Label: NEILMED CLEAR CANAL EARWAX REMOVAL KIT- carbamide peroxide solution/ drops
- NDC Code(s): 13709-314-01
- Packager: NEILMED PHARMACEUTICALS INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 11, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Purpose
- Uses
-
! Warnings
Avoid using the product and ask a doctor before use if you have :
- Ear pain, drainage or discharge
- Impacted earwax
- Ear infection, irritation or rash in the ear.
- Meniere's Syndrome, dizziness, or vertigo
- An injury or perforation (hole) of the eardrum
- Tubes in the ears
- Has recent ear/mastoid surgery
- Had an allergic reaction to carbamide peroxide or other ingredients in this product in the past.
- Heart disease, high blood pressure, thyroid disease or diabetes
To prevent contamination, do not share this product with others.
- When using this product:
- Stop use and ask a doctor if:
- Keep this and all drugs out of the reach of children
-
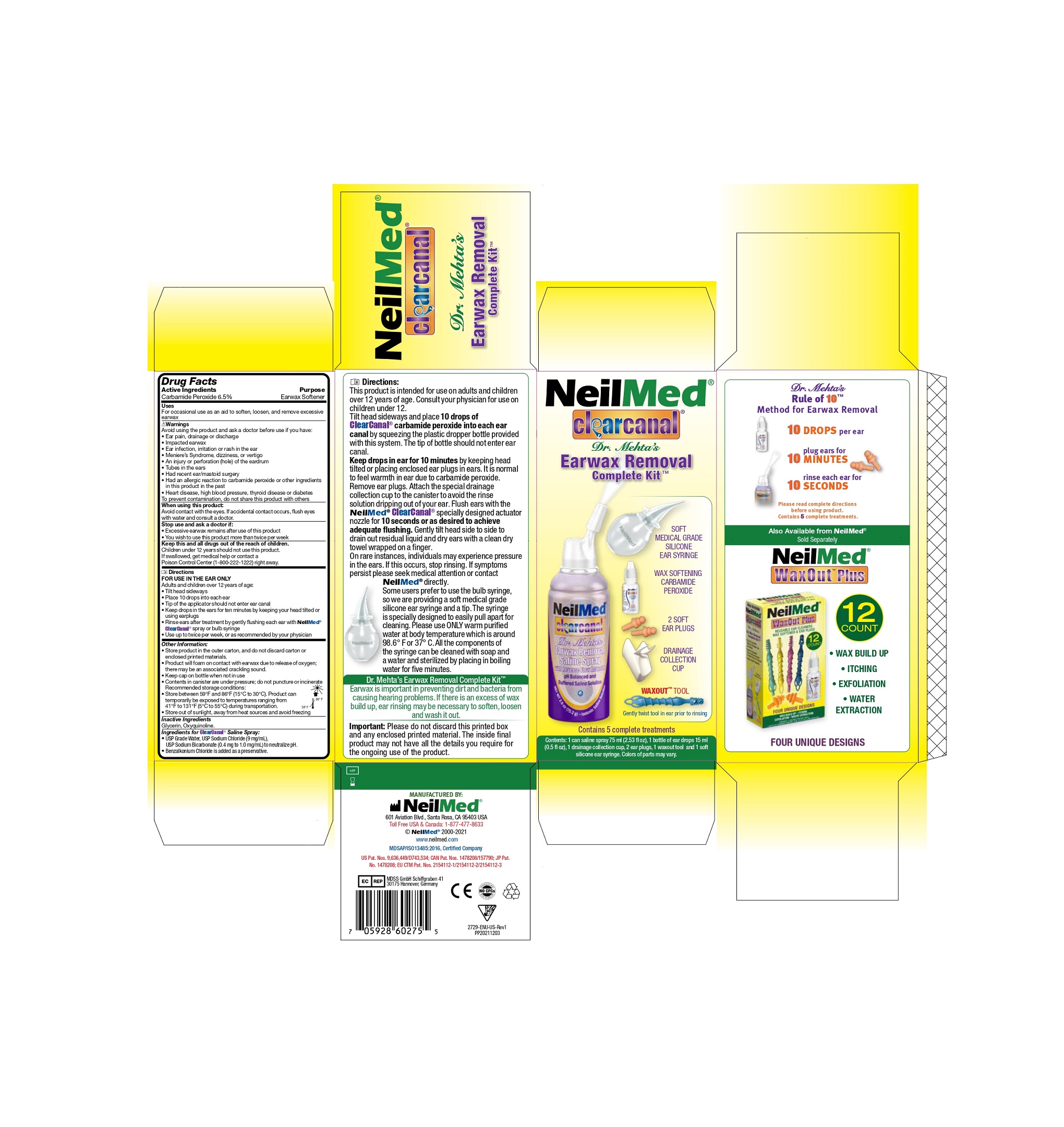
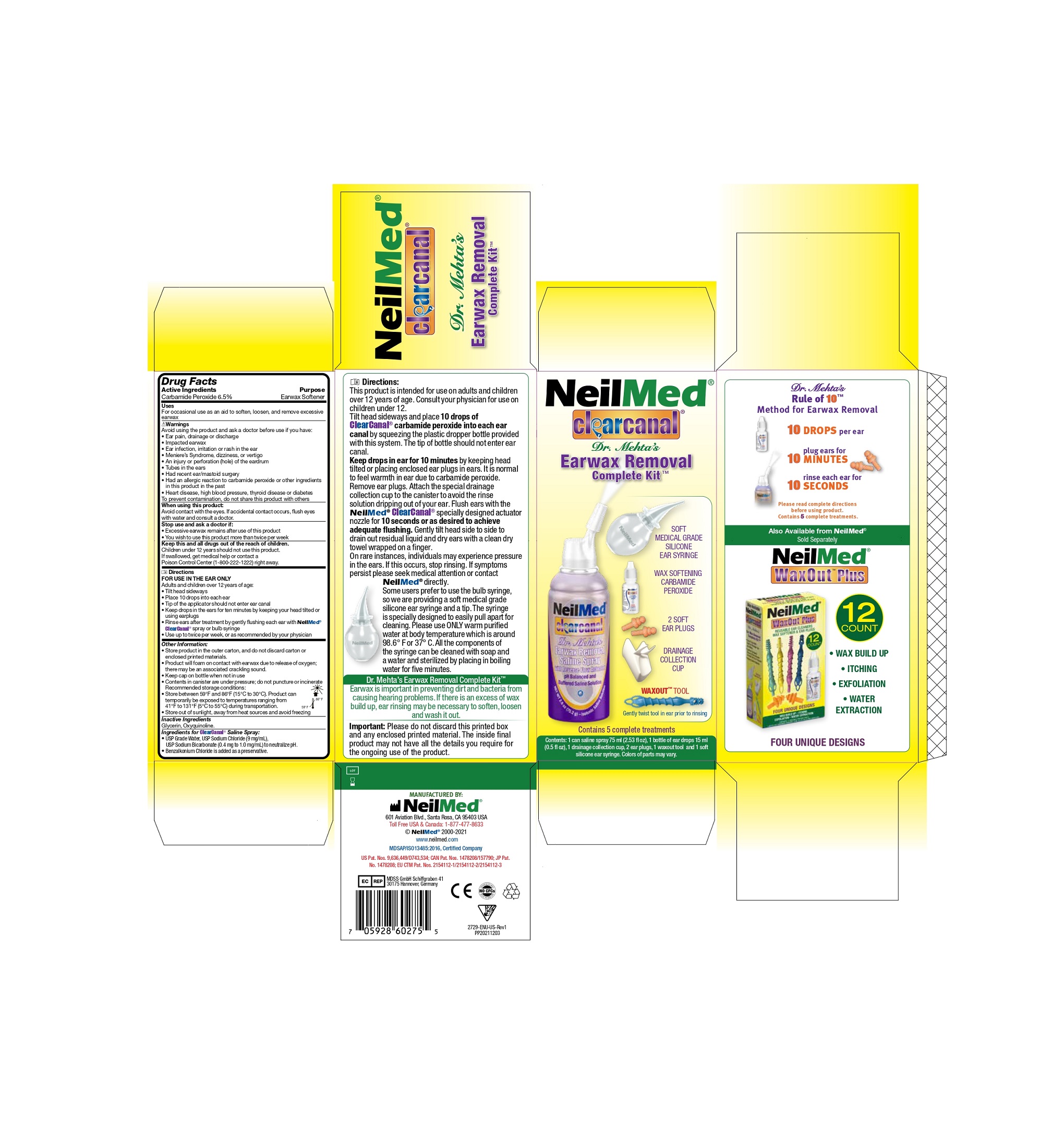
Directions
FOR USE IN THE EAR ONLY
Adults and children over 12 years of age:
- Tilt head sideways
- Place 10 drops into each ear
- Tip of the applicator should not enter ear canal
- Keep drops in the ears for ten minutes by keeping your head tilted or using earplugs
- Rinse ears after treatment by gently flusing each ear with NeilMed®ClearCanal® spray or bulb syringe
- Use up to twice per week, or as recommended by your physician.
-
Other Information:
- Store product in the outer carton, and do not discard carton or enclosed printed materials.
- Product will foam on contact with earwax due to release of oxygen; there may be an associated crackling sound.
- Keep cap on bottle when not in use.
- Contents in canister are under pressure; do not puncture or incinerate
Recommended storage conditions:
- Store between 59°F and 86°F (15°C to 30°C). Product can temporarily be exposed to temperatures ranging from 41ºF to 131ºF (5ºC to 55ºC) during transportation.
