Label: avinza- morphine sulfate capsule, extended release
-
Contains inactivated NDC Code(s)
NDC Code(s): 64365-505-03, 64365-506-03, 64365-507-02, 64365-508-02 - Packager: Ligand Pharmaceuticals Incorporated
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated August 8, 2006
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- N/A - Section Title Not Found In Database
-
BOXED WARNING
(What is this?)
WARNING:
AVINZA capsules are a modified-release formulation of morphine sulfate indicated for once daily administration for the relief of moderate to severe pain requiring continuous, around-the-clock opioid therapy for an extended period of time. AVINZA CAPSULES ARE TO BE SWALLOWED WHOLE OR THE CONTENTS OF THE CAPSULES SPRINKLED ON APPLESAUCE. THE CAPSULE BEADS ARE NOT TO BE CHEWED, CRUSHED, OR DISSOLVED DUE TO THE RISK OF RAPID RELEASE AND ABSORPTION OF A POTENTIALLY FATAL DOSE OF MORPHINE. PATIENTS MUST NOT CONSUME ALCOHOLIC BEVERAGES WHILE ON AVINZA THERAPY. ADDITIONALLY, PATIENTS MUST NOT USE PRESCRIPTION OR NON-PRESCRIPTION MEDICATIONS CONTAINING ALCOHOL WHILE ON AVINZA THERAPY. CONSUMPTION OF ALCOHOL WHILE TAKING AVINZA MAY RESULT IN THE RAPID RELEASE AND ABSORPTION OF A POTENTIALLY FATAL DOSE OF MORPHINE.
-
DESCRIPTION
AVINZA (morphine sulfate extended-release capsules) 30, 60, 90, and 120 mg contain both immediate release and extended release beads of morphine sulfate for once daily oral administration.
Chemically, morphine sulfate is 7,8-didehydro-4,5 alpha-epoxy-17-methyl-morphinan-3,6 alpha-diol sulfate (2:1) (salt) pentahydrate with a molecular weight of 758. Morphine sulfate occurs as white, feathery, silky crystals; cubical masses of crystal; or white crystalline powder. It is soluble in water and slightly soluble in alcohol, but is practically insoluble in chloroform or ether. The octanol:water partition coefficient of morphine is 1.42 at physiologic pH and the pKa is 7.9 for the tertiary nitrogen (the majority is ionized at pH 7.4).
Each AVINZA Capsule contains either 30, 60, 90, or 120 mg of morphine sulfate, USP and the following inactive ingredients: ammonio-methacrylate copolymers, NF, fumaric acid, NF, povidone, USP, sodium lauryl sulfate, NF, sugar starch spheres, NF, and talc, USP. The capsule shell contains black ink, gelatin, titanium dioxide, D&C yellow No. 10 (30 mg), FDC green No. 3 (60 mg), FD&C red No. 40 (90 mg), FD&C red No. 3 (120 mg), and FD&C blue No. 1 (120 mg).
Structure:
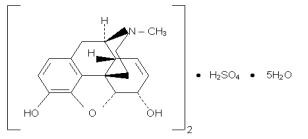
AVINZA uses the proprietary SODAS® (Spheroidal Oral Drug Absorption System) technology to produce the extended release component of AVINZA, which combined with an immediate release component achieves the desired release profile characteristics of AVINZA capsules. Within the gastrointestinal tract, due to the permeability of the ammonio‑methacrylate copolymers of the beads, fluid enters the beads and solubilizes the drug. This is mediated by fumaric acid, which acts as an osmotic agent and a local pH modifier. The resultant solution then diffuses out in a predetermined manner which prolongs the in vivo dissolution and absorption phases. (see Pharmacokinetics)
-
CLINICAL PHARMACOLOGY
Morphine, a pure opioid agonist, is relatively selective for the mu receptor, although it can interact with other opioid receptors at higher doses. In addition to analgesia, the widely diverse effects of morphine include drowsiness, changes in mood, respiratory depression, decreased gastrointestinal motility, nausea, vomiting, and alterations of the endocrine and autonomic nervous system.
