Label: LACTATED RINGERS AND DEXTROSE- sodium chloride, sodium lactate, potassium chloride, calcium chloride, and dextrose monohydrate injection, solution
LACTATED RINGERS- sodium chloride, sodium lactate, potassium chloride, and calcium chloride injection, solution
-
NDC Code(s):
0990-7929-03,
0990-7929-09,
0990-7953-02,
0990-7953-03, view more0990-7953-09
- Packager: ICU Medical Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated September 16, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
These products are sterile, nonpyrogenic solutions each containing isotonic concentrations of electrolytes (with or without dextrose) in water for injection. The solutions containing dextrose and electrolytes are hypertonic; those containing only electrolytes are isotonic. They are administered by intravenous infusion for parenteral replacement of extracellular losses of fluid and electrolytes, with or without minimal carbohydrate calories.
Each 100 mL of Lactated Ringer's Injection, USP contains sodium chloride 600 mg, sodium lactate, anhydrous 310 mg, potassium chloride 30 mg and calcium chloride, dihydrate 20 mg. May contain hydrochloric acid and/or sodium hydroxide for pH adjustment. A liter provides 9 calories (from lactate), sodium (Na+), 130 mEq, potassium (K+) 4 mEq, calcium (Ca++) 3 mEq, chloride (Cl−) 109 mEq and lactate [CH3CH(OH) COO−] 28 mEq. The electrolyte content is isotonic (273 mOsmol/liter, calc.) in relation to the extracellular fluid (approx. 280 mOsmol/liter). The pH of the solution is 6.6 (6.0 − 7.5).
Each 100 mL of Lactated Ringer's and 5% Dextrose Injection, USP contains dextrose, hydrous 5 g plus the same ingredients and mEq values as Lactated Ringer's Injection, USP (contains only hydrochloric acid for pH adjustment). A liter provides 179 calories (from dextrose and lactate) and has a hypertonic osmolar concentration of 525 mOsmol (calc.). The pH is 4.9 (4.0 − 6.5).
The solutions contain no bacteriostat, antimicrobial agent or added buffer (except for pH adjustment) and each is intended only for use as a single-dose injection. When smaller doses are required the unused portion should be discarded.
The solutions are parenteral fluid, nutrient and/or electrolyte replenishers.
Dextrose, USP is chemically designated D-glucose, monohydrate (C6H12O6 • H2O), a hexose sugar freely soluble in water. It has the following structural formula:
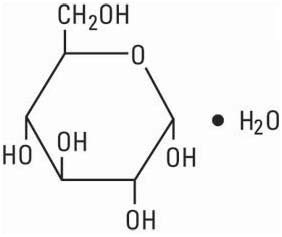
Calcium Chloride, USP is chemically designated calcium chloride, dihydrate (CaCl2 • 2 H2O), white fragments or granules freely soluble in water.
Potassium Chloride, USP is chemically designated KCl, a white granular powder freely soluble in water.
Sodium Chloride, USP is chemically designated NaCl, a white crystalline powder freely soluble in water.
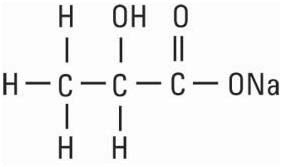
Sodium Lactate, USP is chemically designated monosodium lactate [CH3CH(OH)COONa], a 60% aqueous solution miscible in water.
It has the following structural formula:
Water for Injection, USP is chemically designated H2O.
The flexible plastic container is fabricated from a specially formulated polyvinylchloride. Water can permeate from inside the container into the overwrap but not in amounts sufficient to affect the solution significantly. Solutions inside the plastic container also can leach out certain of their chemical components in very small amounts before the expiration period is attained. However, the safety of the plastic has been confirmed by tests in animals according to USP biological standards for plastic containers.
-
CLINICAL PHARMACOLOGY
When administered intravenously, these solutions provide sources of water and electrolytes with or without minimal carbohydrate calories. Their electrolyte content resembles that of the principal ionic constituents of normal plasma and the solutions therefore are suitable for parenteral replacement of extracellular losses of fluid and electrolytes, with or without carbohydrate calories.
Solutions containing carbohydrate in the form of dextrose restore blood glucose levels and provide calories.
