Label: ROXICODONE- oxycodone hydrochloride tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 16590-913-72 - Packager: STAT RX USA LLC
- This is a repackaged label.
- Source NDC Code(s): 66479-582
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: CII
- Marketing Status: New Drug Application
Drug Label Information
Updated October 12, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
ROXICODONE® (oxycodone hydrochloride tablets USP) is an opioid analgesic.
Each tablet for oral administration contains 5 mg, 15 mg or 30 mg of oxycodone hydrochloride USP.
Oxycodone hydrochloride is a white, odorless crystalline powder derived from the opium alkaloid, thebaine. Oxycodone hydrochloride dissolves in water (1 g in 6 to 7 mL) and is considered slightly soluble in alcohol (octanol water partition coefficient is 0.7).
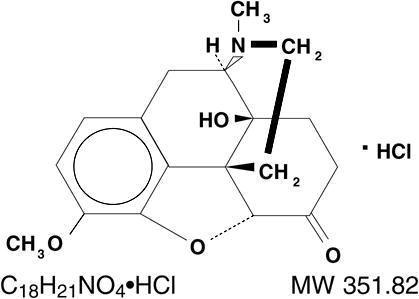
Chemically, oxycodone hydrochloride is 4, 5α-epoxy-14-hydroxy-3-methoxy-17-methylmorphinan-6-one hydrochloride and has the following structural formula:
The 5 mg ROXICODONE® tablet contains the following inactive ingredients: microcrystalline cellulose and stearic acid. The 15 and 30 mg tablets contain the following inactive ingredients: microcrystalline cellulose; sodium starch glycolate; corn starch; lactose; stearic acid; D&C Yellow No. 10 (15 mg tablet); and FD&C Blue No. 2 (15 mg and 30 mg tablets).
The 5 mg, 15 mg and 30 mg tablets contain the equivalent of 4.5 mg, 13.5 mg and 27.0 mg, respectively, of oxycodone free base.
-
CLINICAL PHARMACOLOGY
Pharmacology:
The analgesic ingredient, oxycodone, is a semi-synthetic narcotic with multiple actions qualitatively similar to those of morphine; the most prominent of these involves the central nervous system and organs composed of smooth muscle.
Oxycodone, as the hydrochloride salt, is a pure agonist opioid whose principal therapeutic action is analgesia and has been in clinical use since 1917. Like all pure opioid agonists, there is no ceiling effect to analgesia, such as is seen with partial agonists or non-opioid analgesics. Based upon a single-dose, relative-potency study conducted in humans with cancer pain, 10 to 15 mg of oxycodone given intramuscularly produced an analgesic effect similar to 10 mg of morphine given intramuscularly. Both drugs have a 3 to 4 hour duration of action. Oxycodone retains approximately one half of its analgesic activity when administered orally.
Effects on Central Nervous System: The precise mechanism of the analgesic action is unknown. However, specific CNS opioid receptors for endogenous compounds with opioid-like activity have been identified throughout the brain and spinal cord and play a role in the analgesic effects of this drug. A significant feature of opioid-induced analgesia is that it occurs without loss of consciousness. The relief of pain by morphine-like opioids is relatively selective, in that other sensory modalities, (e.g., touch, vibrations, vision, hearing, etc.) are not obtunded.
Oxycodone produces respiratory depression by direct action on brain stem respiratory centers. The respiratory depression involves both a reduction in the responsiveness of the brain stem respiratory centers to increases in carbon dioxide tension and to electrical stimulation.
Oxycodone depresses the cough reflex by direct effect on the cough center in the medulla. Antitussive effects may occur with doses lower than those usually required for analgesia. Oxycodone causes miosis, even in total darkness. Pinpoint pupils are a sign of opioid overdose but are not pathognomonic (e.g., pontine lesions of hemorrhagic or ischemic origins may produce similar findings). Marked mydriasis rather than miosis may be seen due to hypoxia in overdose situations.
Effects on Gastrointestinal Tract And Other Smooth Muscle: Oxycodone, like other opioid analgesics, produces some degree of nausea and vomiting which is caused by direct stimulation of the chemoreceptor trigger zone (CTZ) located in the medulla. The frequency and severity of emesis gradually diminishes with time.
Oxycodone may cause a decrease in the secretion of hydrochloric acid in the stomach that reduces motility while increasing the tone of the antrum, stomach, and duodenum. Digestion of food in the small intestine is delayed and propulsive contractions are decreased. Propulsive peristaltic waves in the colon are decreased, while tone may be increased to the point of spasm resulting in constipation. Other opioid-induced effects may include a reduction in biliary and pancreatic secretions, spasm of sphincter of Oddi, and transient elevations in serum amylase.
