Label: ALLERGY RELIEF- fexofenadine hcl tablet, coated
- NDC Code(s): 81522-613-30
- Packager: FSA Store Inc. (Caring Mills)
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated January 24, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
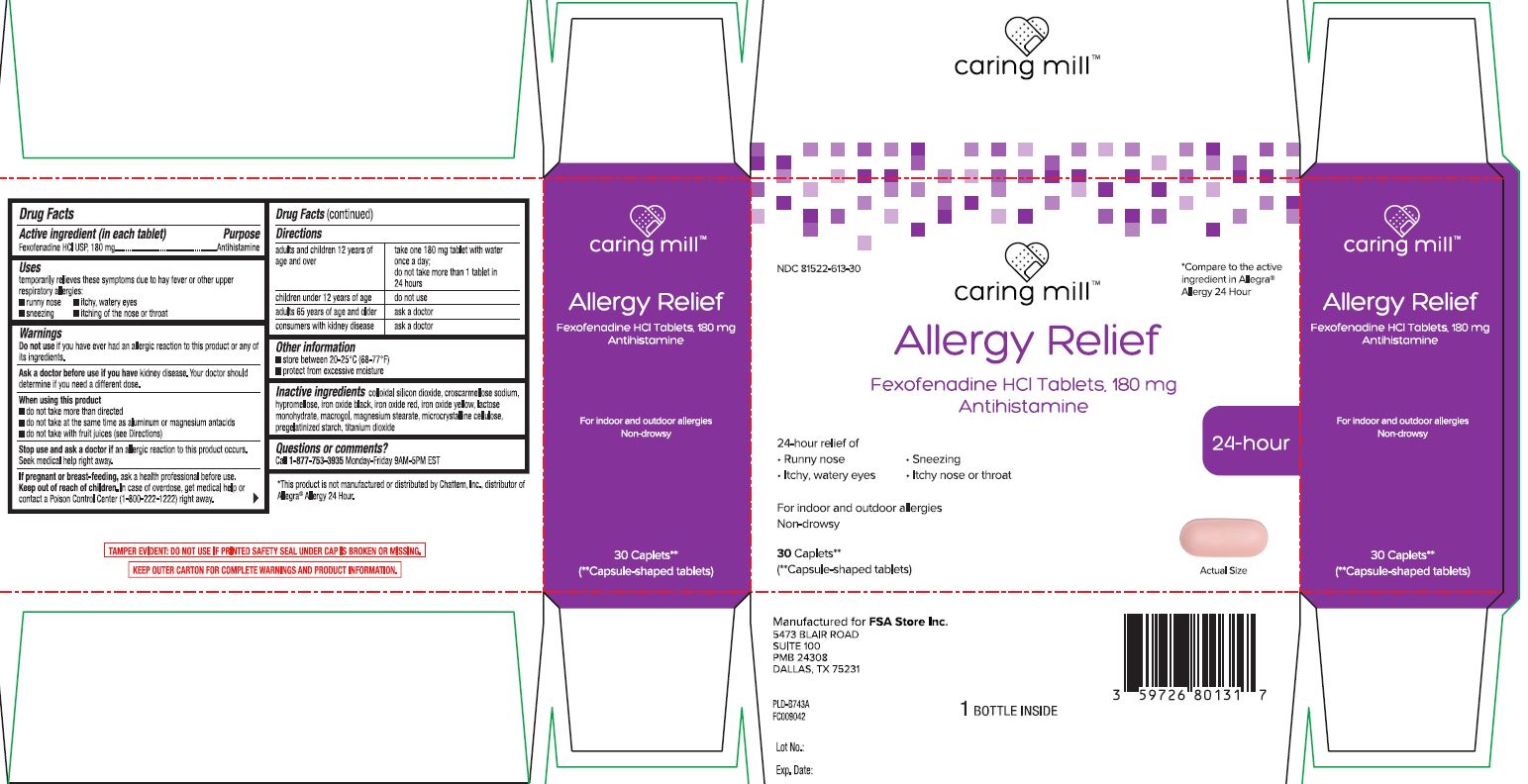
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
kidney disease. Your doctor should determine if you need a different dose.
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
*Compare to the active ingredient in Allegra® Allergy 24 Hour
Allergy Relief
Fexofenadine HCl Tablets, 180 mg
Antihistamine
24-Hour relief of
- Runny nose
- Sneezing
- Itchy, watery eyes
- Itchy nose or throat
For Indoor & outdoor allergies
Non-drowsy
Caplets**
(**Capsule-shaped tablets)
*This product is not manufactured or distributed by Chattem Inc., distributor of Allegra® Allergy 24 Hour.
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
Manufactured for FSA Stores Inc.
5473 BLAIR ROAD
SUITE 100
PMB 24308
DALLAS, TX 75231
- Product Label
-
INGREDIENTS AND APPEARANCE
ALLERGY RELIEF
fexofenadine hcl tablet, coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81522-613 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FEXOFENADINE HYDROCHLORIDE (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) FEXOFENADINE HYDROCHLORIDE 180 mg Inactive Ingredients Ingredient Name Strength FERROSOFERRIC OXIDE (UNII: XM0M87F357) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSES (UNII: 3NXW29V3WO) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color white (peach) Score no score Shape CAPSULE Size 17mm Flavor Imprint Code G6 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81522-613-30 1 in 1 BOX 10/08/2021 1 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA211075 10/08/2021 Labeler - FSA Store Inc. (Caring Mills) (049283340)