Label: SPF BESTSELLERS STARTER KIT- avobenzone, homosalate, octisalate, octocrylene kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 82384-0030-1, 82384-0031-1, 82384-0032-1, 82384-0033-1 - Packager: System Beauty, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 15, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
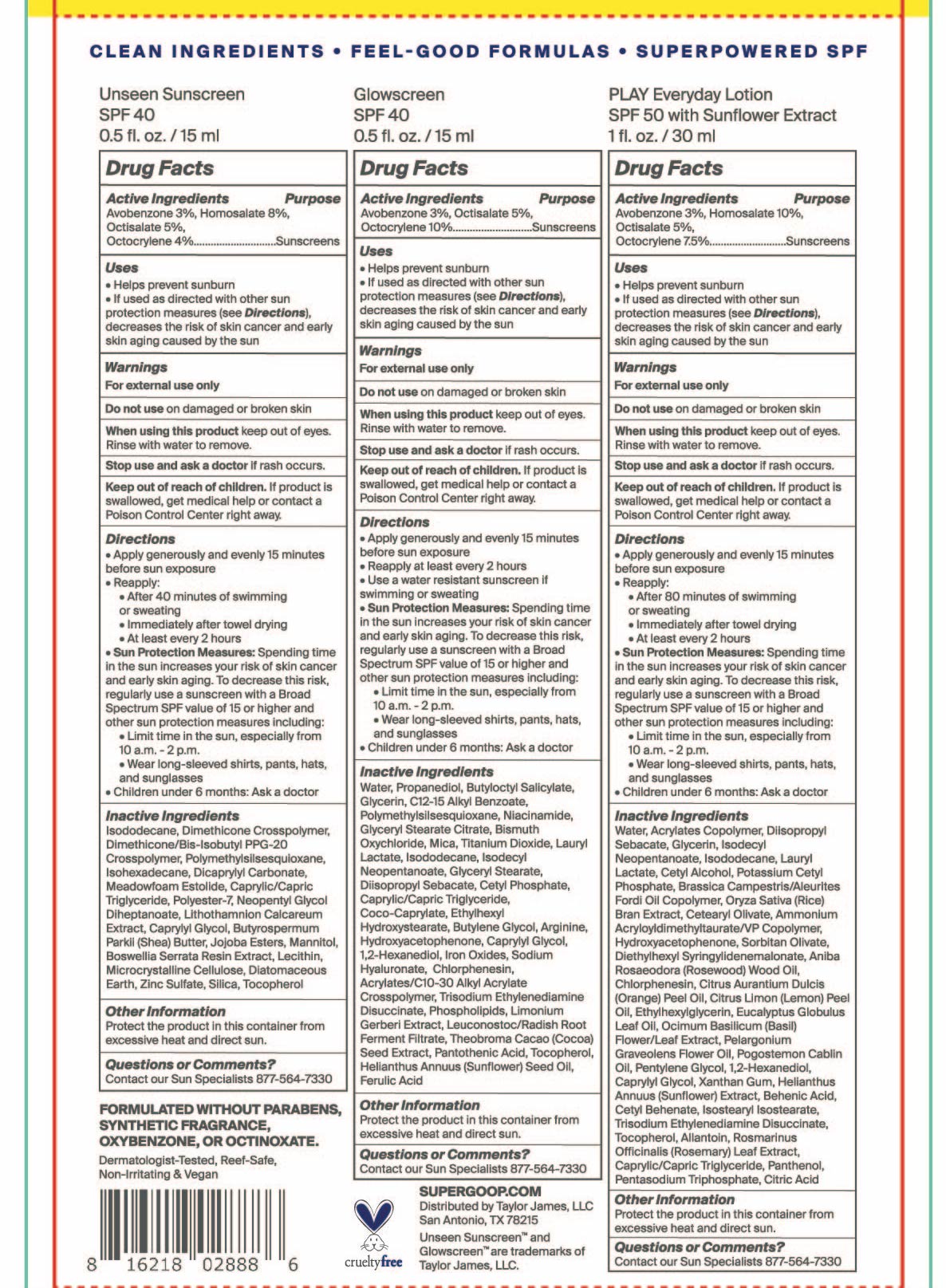
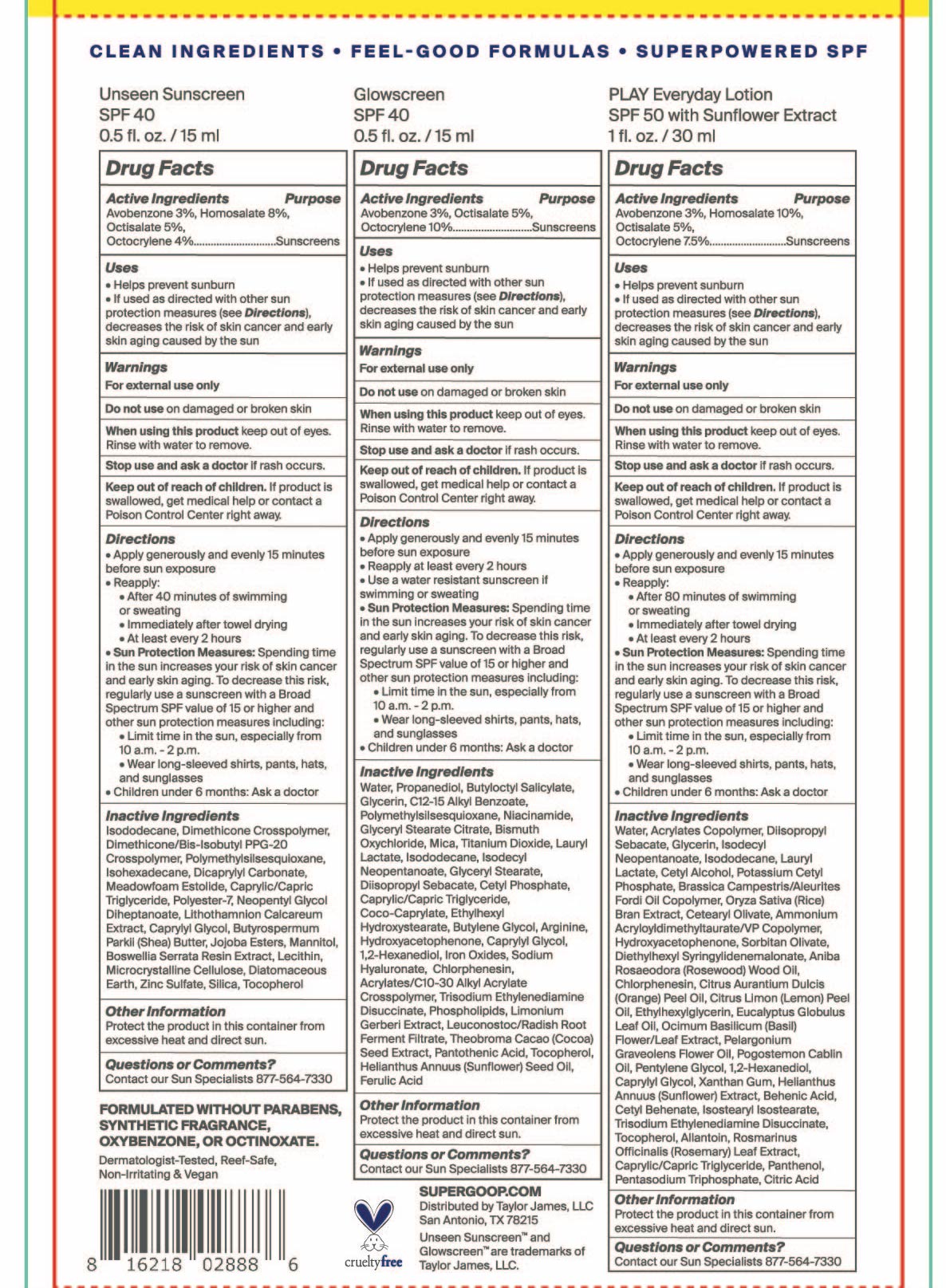
- Unseen Sunscreen Active Ingredient
- Glowscreen SPF 40
- Play Everyday Sunscreen SPF 50 with Sunflower Extract Active ingredients
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
-
Unseen Sunscreen SPF 40 Directions
Directions
- apply generously and evenly 15 minutes before sun exposure
- reapply:
after 40 minutes of swimming or sweating
immediately after towel drying
at least every 2 hours
- Sun Protection Measures Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- Limit time in the sun, especially from 10 a.m.-2 p.m.
- Wear long-sleeved shirts, pants, hats, and sunglasses
- Children under 6 months: ask a doctor
-
Glowscreen SPF 40 Directions
Directions
- Apply generously and evenly 15 minutes before sun exposure
- Reapply at least every 2 hours
Use a water resistant sunscreen if swimming or sweating
Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
Limit time in the sun, especially from 10a.m. – 2p.m.- Wear long-sleeved shirts, pants, hats, and sunglasses
- Children under 6 months: Ask a doctor
-
Play Everyday Sunscreen SPF 50 with Sunflower Extract directions
Directions
- Apply liberally 15 minutes before sun exposure
Reapply:
- after 80 minutes of swimming or sweating
- immediately after towel drying
- at least every 2 hours
- Sun Protection Measures. Spending time in the sun increases your risk of skin
cancer and early skin aging. To decrease this risk, regularly use a sunscreen
with broad spectrum SPF of 15 or higher and other sun protection measures
including:- limit time in the sun, especially from 10 a.m. - 2 p.m.