Effects on Cardiovascular System: Oxycodone, in therapeutic doses, produces peripheral vasodilatation (arteriolar and venous), decreased peripheral resistance, and inhibits baroreceptor reflexes. Manifestations of histamine release and/or peripheral vasodilatation may include pruritus, flushing, red eyes, sweating, and/or orthostatic hypotension.
Caution should be used in hypovolemic patients, such as those suffering acute myocardial infarction, because oxycodone may cause or further aggravate their hypotension. Caution should also be used in patients with cor pulmonale who have received therapeutic doses of opioids.
Pharmacodynamics:
The relationship between the plasma level of oxycodone and the analgesic response will depend on the patient's age, state of health, medical condition and extent of previous opioid treatment.
The minimum effective plasma concentration of oxycodone to achieve analgesia will vary widely among patients, especially among patients who have been previously treated with potent agonist opioids. Thus, patients need to be treated with individualized titration of dosage to the desired effect. The minimum effective analgesic concentration of oxycodone for any individual patient may increase with repeated dosing due to an increase in pain and/or development of tolerance.
Pharmacokinetics:
The activity of ROXICODONE® (oxycodone hydrochloride) tablets is primarily due to the parent drug oxycodone. ROXICODONE® tablets are designed to provide immediate release of oxycodone.
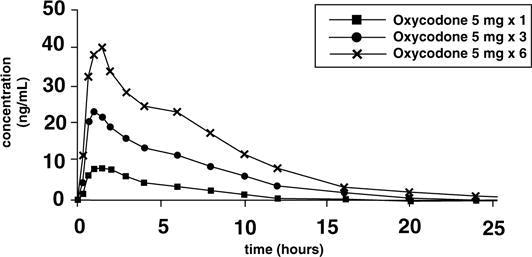
Table 1 Pharmacokinetic Parameters (Mean±SD) Dose\Parameters AUC Cmax Tmax Cmin Cavg Half-Life (ngxhr/mL) (ng/mL) (hr) (ng/mL) (ng/mL) (hr) Single Dose Pharmacokinetics ROXICODONE 133.2±33 22.3±8.2 1.8±1.8 n/a n/a 3.73±0.9 5 mg tabs x 3 ROXICODONE 128.2±35.1 22.2±7.6 1.4±0.7 n/a n/a 3.55±1.0 15 mg tab ROXICODONE Liquid 130.6±34.7 21.1±6.1 1.9±1.5 n/a n/a 3.71±0.8 Concentrate 15 mg oral solution ROXICODONE 268.2±60.7 39.3±14.0 2.6±3.0 n/a n/a 3.85±1.3 30 mg tab Food-Effect, Single Dose ROXICODONE 105±6.2 19.0±3.7 1.25±0.5 n/a n/a 2.9±0.4 10 mg/10 mL oral sol'n (fasted) ROXICODONE 133±25.2 17.7±3.0 2.54±1.2 n/a n/a 3.3±0.5 10 mg/10 mL oral sol'n (fed) Multiple-Dose AUC(72-84) Studies ROXICODONE 113.3±24.0 15.7±3.2 1.3±0.3 7.4±1.8 9.4±2.0 n/a 5 mg tabs q6h x 14 doses ROXICODONE 99.0±24.8 12.9±3.1 1.0±0.3 7.2±2.3 9.7±2.6 n/a 3.33 mg (3.33 mL) oral sol'n. q4h x 21 doses Absorption: About 60% to 87% of an oral dose of oxycodone reaches the systemic circulation in comparison to a parenteral dose. This high oral bioavailability (compared to other oral opioids) is due to lower pre-systemic and/or first-pass metabolism of oxycodone. The relative oral bioavailability of ROXICODONE® 15 mg and 30 mg tablets, compared to the 5 mg ROXICODONE® tablets, is 96% and 101% respectively. ROXICODONE® 15 mg tablets and 30 mg tablets are bioequivalent to the 5 mg ROXICODONE® tablet (see Table 1 for pharmacokinetic parameters). Dose proportionality of oxycodone has been established using the ROXICODONE® 5 mg tablets at doses of 5 mg, 15 mg (three 5 mg tablets) and 30 mg (six 5 mg tablets) based on extent of absorption (AUC) (see Figure 1). It takes approximately 18 to 24 hours to reach steady-state plasma concentrations of oxycodone with ROXICODONE®.
