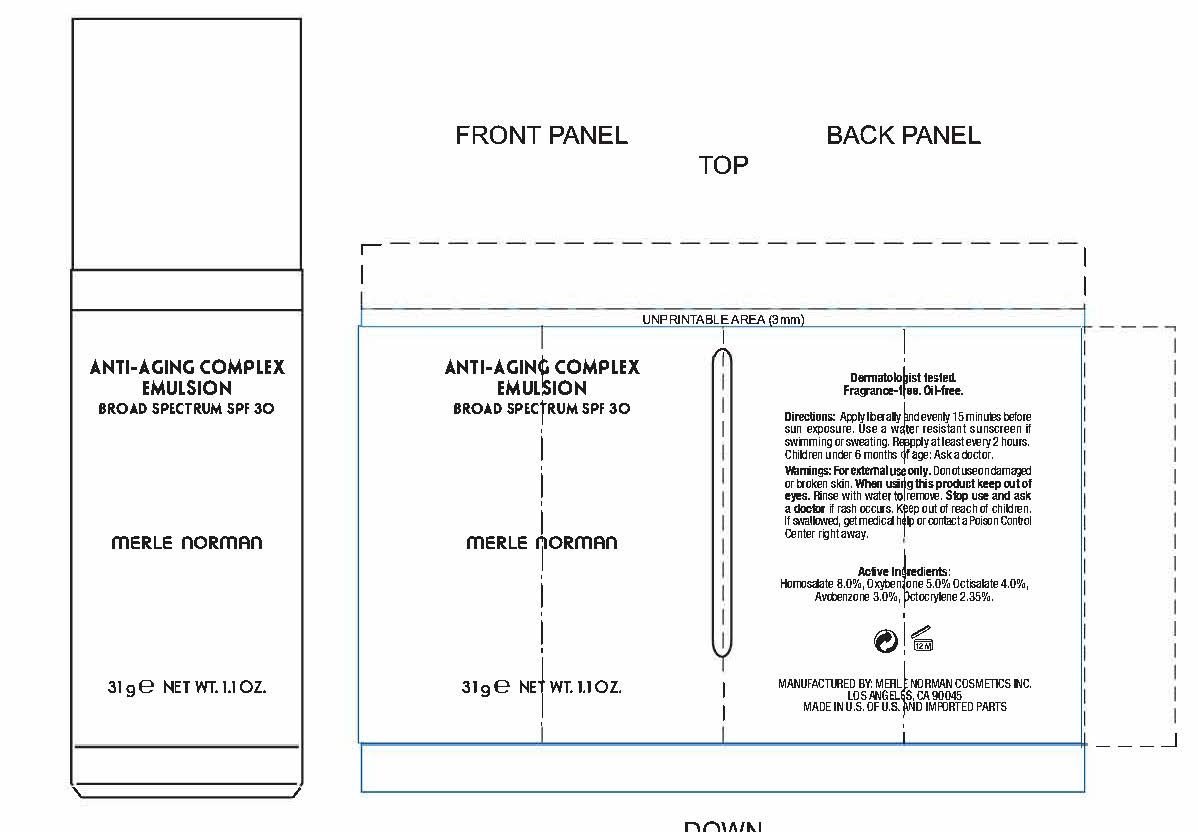
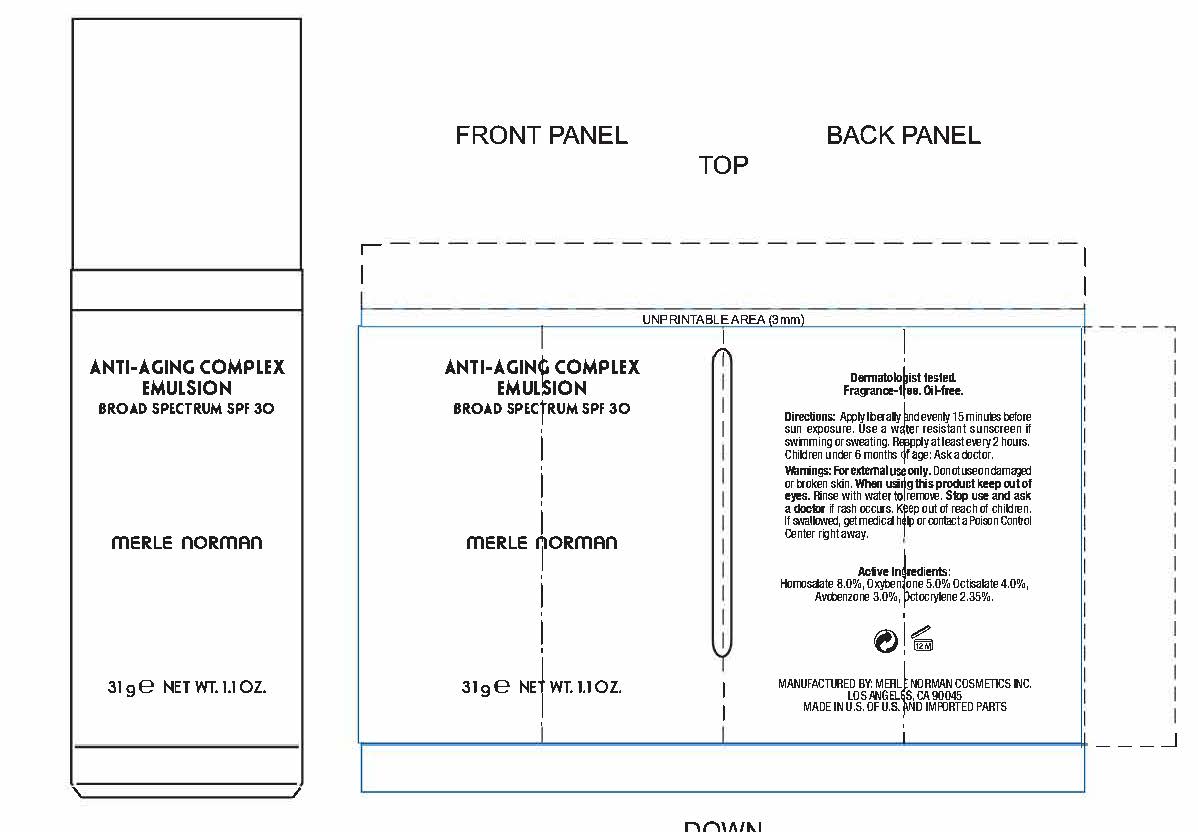
Label: ANTI-AGING COMPLEX BROAD SPECTRUM SPF 30- avobenzone, homosalate, octisalate, octocrylene, oxybenzone cream
- NDC Code(s): 57627-204-01
- Packager: Merle Norman Cosmetics, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- WARNINGS
- INDICATIONS & USAGE
-
DOSAGE & ADMINISTRATION
Directions
- For sunscreen use:
- apply liberally and evenly 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- children under 6 months: Ask a doctor
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
- children under 6 months: Ask a doctor
- Apply to cleansed and toned skin prior to sun exposure.
-
INACTIVE INGREDIENT
Inactive ingredients: Aqua/Water/Eau, Glycerin, Ethoxydiglycol, Butylene Glycol, Alcohol Denat., Cyclopentasiloxane, Glycereth-26, Isostearyl Neopentanoate, Nylon-12, Glyceryl Stearate, Bis-Ethylhexyl Hydroxydimethoxy Benzylmalonate, Isononyl Isononanoate, Phenoxyethanol, Ammonium Acryloyldimethyltaurate/VP Copolymer, Polymethyl Methacrylate, Hydrolyzed Malt Extract, Sodium Acrylate/Sodium Acryloyldimethyl Taurate Copolymer, Polyisobutene, Hydrogenated Lecithin, PEG-100 Stearate, Caprylyl Glycol, Mica (CI 77019), Coco-Glucoside, Ethylhexylglycerin, Tetrasodium EDTA, Xanthan Gum, Coconut Alcohol, BHT, Bisabolol, Hexylene Glycol, Propylene Glycol, Caprylyl/Capryl Glucoside, Opuntia Ficus-Indica Stem Extract, Hydroxypropyl Cyclodextrin, Acetyl Tyrosine, Phospholipids, Panax Ginseng Root Extract, Camellia Sinensis Leaf Extract, Proline, Silica, Adenosine Triphosphate, Hydrolyzed Vegetable Protein, Acetyl Octapeptide-3, Equisetum Arvense Extract, Acetylarginyltryptophyl Diphenylglycine, Ascorbyl Palmitate, Retinyl Palmitate, Palmitoyl Tripeptide-38, Tocopheryl Acetate.