- Wear Long-sleeved shirts, pants, hats, and sunglasses
- Children under 6 months: Ask a doctor
-
Unseen Sunscreen Inactive Ingredients
Inactive Ingredients
Isododecane, Dimethicone Crosspolymer, Dimethicone/Bis-Isobutyl PPG-20 Crosspolymer, Polymethylsilsesquioxane, Isohexadecane, Dicrapylyl Carbonate, Meadowfoam Estolide, Caprylic/Capric Triglyceride, Polyester-7, Neopentyl Glycol Diheptanoate, Lithothamnion Calcareum Extract,Butyrospermum Parkii (Shea) Butter, Jojoba Esters, Mannitol, Boswellia Serrata Resin Extract, Lecithin, Microcrystalline Cellulose, Diatomaceous Earth, Zinc Sulfate, Silica, Tocopherol
-
Glowscreen SPF 40 inactive ingredients
Inactive Ingredients
Water, Propanediol, Butyloctyl Salicylate, Glycerin, C12-15 Alkyl Benzoate, Polymethylsilsesquioxane, Niacinamide, Glyceryl Stearate Citrate, Bismuth Oxychloride, Mica, Titanium Dioxide, Lauryl Lactate, Isododecane, Isodecyl Neopentanoate, Glyceryl Stearate, Diisopropyl Sebacate, Cetyl Phosphate, Caprylic/Capric Triglyceride, Coco-Caprylate, Ethylhexyl Hydroxystearate, Butylene Glycol, Arginine, Hydroxyacetophenone, Caprylyl Glycol, 1,2-Hexanediol, Iron Oxides, Sodium Hyaluronate, Chlorphenesin, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Trisodium Ethylenediamine Disuccinate, Phospholipids, Limonium Gerberi Extract, Leuconostoc/Radish Root Ferment Filtrate, Theobroma Cacao (Cocoa) Seed Extract, Pantothenic Acid, Tocopherol, Helianthus Annuus (sunflower) Seed Oil, Ferulic Acid -
Play Everyday Lotion SPF 50 with Sunflower extract
Inactive Ingredients
Water, Acrylates Copolymer, Diisopropyl Sebacate, Glycerin, Isodecyl Neopentanoate, Isododecane, Lauryl Lactate, Cetyl Alcohol, Potassium Cetyl Phosphate, Brassica Camprestris/Aleurites Fordi Copolymer, Oryza Sativa (Rice) Bran Extract, Cetearyl Olivate, Ammonium Acryloyldimethyltaurate/VP Copolymer, Hydroxyacetophenone, Sorbitan Olivate, Diethylhexyl Syringylidenemalonate, Aniba Rosaeodora (Rosewood) Wood Oil, Chlorphenesin, Citrus Aurantium Dulcis (Orange) Peel Oil, Citrus Limon (Lemon) Peel Oil, Ethylhexylglycerin, Eucalyptus Globulus Leaf Oil, Ocimum Bascilicum (Basil) Flower/Leaf Extract, Pelargonium Graveolens Flower Oil, Pogostemon Cablin Oil, Pentylene Glycol, 1,2-hexanediol, Caprylyl Glycol, Xanthan Gum, Helianthus Annuus (Sunflower) Extract, Behenic Acid, Cetyl Behenate, Isostearyl Isostearate, Trisodium Ethylenediamene Disuccinate, Tocopherol, Allantoin, Rosmarinus Officinalis (Rosemary) Leaf Extract, Caprylic/Capric Triglyceride,Panthenol, Pentasodium Triphosphate, Citric Acid
- SPF Bestsellers Starter Kit
-
INGREDIENTS AND APPEARANCE
SPF BESTSELLERS STARTER KIT
avobenzone, homosalate, octisalate, octocrylene kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82384-0030 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82384-0030-1 1 in 1 KIT; Type 1: Convenience Kit of Co-Package 12/17/2021 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 15 mL Part 2 1 BOTTLE 15 mL Part 3 1 BOTTLE 30 mL Part 1 of 3 UNSEEN SUNSCREEN BROAD SPECTRUM SPF 40
avobenzone, homosalate, octocrylene, octisalate creamProduct Information Item Code (Source) NDC:82384-0031 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 8 g in 100 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 100 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 4 g in 100 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5 g in 100 mL Inactive Ingredients Ingredient Name Strength POLYESTER-7 (UNII: 0841698D2F) NEOPENTYL GLYCOL DIHEPTANOATE (UNII: 5LKW3C543X) ZINC SULFATE (UNII: 89DS0H96TB) TOCOPHEROL (UNII: R0ZB2556P8) ISODODECANE (UNII: A8289P68Y2) PHYMATOLITHON