Food Effect: A single-dose food effect study was conducted in normal volunteers using the 5 mg/5 mL solution. The concurrent intake of a high fat meal was shown to enhance the extent (27% increase in AUC), but not the rate of oxycodone absorption from the oral solution (see Table 1). In addition, food caused a delay in Tmax (1.25 to 2.54 hour). Similar effects of food are expected with the 15 mg and 30 mg tablets.
Distribution: Following intravenous administration, the volume of distribution (Vss) for oxycodone was 2.6 L/kg. Plasma protein binding of oxycodone at 37°C and a pH of 7.4 was about 45%. Oxycodone has been found in breast milk (see PRECAUTIONS-Nursing Mothers).
Metabolism: Oxycodone hydrochloride is extensively metabolized to noroxycodone, oxymorphone, and their glucuronides. The major circulating metabolite is noroxycodone with an AUC ratio of 0.6 relative to that of oxycodone. Oxymorphone is present in the plasma only in low concentrations. The analgesic activity profile of other metabolites is not known at present.
The formation of oxymorphone, but not noroxycodone, is mediated by CYP2D6 and as such its formation can, in theory, be affected by other drugs (see PRECAUTIONS-Drug Interactions).
Elimination: Oxycodone and its metabolites are excreted primarily via the kidney. The amounts measured in the urine have been reported as follows: free oxycodone up to 19%; conjugated oxycodone up to 50%; free oxymorphone 0%; conjugated oxymorphone ≤ 14%; both free and conjugated noroxycodone have been found in the urine but not quantified. The total plasma clearance was 0.8 L/min for adults. Apparent elimination half-life of oxycodone following the administration of ROXICODONE® was 3.5 to 4 hours.
Special Populations:
Geriatric: Population pharmacokinetic studies conducted with ROXICODONE®, indicated that the plasma concentrations of oxycodone did not appear to be increased in patients over the age of 65.
Gender: Population pharmacokinetic analyses performed in the clinical study support the lack of gender effect on the pharmacokinetics of oxycodone from ROXICODONE®.
Race: Population pharmacokinetic analyses support the lack of race effect on oxycodone pharmacokinetics after administration of ROXICODONE®, but these data should be interpreted conservatively, since the majority of patients enrolled into the studies were Caucasians (94%).
Renal Insufficiency: In a clinical trial supporting the development of ROXICODONE®, too few patients with decreased renal function were evaluated to study these potential differences. In previous studies, patients with renal impairment (defined as a creatinine clearance < 60 mL/min) had concentrations of oxycodone in the plasma that were higher than in subjects with normal renal function. Based on information available on the metabolism and excretion of oxycodone, dose initiation in patients with renal impairment should follow a conservative approach. Dosages should be adjusted according to the clinical situation.
Hepatic Failure: In a clinical trial supporting the development of ROXICODONE®, too few patients with decreased hepatic function were evaluated to study these potential differences. However, since oxycodone is extensively metabolized, its clearance may decrease in hepatic failure patients. Dose initiation in patients with hepatic impairment should follow a conservative approach. Dosages should be adjusted according to the clinical situation.
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
ROXICODONE® is contraindicated in patients with known hypersensitivity to oxycodone, or in any situation where opioids are contraindicated. This includes patients with significant respiratory depression (in unmonitored settings or the absence of resuscitative equipment) and patients with acute or severe bronchial asthma or hypercarbia. ROXICODONE® is contraindicated in any patient who has or is suspected of having paralytic ileus.
-
WARNINGS
Respiratory Depression:
Respiratory depression is the chief hazard from all opioid agonist preparations. Respiratory depression occurs most frequently in elderly or debilitated patients, usually following large initial doses in non-tolerant patients, or when opioids are given in conjunction with other agents that depress respiration.
ROXICODONE® should be used with extreme caution in patients with significant chronic obstructive pulmonary disease or cor pulmonale, and in patients having substantially decreased respiratory reserve, hypoxia, hypercapnia, or pre-existing respiratory depression. In such patients, even usual therapeutic doses of ROXICODONE® may decrease respiratory drive to the point of apnea. In these patients alternative non-opioid analgesics should be considered, and opioids should be employed only under careful medical supervision at the lowest effective dose.
Hypotensive Effect:
ROXICODONE®, like all opioid analgesics, may cause severe hypotension in an individual whose ability to maintain blood pressure has been compromised by a depleted blood volume, or after concurrent administration with drugs such as phenothiazines or other agents which compromise vasomotor tone. ROXICODONE® may produce orthostatic hypotension in ambulatory patients. ROXICODONE®, like all opioid analgesics, should be administered with caution to patients in circulatory shock, since vasodilatation produced by the drug may further reduce cardiac output and blood pressure.