Carbohydrate in the form of dextrose may aid in minimizing liver glycogen depletion and exerts a protein-sparing action. Dextrose injected parenterally undergoes oxidation to carbon dioxide and water.
Calcium chloride in water dissociates to provide calcium (Ca++) and chloride (Cl−) ions. They are normal constituents of the body fluids and are dependent on various physiologic mechanisms for maintenance of balance between intake and output. Approximately 80% of body calcium is excreted in the feces as insoluble salts; urinary excretion accounts for the remaining 20%.
Potassium chloride in water dissociates to provide potassium (K+) and chloride (Cl−) ions. Potassium is found in low concentration in plasma and extracellular fluids (3.5 to 5.0 mEq/liter in a healthy adult). It is the chief cation of body cells (160 mEq/liter of intracellular water). Potassium plays an important role in electrolyte balance. Normally about 80 to 90% of the potassium intake is excreted in the urine; the remainder in the stools and to a small extent, in the perspiration. The kidney does not conserve potassium well so that during fasting or in patients on a potassium-free diet, potassium loss from the body continues resulting in potassium depletion.
Sodium chloride in water dissociates to provide sodium (Na+) and chloride (Cl−) ions. Sodium (Na+) is the principal cation of the extracellular fluid and plays a large part in the therapy of fluid and electrolyte disturbances. Chloride (Cl−) has an integral role in buffering action when oxygen and carbon dioxide exchange occurs in the red blood cells. The distribution and excretion of sodium (Na+) and chloride (Cl−) are largely under the control of the kidney which maintains a balance between intake and output.
Sodium lactate provides sodium (Na+) and lactate (C3H503−) ions. The lactate anion is in equilibrium with pyruvate and has an alkalizing effect resulting from simultaneous removal by the liver of lactate and hydrogen ions. In the liver, lactate is metabolized to glycogen which is ultimately converted to carbon dioxide and water by oxidative metabolism. The sodium (Na+) ion combines with bicarbonate ion produced from carbon dioxide of the body and thus retains bicarbonate to combat metabolic acidosis (bicarbonate deficiency). The normal plasma level of lactate ranges from 0.9 to 1.9 mEq/liter.
Water is an essential constituent of all body tissues and accounts for approximately 70% of total body weight. Average normal adult daily requirement ranges from two to three liters (1.0 to 1.5 liters each for insensible water loss by perspiration and urine production).
Water balance is maintained by various regulatory mechanisms. Water distribution depends primarily on the concentration of electrolytes in the body compartments and sodium (Na+) plays a major role in maintaining physiologic equilibrium.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
Solutions containing calcium ions should not be administered simultaneously through the same administration set as blood because of the likelihood of coagulation.
Solutions which contain potassium should be used with great care, if at all, in patients with hyperkalemia, severe renal failure and in conditions in which potassium retention is present.
Solutions containing sodium ions should be used with great care, if at all, in patients with congestive heart failure, severe renal insufficiency and in clinical states in which there exists edema with sodium retention.
In patients with diminished renal function, administration of solutions containing sodium or potassium ions may result in sodium or potassium retention.
Solutions containing lactate ions should be used with great care in patients with metabolic or respiratory alkalosis. The administration of lactate ions should be done with great care where there is an increased level or an impaired utilization of lactate ions, as in severe hepatic insufficiency.
The intravenous administration of these solutions can cause fluid and/or solute overloading resulting in dilution of serum electrolyte concentrations, overhydration, congested states or pulmonary edema. The risk of dilutional states is inversely proportional to the electrolyte concentrations of administered parenteral solutions.
The risk of solute overload causing congested states with peripheral and pulmonary edema is directly proportional to the electrolyte concentrations of such solutions.
-
PRECAUTIONS
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations and acid-base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such evaluation.
Solutions containing dextrose should be used with caution in patients with known subclinical or overt diabetes mellitus.
Caution must be exercised in the administration of parenteral fluids, especially those containing sodium ions, to patients receiving corticosteroids or corticotropin.
Potassium containing solutions should be used with caution in the presence of cardiac disease, particularly in digitalized patients or in the presence of renal disease.
Solutions containing lactate ions should be used with caution as excess administration may result in metabolic alkalosis.