CALCAREUM (UNII: 6J1M3WA0ZK) SHEA BUTTER (UNII: K49155WL9Y) INDIAN FRANKINCENSE (UNII: 4PW41QCO2M) DIATOMACEOUS EARTH (UNII: 2RF6EJ0M85) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) MANNITOL (UNII: 3OWL53L36A) POLYMETHYLSILSESQUIOXANE (11 MICRONS) (UNII: Z570VEV8XK) ISOHEXADECANE (UNII: 918X1OUF1E) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) LECITHIN, SUNFLOWER (UNII: 834K0WOS5G) DIMETHICONE CROSSPOLYMER (450000 MPA.S AT 12% IN CYCLOPENTASILOXANE) (UNII: UF7620L1W6) DIMETHICONE/BIS-ISOBUTYL PPG-20 CROSSPOLYMER (UNII: O4I3UFO6ZF) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82384-0031-1 15 mL in 1 TUBE; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 12/17/2021 Part 2 of 3 GLOWSCREEN SPF40
avobenzone, octisalate, octocrylene creamProduct Information Item Code (Source) NDC:82384-0032 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 10 g in 100 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 100 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5 g in 100 mL Inactive Ingredients Ingredient Name Strength NIACINAMIDE (UNII: 25X51I8RD4) ETHYLHEXYL HYDROXYSTEARATE (UNII: B7I80BVV5E) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) CAPRYLYL GLYCOL (UNII: 00YIU5438U) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) LIMONIUM GERBERI WHOLE (UNII: 2J5K7YCF9F) LEUCONOSTOC/RADISH ROOT FERMENT FILTRATE (UNII: D2QHA03458) PANTOTHENIC ACID (UNII: 19F5HK2737) TOCOPHEROL (UNII: R0ZB2556P8) SUNFLOWER OIL (UNII: 3W1JG795YI) PROPANEDIOL (UNII: 5965N8W85T) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) POLYMETHYLSILSESQUIOXANE (4.5 MICRONS) (UNII: 59Z907ZB69) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL STEARATE CITRATE (UNII: WH8T92A065) CARBOMER INTERPOLYMER TYPE A (ALLYL SUCROSE CROSSLINKED) (UNII: 59TL3WG5CO) ISODODECANE (UNII: A8289P68Y2) ARGININE (UNII: 94ZLA3W45F) CETYL PHOSPHATE (UNII: VT07D6X67O) COCO-CAPRYLATE (UNII: 4828G836N6) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) WATER (UNII: 059QF0KO0R) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) ISODECYL NEOPENTANOATE (UNII: W60VYE24XC) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) COCOA (UNII: D9108TZ9KG) FERULIC ACID (UNII: AVM951ZWST) DIISOPROPYL SEBACATE (UNII: J8T3X564IH) TRISODIUM ETHYLENEDIAMINE DISUCCINATE (UNII: YA22H34H9Q) BISMUTH OXYCHLORIDE (UNII: 4ZR792I587) MICA (UNII: V8A1AW0880) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) LAURYL LACTATE (UNII: G5SU0BFK7O) CHLORPHENESIN (UNII: I670DAL4SZ) HYALURONATE SODIUM (UNII: YSE9PPT4TH) FERRIC OXIDE RED (UNII: 1K09F3G675) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82384-0032-1 15 mL in 1 BOTTLE; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 12/17/2021 Part 3 of 3 PLAY EVERYDAY SPF 50 WITH SUNFLOWER EXTRACT
avobenzone, homosalate, octisalate, octocrylene lotionProduct Information Item Code (Source) NDC:82384-0033 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 10 g in 100 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5 g in 100 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 7.5 g in 100 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 100 mL Inactive Ingredients Ingredient Name Strength ISODODECANE (UNII: A8289P68Y2) ISODECYL NEOPENTANOATE (UNII: W60VYE24XC) WATER (UNII: 059QF0KO0R) DIISOPROPYL SEBACATE (UNII: J8T3X564IH) GLYCERIN (UNII: PDC6A3C0OX) LAURYL LACTATE (UNII: G5SU0BFK7O) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82384-0033-1 30 mL in 1 BOTTLE; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 12/17/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 12/17/2021 Labeler - System Beauty, LLC (809198398)