Head Injury and Increased Intracranial Pressure:
The respiratory depressant effects of narcotics and their capacity to elevate cerebrospinal fluid pressure may be markedly exaggerated in the presence of head injury, other intracranial lesions or a pre-existing increase in intracranial pressure. Furthermore, narcotics produce adverse reactions which may obscure the clinical course of patients with head injuries.
-
PRECAUTIONS
General:
ROXICODONE® tablets are intended for use in patients who require oral pain therapy with an opioid agonist. As with any opioid analgesic, it is critical to adjust the dosing regimen individually for each patient (see DOSAGE AND ADMINISTRATION).
Selection of patients for treatment with ROXICODONE® should be governed by the same principles that apply to the use of other potent opioid analgesics. Opioid analgesics given on a fixed-dosage schedule have a narrow therapeutic index in certain patient populations, especially when combined with other drugs, and should be reserved for cases where the benefits of opioid analgesia outweigh the known risks of respiratory depression, altered mental state, and postural hypotension. Physicians should individualize treatment in every case, using nonopioid analgesics, prn opioids and /or combination products, and chronic opioid therapy with drugs such as ROXICODONE® (oxycodone hydrochloride) in a progressive plan of pain management such as outlined by the World Health Organization, the Agency for Health Care Policy and Research, and the American Pain Society.
Use of ROXICODONE® is associated with increased potential risks and should be used only with caution in the following conditions: acute alcoholism; adrenocortical insufficiency (e.g., Addison's disease); convulsive disorders; CNS depression or coma; delirium tremens; debilitated patients; kyphoscoliosis associated with respiratory depression; myxedema or hypothyroidism; prostatic hypertrophy or urethral stricture; severe impairment of hepatic, pulmonary or renal function; and toxic psychosis.
The administration of ROXICODONE®, like all opioid analgesics, may obscure the diagnosis or clinical course in patients with acute abdominal conditions. Oxycodone may aggravate convulsions in patients with convulsive disorders, and all opioids may induce or aggravate seizures in some clinical settings.
Tolerance and Physical Dependence:
Physical dependence and tolerance are not unusual during chronic opioid therapy. Significant tolerance should not occur in most patients treated with the lowest doses of oxycodone. It should be expected, however, that a fraction of patients will develop some degree of tolerance and require progressively higher dosages of ROXICODONE® to maintain pain control during chronic treatment. The dosage should be selected according to the patient's individual analgesic response and ability to tolerate side effects. Tolerance to the analgesic effects of opioids is usually paralleled by tolerance to side effects except for constipation.
Physical dependence results in withdrawal symptoms in patients who abruptly discontinue the drug or may be precipitated through the administration of drugs with opioid antagonist activity. If ROXICODONE® is abruptly discontinued in a physically dependent patient, an abstinence syndrome may occur (see DRUG ABUSE AND DEPENDENCE). If signs and symptoms of withdrawal occur, patients should be treated by reinstitution of opioid therapy followed by gradual tapered dose reduction of ROXICODONE® combined with symptomatic support (see DOSAGE AND ADMINISTRATION: Cessation of Therapy).
Use In Pancreatic/Biliary Tract Disease:
ROXICODONE® may cause spasm of the sphincter of Oddi and should be used with caution in patients with biliary tract disease, including acute pancreatitis. Opioids like ROXICODONE® may cause increases in the serum amylase level.
Information for Patients/Caregivers:
If clinically advisable, patients (or their caregivers) receiving ROXICODONE® (oxycodone hydrochloride) tablets should be given the following information by the physician, nurse, pharmacist or caregiver:
- Patients should be advised to report episodes of breakthrough pain and adverse experiences occurring during therapy. Individualization of dosage is essential to make optimal use of this medication.
- Patients should be advised not to adjust the dose of ROXICODONE® without consulting the prescribing professional.
- Patients should be advised that ROXICODONE® may impair mental and/or physical ability required for the performance of potentially hazardous tasks (e.g., driving, operating heavy machinery).
- Patients should not combine ROXICODONE® with alcohol or other central nervous system depressants (sleep aids, tranquilizers) except by the orders of the prescribing physician, because additive effects may occur.
- Women of childbearing potential who become, or are planning to become pregnant, should be advised to consult their physician regarding the effects of analgesics and other drug use during pregnancy on themselves and their unborn child.