Do not administer unless solution is clear and container is undamaged. Discard unused portion.
Pregnancy Category C. Animal reproduction studies have not been conducted with Ringer's Injection, USP, Ringer's and Dextrose Injection, USP, Lactated Ringer's Injection, USP or Lactated Ringer's and Dextrose Injection, USP. It is also not known whether these injections can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. These injections should be given to a pregnant woman only if clearly needed.
Pediatric Use: The safety and effectiveness in the pediatric population are based on the similarity of the clinical conditions of the pediatric and adult populations. In neonates or very small infants the volume of fluid may affect fluid and electrolyte balance.
Frequent monitoring of serum glucose concentrations is required when dextrose is prescribed to pediatric patients, particularly neonates and low birth weight infants.
In very low birth weight infants, excessive or rapid administration of dextrose injection may result in increased serum osmolarity and possible intracerebral hemorrhage.
-
ADVERSE REACTIONS
Reactions which may occur because of the solution or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
- OVERDOSAGE
-
DOSAGE AND ADMINISTRATION
The dose is dependent upon the age, weight and clinical condition of the patient.
As reported in the literature, the dosage and constant infusion rate of intravenous dextrose must be selected with caution in pediatric patients, particularly neonates and low birth weight infants, because of the increased risk of hyperglycemia/hypoglycemia.
Drug Interactions
Additives may be incompatible. Consult with pharmacist, if available. When introducing additives, use aseptic technique, mix thoroughly and do not store.
The presence of calcium limits their compatibility with certain drugs that form precipitates of calcium salts, and also prohibits their simultaneous infusion through the same administration set as blood because of the likelihood of coagulation.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. See PRECAUTIONS.
-
INSTRUCTIONS FOR USE
To Open:
Tear outer wrap at notch and remove solution container. If supplemental medication is desired, follow directions below before preparing for administration. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually.
To Add Medication
- Prepare additive port.
- Using aseptic technique and an additive delivery needle of appropriate length, puncture resealable additive port at target area, inner diaphragm and inject. Withdraw needle after injecting medication.
- The additive port may be protected by covering with an additive cap.
- Mix container contents thoroughly.
Preparation for Administration
(Use aseptic technique)
- Close flow control clamp of administration set.
- Remove cover from outlet port at bottom of container.
- Insert piercing pin of administration set into port with a twisting motion until the set is firmly seated. NOTE: See full directions on administration set carton.
- Suspend container from hanger.
- Squeeze and release drip chamber to establish proper fluid level in chamber.
- Open flow control clamp and clear air from set. Close clamp.
- Attach set to venipuncture device. If device is not indwelling, prime and make venipuncture.
- Regulate rate of administration with flow control clamp.
WARNING: DO NOT USE FLEXIBLE CONTAINER IN SERIES CONNECTIONS.
-
HOW SUPPLIED
These solutions are supplied in single-dose flexible plastic containers as follows:
NDC No. Product Name Container Size (mL) 0409-7953-02* Lactated Ringer's Inj., USP 250 0990-7953-02* Lactated Ringer's Inj., USP 250 0409-7953-03† Lactated Ringer's Inj., USP 500 0990-7953-03*,† Lactated Ringer's Inj., USP 500 0409-7953-09*,† Lactated Ringer's Inj., USP 1000 0990-7953-09*,† Lactated Ringer's Inj., USP 1000 0409-7929-03† Lactated Ringer's and 5% Dextrose Inj., USP 500 0990-7929-03*,† Lactated Ringer's and 5% Dextrose Inj., USP 500 0409-7929-09*,† Lactated Ringer's and 5% Dextrose Inj., USP 1000 0990-7929-09*,† Lactated Ringer's and 5% Dextrose Inj., USP 1000 ICU Medical is transitioning NDC codes from "0409" to a"0990" labeler code. Both NDC codes are expected to be in the market for a period of time.
Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.] Protect from freezing.