- Store out of sunlight, away from heat sources and avoid freezing
- Inactive Ingredients
- Ingredients for ClearCanal® Saline Spray :
- Contents :
-
Directions:
This product is intended for use on adults and children over 12 years of age. Consult your physician for use on children under 12.
Tilt head sideways and place 10 drops of ClearCanal Carbamide peroxide into each ear canal by squeezing the plastic dropper bottle provided with this system. The tip of bottle should not enter ear canal.
Keep drops in ear for 10 minutes by head tilted or placing enclosed ear plugs in ears. It is normal to feel warmth in ear due to carbamide peroxide. Remove ear plugs. Attach the special drainage collection cup to the canister to avoid the rinse solution dripping out of your ear. Flush ears with the NeilMed ClearCanal specially designed actuator nozzle for 10 seconds or as desired to achieve adequate flushing. Gently tilt head side to side to drain out residual liquid and dry ears with a clean dry towel wrapped on a finger.
- ClearCanal Earwax Removal Complete Kit
- ClearCanal Earwax Removal Saline Spray
-
INGREDIENTS AND APPEARANCE
NEILMED CLEAR CANAL EARWAX REMOVAL KIT
carbamide peroxide solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13709-314 Route of Administration AURICULAR (OTIC) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARBAMIDE PEROXIDE (UNII: 31PZ2VAU81) (HYDROGEN PEROXIDE - UNII:BBX060AN9V) CARBAMIDE PEROXIDE 65 mg in 1 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) OXYQUINOLINE (UNII: 5UTX5635HP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13709-314-01 1 in 1 KIT 12/25/2021 1 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M014 12/25/2021 Labeler - NEILMED PHARMACEUTICALS INC (799295915) Establishment Name Address ID/FEI Business Operations NEILMED PHARMACEUTICALS INC 799295915 manufacture(13709-314)