- Patients should be advised that ROXICODONE® is a potential drug of abuse. They should protect it from theft, and it should never be given to anyone other than the individual for whom it was prescribed.
- Patients should be advised that if they have been receiving treatment with ROXICODONE® for more than a few weeks and cessation of therapy is indicated, it may be appropriate to taper the ROXICODONE® dose, rather than abruptly discontinue it, due to the risk of precipitating withdrawal symptoms. Their physician can provide a dose schedule to accomplish a gradual discontinuation of the medication.
Drug Interactions:
Oxycodone is metabolized in part to oxymorphone via the cytochrome p450 isoenzyme CYP2D6. While this pathway may be blocked by a variety of drugs (e.g., certain cardiovascular drugs and antidepressants), such blockade has not yet been shown to be of clinical significance with this agent. However, clinicians should be aware of this possible interaction.
Neuromuscular Blocking Agents: Oxycodone, as well as other opioid analgesics, may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression.
CNS Depressants: Patients receiving narcotic analgesics, general anesthetics, phenothiazines, other tranquilizers, sedative-hypnotics or other CNS depressants (including alcohol) concomitantly with ROXICODONE® may exhibit an additive CNS depression. Interactive effects resulting in respiratory depression, hypotension, profound sedation, or coma may result if these drugs are taken in combination with the usual dosage of ROXICODONE®. When such combined therapy is contemplated, the dose of one or both agents should be reduced.
Mixed Agonist/Antagonist Opioid Analgesics: Agonist/antagonist analgesics (i.e., pentazocine, nalbuphine, butorphanol and buprenorphine) should be administered with caution to patients who have received or are receiving a course of therapy with a pure opioid agonist analgesic such as ROXICODONE®. In this situation, mixed agonist/antagonist analgesics may reduce the analgesic effect of ROXICODONE® and/or may precipitate withdrawal symptoms in these patients.
Monoamine Oxidase Inhibitors (MAOIs): MAOIs have been reported to intensify the effects of at least one opioid drug causing anxiety, confusion and significant depression of respiration or coma. The use of ROXICODONE® is not recommended for patients taking MAOIs or within 14 days of stopping such treatment.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Long-term studies have not been performed in animals to evaluate the carcinogenic potential of ROXICODONE® or oxycodone. The possible effects on male or female fertility have not been studied in animals.
Oxycodone hydrochloride was genotoxic in an in vitro mouse lymphoma assay in the presence of metabolic activation. There was no evidence of genotoxic potential in an in vitro bacterial reverse mutation assay (Salmonella typhimurium and Escherichia coli) or in an assay for chromosomal aberrations (in vivo mouse bone marrow micronucleus assay).
Pregnancy:
Teratogenic Effects: Category B: Reproduction studies in Sprague-Dawley rats and New Zealand rabbits revealed that when oxycodone was administered orally at doses up to 16 mg/kg (approximately 2 times the daily oral dose of 90 mg for adults on a mg/m2 basis) and 25 mg/kg (approximately 5 times the daily oral dose of 90 mg on a mg/m2 basis), respectively was not teratogenic or embryo-fetal toxic. There are no adequate and well controlled studies of oxycodone in pregnant women. Because animal reproductive studies are not always predictive of human responses, ROXICODONE® should be used during pregnancy only if potential benefit justifies the potential risk to the fetus.
Labor and Delivery:
ROXICODONE® is not recommended for use in women during or immediately prior to labor. Occasionally, opioid analgesics may prolong labor through actions which temporarily reduce the strength, duration and frequency of uterine contractions. Neonates, whose mothers received opioid analgesics during labor, should be observed closely for signs of respiratory depression. A specific narcotic antagonist, naloxone, should be available for reversal of narcotic-induced respiratory depression in the neonate.
Nursing Mothers:
Oxycodone has been detected in breast milk. Withdrawal symptoms can occur in breast-feeding infants when maternal administration of an opioid analgesic is stopped. Ordinarily, nursing should not be undertaken while a patient is receiving ROXICODONE® since oxycodone may be excreted in milk.
Geriatric Use:
Of the total number of subjects in clinical studies of ROXICODONE®, 20.8% (112/538) were 65 and over, while 7.2% (39/538) were 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Hepatic Impairment:
Since oxycodone is extensively metabolized, its clearance may decrease in hepatic failure patients. Dose initiation in patients with hepatic impairment should follow a conservative approach. Dosages should be adjusted according to the clinical situation.