Revised: April, 2020
IFU0000184
-
PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - NDC 0990-7929-09
1000 mL
NDC 0990-7929-09LACTATED RINGER'S and
5% DEXTROSE
Injection, USPEACH 100 mL CONTAINS DEXTROSE, HYDROUS
5 g; SODIUM LACTATE, ANHYD. 310 mg; SODIUM
CHLORIDE 600 mg; POTASSIUM CHLORIDE
30 mg; CALCIUM CHLORIDE, DIHYDRATE 20 mg
IN WATER FOR INJECTION. pH ADJUSTED WITH
HCl.
ELECTROLYTES PER 1000 mL (NOT INCLUDING
pH ADJUSTMENT): SODIUM 130 mEq;
POTASSIUM 4 mEq; CALCIUM 3 mEq; CHLORIDE
109 mEq; LACTATE 28 mEq.
525 mOsmol/LITER (CALC). pH 4.9 (4.0 to 6.5).
CAUTION: DO NOT ADMINISTER CALCIUM
CONTAINING SOLUTIONS CONCURRENTLY
WITH STORED BLOOD. NOT FOR USE IN THE
TREATMENT OF LACTIC ACIDOSIS.
ADDITIVES MAY BE INCOMPATIBLE. CONSULT
WITH PHARMACIST, IF AVAILABLE. WHEN
INTRODUCING ADDITIVES, USE ASEPTIC
TECHNIQUE, MIX THOROUGHLY AND DO NOT
STORE. SINGLE-DOSE CONTAINER. FOR I.V.
USE. USUAL DOSAGE: SEE INSERT. STERILE,
NONPYROGENIC. USE ONLY IF SOLUTION IS
CLEAR AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES CONNECTIONS.3
V
CONTAINS DEHPRX ONLY
icumedical
IM-4426
ICU Medical, Inc., Lake Forest, Illinois, 60045, USA

-
PRINCIPAL DISPLAY PANEL - 1000 mL Bag Overwrap - NDC 0409-7929-09
TO OPEN TEAR AT NOTCH
2
HDPEDO NOT REMOVE FROM OVERWRAP UNTIL READY FOR USE. AFTER REMOVING
THE OVERWRAP, CHECK FOR MINUTE LEAKS BY SQUEEZING CONTAINER FIRMLY.
IF LEAKS ARE FOUND, DISCARD SOLUTION AS STERILITY MAY BE IMPAIRED.
RECOMMENDED STORAGE: ROOM TEMPERATURE (25°C). AVOID EXCESSIVE
HEAT. PROTECT FROM FREEZING. SEE INSERT.
98-4321-R14-3/98
-
PRINCIPAL DISPLAY PANEL - 1000 mL Bag Label - NDC 0990-7953-09
1000 mL
NDC 0990-7953-09LACTATED RINGER'S
Injection, USPEACH 100 mL CONTAINS SODIUM CHLORIDE
600 mg; SODIUM LACTATE, ANHYD. 310 mg;
POTASSIUM CHLORIDE 30 mg; CALCIUM
CHLORIDE, DIHYDRATE 20 mg IN WATER FOR
INJECTION. MAY CONTAIN HCl OR NaOH FOR
pH ADJUSTMENT.
ELECTROLYTES PER 1000 mL (NOT INCLUDING
pH ADJUSTMENT): SODIUM 130 mEq;
POTASSIUM 4 mEq; CALCIUM 3 mEq;
CHLORIDE 109 mEq; LACTATE 28 mEq.
273 mOsmol/LITER (CALC). pH 6.6 (6.0 to 7.5).
CAUTION: DO NOT ADMINISTER CALCIUM
CONTAINING SOLUTIONS CONCURRENTLY
WITH STORED BLOOD. NOT FOR USE IN
THE TREATMENT OF LACTIC ACIDOSIS.
ADDITIVES MAY BE INCOMPATIBLE. CONSULT
WITH PHARMACIST, IF AVAILABLE. WHEN
INTRODUCING ADDITIVES, USE ASEPTIC
TECHNIQUE, MIX THOROUGHLY AND DO NOT
STORE. SINGLE-DOSE CONTAINER. FOR I.V. OR
SUBCUTANEOUS USE. USUAL DOSAGE: SEE
INSERT. STERILE, NONPYROGENIC. USE ONLY
IF SOLUTION IS CLEAR AND CONTAINER IS
UNDAMAGED. MUST NOT BE USED IN SERIES
CONNECTIONS.RX ONLY
3
V
CONTAINS DEHPicumedical
IM-4389
ICU Medical, Inc., Lake Forest, Illinois, 60045 USA

-
PRINCIPAL DISPLAY PANEL - 1000 mL Bag Overwrap - NDC 0409-7953-09
TO OPEN TEAR AT NOTCH
2
HDPEDO NOT REMOVE FROM OVERWRAP UNTIL READY FOR USE. AFTER REMOVING
THE OVERWRAP, CHECK FOR MINUTE LEAKS BY SQUEEZING CONTAINER FIRMLY.