- OTHER SAFETY INFORMATION
- STORAGE AND HANDLING
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ANTI-AGING COMPLEX BROAD SPECTRUM SPF 30
avobenzone, homosalate, octisalate, octocrylene, oxybenzone creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57627-204 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 4 g in 100 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 2.35 g in 100 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 5 g in 100 g AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 100 g HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 8 g in 100 g Inactive Ingredients Ingredient Name Strength GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) BIS-ETHYLHEXYL HYDROXYDIMETHOXY BENZYLMALONATE (UNII: 7D4Q5YJ8NV) ISONONYL ISONONANOATE (UNII: S4V5BS6GCX) MICA (UNII: V8A1AW0880) EDETATE SODIUM (UNII: MP1J8420LU) XANTHAN GUM (UNII: TTV12P4NEE) GREEN TEA LEAF (UNII: W2ZU1RY8B0) ASIAN GINSENG (UNII: CUQ3A77YXI) AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) WATER (UNII: 059QF0KO0R) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) GLYCERETH-26 (UNII: NNE56F2N14) PALMITOYL LYSYLDIOXYMETHIONYLLYSINE (UNII: T7A529FB8O) ISOSTEARYL NEOPENTANOATE (UNII: 411THY156Q) NYLON-12 (UNII: 446U8J075B) VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) EQUISETUM ARVENSE BRANCH (UNII: 1L0VKZ185E) PHENOXYETHANOL (UNII: HIE492ZZ3T) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) ACETYL OCTAPEPTIDE-3 (UNII: 8K14HJF88S) PROLINE (UNII: 9DLQ4CIU6V) ASCORBYL PALMITATE (UNII: QN83US2B0N) DIETHYLENE GLYCOL MONOETHYL ETHER (UNII: A1A1I8X02B) ALCOHOL (UNII: 3K9958V90M) PEG-100 STEARATE (UNII: YD01N1999R) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) COCONUT ALCOHOL (UNII: 13F4MW8Y9K) CAPRYLYL/CAPRYL OLIGOGLUCOSIDE (UNII: E00JL9G9K0) OPUNTIA FICUS-INDICA STEM (UNII: MUD8892KHL) ACETYLARGINYLTRYPTOPHYL DIPHENYLGLYCINE (UNII: 9O32SH1GPH) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) HYDROGENATED SOYBEAN LECITHIN (UNII: H1109Z9J4N) CAPRYLYL GLYCOL (UNII: 00YIU5438U) ACETYL L-TYROSINE (UNII: DA8G610ZO5) ADENOSINE (UNII: K72T3FS567) GLYCERIN (UNII: PDC6A3C0OX) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) LEVOMENOL (UNII: 24WE03BX2T) HEXYLENE GLYCOL (UNII: KEH0A3F75J) SODIUM ACRYLATE/SODIUM ACRYLOYLDIMETHYLTAURATE COPOLYMER (4000000 MW) (UNII: 1DXE3F3OZX) TRIPOLYPHOSPHATE ION (UNII: 5798IYA5AY) COCO GLUCOSIDE (UNII: ICS790225B) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57627-204-01 31 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 11/29/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 11/29/2021 Labeler - Merle Norman Cosmetics, Inc. (008479388)