Renal Impairment:
Published data reported that elimination of oxycodone was impaired in end-stage renal failure. Mean elimination half-life was prolonged in uremic patients due to increased volume of distribution and reduced clearance. Dose initiation should follow a conservative approach. Dosages should be adjusted according to the clinical situation.
-
ADVERSE REACTIONS
ROXICODONE® tablets have been evaluated in open label clinical trials in patients with cancer and nonmalignant pain. ROXICODONE® tablets are associated with adverse experiences similar to those seen with other opioids.
Serious adverse reactions that may be associated with ROXICODONE® therapy in clinical use are those observed with other opioid analgesics and include: respiratory depression, respiratory arrest, circulatory depression, cardiac arrest, hypotension, and/or shock (see OVERDOSE, WARNINGS).
The less severe adverse events seen on initiation of therapy with ROXICODONE® are also typical opioid side effects. These events are dose dependent, and their frequency depends on the clinical setting, the patient's level of opioid tolerance, and host factors specific to the individual. They should be expected and managed as a part of opioid analgesia. The most frequent of these include nausea, constipation, vomiting, headache, and pruritus.
In many cases the frequency of adverse events during initiation of opioid therapy may be minimized by careful individualization of starting dosage, slow titration and the avoidance of large rapid swings in plasma concentration of the opioid. Many of these adverse events will abate as therapy is continued and some degree of tolerance is developed, but others may be expected to remain throughout therapy.
In all patients for whom dosing information was available (n=191) from the open-label and double-blind studies involving ROXICODONE®, the following adverse events were recorded in ROXICODONE® treated patients with an incidence ≥ 3%. In descending order of frequency they were: nausea, constipation, vomiting, headache, pruritus, insomnia, dizziness, asthenia, and somnolence.
The following adverse experiences occurred in less than 3% of patients involved in clinical trials with oxycodone:
Body as a Whole: abdominal pain, accidental injury, allergic reaction, back pain, chills and fever, fever, flu syndrome, infection, neck pain, pain, photosensitivity reaction, and sepsis.
Cardiovascular: deep thrombophlebitis, heart failure, hemorrhage, hypotension, migraine, palpitation, and tachycardia.
Digestive: anorexia, diarrhea, dyspepsia, dysphagia, gingivitis, glossitis, and nausea and vomiting.
Hemic and Lymphatic: anemia and leukopenia.
Metabolic and Nutritional: edema, gout, hyperglycemia, iron deficiency anemia and peripheral edema.
Musculoskeletal: arthralgia, arthritis, bone pain, myalgia and pathological fracture.
Nervous: agitation, anxiety, confusion, dry mouth, hypertonia, hypesthesia, nervousness, neuralgia, personality disorder, tremor, and vasodilation.
Respiratory: bronchitis, cough increased, dyspnea, epistaxis, laryngismus, lung disorder, pharyngitis, rhinitis, and sinusitis.
Skin and Appendages: herpes simplex, rash, sweating, and urticaria.
Special Senses: amblyopia.
Urogenital: urinary tract infection.
-
DRUG ABUSE AND DEPENDENCE
Controlled Substance:
ROXICODONE® contains oxycodone, a mu-agonist opioid of the morphine type and is a Schedule II controlled substance. ROXICODONE®, like other opioids used in analgesia, can be abused and is subject to criminal diversion.
Abuse:
Drug addiction is characterized by compulsive use, use for non-medical purposes, and continued use despite harm or risk of harm. Drug addiction is a treatable disease, utilizing a multi-disciplinary approach, but relapse is common.
“Drug-seeking” behavior is very common in addicts and drug abusers. Drug-seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing or referral, repeated “loss” of prescriptions, tampering with prescriptions and reluctance to provide prior medical records or contact information for other treating physician(s). “Doctor shopping” to obtain additional prescriptions is common among drug abusers and people suffering from untreated addiction.
Abuse and addiction are separate and distinct from physical dependence and tolerance. Physicians should be aware that addiction may not be accompanied by concurrent tolerance and symptoms of physical dependence. In addition, abuse of opioids can occur in the absence of true addiction and is characterized by misuse for nonmedical purposes, often in combination with other psychoactive substances. Careful record-keeping of prescribing information, including quantity, frequency, and renewal requests is strongly advised.
ROXICODONE® is intended for oral use only. Abuse of ROXICODONE® poses a risk of overdose and death. The risk is increased with concurrent abuse of alcohol and other substances. Parenteral drug abuse is commonly associated with transmission of infectious diseases such as hepatitis and HIV.