IF LEAKS ARE FOUND, DISCARD SOLUTION AS STERILITY MAY BE IMPAIRED.
RECOMMENDED STORAGE: ROOM TEMPERATURE (25°C). AVOID EXCESSIVE
HEAT. PROTECT FROM FREEZING. SEE INSERT.
98-4321-R14-3/98
-
INGREDIENTS AND APPEARANCE
LACTATED RINGERS AND DEXTROSE
sodium chloride, sodium lactate, potassium chloride, calcium chloride, and dextrose monohydrate injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0990-7929 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 600 mg in 100 mL SODIUM LACTATE (UNII: TU7HW0W0QT) (SODIUM CATION - UNII:LYR4M0NH37, LACTIC ACID, UNSPECIFIED FORM - UNII:33X04XA5AT) SODIUM LACTATE 310 mg in 100 mL POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152, CHLORIDE ION - UNII:Q32ZN48698) POTASSIUM CHLORIDE 30 mg in 100 mL CALCIUM CHLORIDE (UNII: M4I0D6VV5M) (CALCIUM CATION - UNII:2M83C4R6ZB, CHLORIDE ION - UNII:Q32ZN48698) CALCIUM CHLORIDE 20 mg in 100 mL DEXTROSE MONOHYDRATE (UNII: LX22YL083G) (ANHYDROUS DEXTROSE - UNII:5SL0G7R0OK) DEXTROSE MONOHYDRATE 5 g in 100 mL Inactive Ingredients Ingredient Name Strength HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0990-7929-03 24 in 1 CASE 08/01/2019 1 1 in 1 POUCH 1 500 mL in 1 BAG; Type 0: Not a Combination Product 2 NDC:0990-7929-09 12 in 1 CASE 08/01/2019 2 1 in 1 POUCH 2 1000 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA017608 06/24/2019 LACTATED RINGERS
sodium chloride, sodium lactate, potassium chloride, and calcium chloride injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0990-7953 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37, CHLORIDE ION - UNII:Q32ZN48698) SODIUM CHLORIDE 600 mg in 100 mL SODIUM LACTATE (UNII: TU7HW0W0QT) (SODIUM CATION - UNII:LYR4M0NH37, LACTIC ACID, UNSPECIFIED FORM - UNII:33X04XA5AT) SODIUM LACTATE 310 mg in 100 mL POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152, CHLORIDE ION - UNII:Q32ZN48698) POTASSIUM CHLORIDE 30 mg in 100 mL CALCIUM CHLORIDE (UNII: M4I0D6VV5M) (CALCIUM CATION - UNII:2M83C4R6ZB, CHLORIDE ION - UNII:Q32ZN48698) CALCIUM CHLORIDE 20 mg in 100 mL Inactive Ingredients Ingredient Name Strength HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0990-7953-02 24 in 1 CASE 06/24/2019 1 1 in 1 POUCH 1 250 mL in 1 BAG; Type 0: Not a Combination Product 2 NDC:0990-7953-03 24 in 1 CASE 08/01/2019 2 1 in 1 POUCH 2 500 mL in 1 BAG; Type 0: Not a Combination Product 3 NDC:0990-7953-09 12 in 1 CASE 08/01/2019 3 1 in 1 POUCH 3 1000 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA017641 06/24/2019 Labeler - ICU Medical Inc. (118380146) Establishment Name Address ID/FEI Business Operations ICU MEDICAL INC. 117395980 ANALYSIS(0990-7929, 0990-7953) , MANUFACTURE(0990-7929, 0990-7953) , PACK(0990-7929, 0990-7953) , LABEL(0990-7929, 0990-7953)