Proper assessment of the patient, proper prescribing practices, periodic re-evaluation of therapy, and proper dispensing and storage are appropriate measures that help to limit abuse of opioid drugs.
Infants born to mothers physically dependent on opioids will also be physically dependent and may exhibit respiratory difficulties and withdrawal symptoms.
Dependence:
Tolerance is the need for increasing doses of opioids to maintain a defined effect such as analgesia (in the absence of disease progression or other external factors). Physical dependence is manifested by withdrawal symptoms after abrupt discontinuation of a drug or upon administration of an antagonist. Physical dependence and tolerance are not unusual during chronic opioid therapy.
The opioid abstinence or withdrawal syndrome is characterized by some or all of the following: restlessness, lacrimation, rhinorrhea, yawning, perspiration, chills, myalgia, and mydriasis. Other symptoms also may develop, including irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate. In general, opioids should not be abruptly discontinued.
-
OVERDOSAGE
Signs and Symptoms:
Acute overdose with ROXICODONE® can be manifested by respiratory depression, somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, constricted pupils, bradycardia, hypotension, and death.
Treatment:
To treat ROXICODONE® overdose, primary attention should be given to the re-establishment of a patent airway and institution of assisted or controlled ventilation. Supportive measures (including oxygen and vasopressors) should be employed in the management of circulatory shock and pulmonary edema accompanying overdose as indicated. Cardiac arrest or arrhythmias may require cardiac massage or defibrillation.
The narcotic antagonists, naloxone or nalmefene, are specific antidotes for opioid overdose. Opioid antagonists should not be administered in the absence of clinically significant respiratory or circulatory depression secondary to ROXICODONE® overdose. If needed the appropriate dose of naloxone hydrochloride or nalmefene should be administered simultaneously with efforts at respiratory resuscitation (see package insert for each drug for the details). Since the duration of action of oxycodone may exceed that of the antagonist, the patient should be kept under continued surveillance and repeated doses of the antagonist should be administered as needed to maintain adequate respiration. Gastric emptying may be useful in removing unabsorbed drug.
Opioid antagonists should be administered cautiously to persons who are suspected to be physically dependent on any opioid agonist, including oxycodone (see Opioid-Tolerant Individuals).
Opioid-Tolerant Individuals: In an individual physically dependent on opioids, administration of a usual dose of antagonist will precipitate an acute withdrawal. The severity of the withdrawal syndrome produced will depend on the degree of physical dependence and the dose of the antagonist administered. Use of an opioid antagonist should be reserved for cases where such treatment is clearly needed. If it is necessary to treat serious respiratory depression in the physically dependent patient, administration of the antagonist should be begun with care and by titration with smaller than usual doses.
-
DOSAGE AND ADMINISTRATION
ROXICODONE® is intended for the management of moderate to severe pain in patients who require treatment with an oral opioid analgesic. The dose should be individually adjusted according to severity of pain, patient response and patient size. If the pain increases in severity, if analgesia is not adequate, or if tolerance occurs, a gradual increase in dosage may be required.
Patients who have not been receiving opioid analgesics should be started on ROXICODONE® in a dosing range of 5 to 15 mg every 4 to 6 hours as needed for pain. The dose should be titrated based upon the individual patient's response to their initial dose of ROXICODONE®. Patients with chronic pain should have their dosage given on an around-the-clock basis to prevent the reoccurrence of pain rather than treating the pain after it has occurred. This dose can then be adjusted to an acceptable level of analgesia taking into account side effects experienced by the patient.
For control of severe chronic pain, ROXICODONE® should be administered on a regularly scheduled basis, every 4-6 hours, at the lowest dosage level that will achieve adequate analgesia.
As with any potent opioid, it is critical to adjust the dosing regimen for each patient individually, taking into account the patient's prior analgesic treatment experience. Although it is not possible to list every condition that is important to the selection of the initial dose of ROXICODONE®, attention should be given to: 1) the daily dose, potency, and characteristics of a pure agonist or mixed agonist/antagonist the patient has been taking previously, 2) the reliability of the relative potency estimate to calculate the dose of oxycodone needed, 3) the degree of opioid tolerance, 4) the general condition and medical status of the patient, and 5) the balance between pain control and adverse experiences.
Conversion From Fixed-Ratio Opioid/Acetaminophen, Opioid/Aspirin, or Opioid/Nonsteroidal Combination Drugs:
When converting patients from fixed ratio opioid/non-opioid drug regimens a decision should be made whether or not to continue the non-opioid analgesic. If a decision is made to discontinue the use of non-opioid analgesic, it may be necessary to titrate the dose of ROXICODONE® in response to the level of analgesia and adverse effects afforded by the dosing regimen. If the non-opioid regimen is continued as a separate single entity agent, the starting dose ROXICODONE® should be based upon the most recent dose of opioid as a baseline for further titration of oxycodone. Incremental increases should be gauged according to side effects to an acceptable level of analgesia.
Patients Currently on Opioid Therapy:
If a patient has been receiving opioid-containing medications prior to taking ROXICODONE®, the potency of the prior opioid relative to oxycodone should be factored into the selection of the total daily dose (TDD) of oxycodone.
In converting patients from other opioids to ROXICODONE® close observation and adjustment of dosage based upon the patient's response to ROXICODONE® is imperative. Administration of supplemental analgesia for breakthrough or incident pain and titration of the total daily dose of ROXICODONE® may be necessary, especially in patients who have disease states that are changing rapidly.
Maintenance of Therapy:
Continual re-evaluation of the patient receiving ROXICODONE® is important, with special attention to the maintenance of pain control and the relative incidence of side effects associated with therapy. If the level of pain increases, effort should be made to identify the source of increased pain, while adjusting the dose as described above to decrease the level of pain.
During chronic therapy, especially for non-cancer-related pain (or pain associated with other terminal illnesses), the continued need for the use of opioid analgesics should be re-assessed as appropriate.
Cessation of Therapy:
When a patient no longer requires therapy with ROXICODONE® or other opioid analgesics for the treatment of their pain, it is important that therapy be gradually discontinued over time to prevent the development of an opioid abstinence syndrome (narcotic withdrawal). In general, therapy can be decreased by 25% to 50% per day with careful monitoring for signs and symptoms of withdrawal (see DRUG ABUSE AND DEPENDENCE section for description of the signs and symptoms of withdrawal). If the patient develops these signs or symptoms, the dose should be raised to the previous level and titrated down more slowly, either by increasing the interval between decreases, decreasing the amount of change in dose, or both. It is not known at what dose of ROXICODONE® that treatment may be discontinued without risk of the opioid abstinence syndrome.
-
HOW SUPPLIED
ROXICODONE® (oxycodone hydrochloride tablets USP) are available as follows:
5 mg white scored tablets (Identified 54 582)
[Embossed 54 582 on one side]
NDC 66479-580-25: Unit dose, 25 tablets per card,
4 cards per shipperNDC 66479-580-10: Bottles of 100 tablets
15 mg green tablets scored (Identified 54 710)
[Embossed 54 710 on one side]
NDC 66479-581-10: Bottles of 100 tablets30 mg blue tablets scored (Identified 54 199)
[Embossed 54 199 on one side]
NDC 66479-582-10: Bottles of 100 tabletsDEA Order Form Required
Dispense in a tight, light-resistant container.
Protect from moisture.
Store at 25°C (77°F); excursions are permitted to
15°-30°C (59°-86°F) [see USP Controlled Room Temperature]ROXICODONE® is a registered trademark of Xanodyne Pharmaceuticals, Inc.
© 2009 Xanodyne Pharmaceuticals, Inc.
Marketed by:
Xanodyne®
Pharmaceuticals, Inc.
Newport, KY 41071PI - 580/581/582-A
Rev. 05-2009To request medical information or to report suspected adverse reactions, contact Xanodyne Medical Affairs at 1-877-773-7793.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ROXICODONE
oxycodone hydrochloride tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:16590-913(NDC:66479-582) Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength oxycodone hydrochloride (UNII: C1ENJ2TE6C) (oxycodone - UNII:CD35PMG570) oxycodone hydrochloride 30 mg Inactive Ingredients Ingredient Name Strength Cellulose, Microcrystalline (UNII: OP1R32D61U) Stearic Acid (UNII: 4ELV7Z65AP) FD&C Blue No. 2 (UNII: L06K8R7DQK) Sodium Starch Glycolate Type A Potato (UNII: 5856J3G2A2) Starch, Corn (UNII: O8232NY3SJ) Lactose Monohydrate (UNII: EWQ57Q8I5X) Water (UNII: 059QF0KO0R) Product Characteristics Color blue (BLUE) Score 2 pieces Shape ROUND (ROUND) Size 6mm Flavor Imprint Code 54;199 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:16590-913-72 120 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021011 08/31/2000 Labeler - STAT RX USA LLC (786036330) Establishment Name Address ID/FEI Business Operations STAT RX USA LLC 786036330 relabel, repack