Effects on the Central Nervous System (CNS): The principal therapeutic action of morphine is analgesia. Other therapeutic effects of morphine include anxiolysis, euphoria and feelings of relaxation. Although the precise mechanism of the analgesic action is unknown, specific CNS opiate receptors and endogenous compounds with morphine-like activity have been identified throughout the brain and spinal cord and are likely to play a role in the expression and perception of analgesic effects. In common with other opioids, morphine causes respiratory depression, in part by a direct effect on the brainstem respiratory centers. Morphine and related opioids depress the cough reflex by direct effect on the cough center in the medulla. Antitussive effects may occur with doses lower than those usually required for analgesia. Morphine causes miosis, even in total darkness. Pinpoint pupils are a sign of opioid overdose; however, when asphyxia is present during opioid overdose, marked mydriasis occurs.
Effects on the Gastrointestinal Tract and on Other Smooth Muscle: Gastric, biliary and pancreatic secretions are decreased by morphine. Morphine causes a reduction in motility and is associated with an increase in tone in the antrum of the stomach and duodenum. Digestion of food in the small intestine is delayed and propulsive contractions are decreased. Propulsive peristaltic waves in the colon are decreased, while tone is increased to the point of spasm. The end result may be constipation. Morphine can cause a marked increase in biliary tract pressure as a result of spasm of the sphincter of Oddi. Morphine may also cause spasm of the sphincter of the urinary bladder.
Effects on the Cardiovascular System: In therapeutic doses, morphine does not usually exert major effects on the cardiovascular system. Morphine produces peripheral vasodilation which may result in orthostatic hypotension and fainting. Release of histamine can occur, which may play a role in opioid-induced hypotension. Manifestations of histamine release and/or peripheral vasodilation may include pruritus, flushing, red eyes and sweating.
Pharmacodynamics
Morphine concentrations are not predictive of analgesic response, especially in patients previously treated with opioids. The minimum effective concentration varies widely and is influenced by a variety of factors, including the extent of previous opioid use, age, and general medical condition. Effective doses in tolerant patients may be significantly higher than in opioid-naïve patients.
In all patients, the dose of morphine should be titrated on the basis of clinical evaluation of the patient and to achieve a balance between therapeutic and adverse effects.
Pharmacokinetics
AVINZA consists of two components, an immediate release component that rapidly achieves plateau morphine plasma concentrations and an extended release component that maintains plasma concentrations throughout the 24‑hour dosing interval. The amount of morphine absorbed from AVINZA following oral administration is similar to that absorbed from other oral morphine formulations.
The oral bioavailability of morphine is less than 40% and shows large inter-individual variability due to extensive pre-systemic metabolism.
Absorption
Following single-dose oral administration of a 60 mg dose of AVINZA under fasting conditions, morphine concentrations of approximately 3 to 6 ng/ml were achieved within 30 minutes after dosing and maintained for the 24-hour dosing interval. The pharmacokinetics of AVINZA were shown to be dose-proportional over a single oral dose range of 30 to 120 mg in healthy volunteers and a multiple oral dose range of at least 30 to 180 mg in patients with chronic moderate to severe pain.
Food Effects: When a 60 mg dose of AVINZA was administered immediately following a high fat meal, peak morphine concentrations and AUC values were similar to those observed when the dose of AVINZA was administered in a fasting state, although achievement of initial concentrations was delayed by approximately 1 hour under fed conditions. Therefore, AVINZA can be administered without regard to food. When the contents of AVINZA were administered by sprinkling on applesauce, the rate and extent of morphine absorption were found to be bioequivalent to the same dose when administered as an intact capsule.
Steady‑State: When dosed once-daily, AVINZA steady‑state pharmacokinetics are characterized by a plateau-like plasma concentration profile. Steady‑state plasma concentrations of morphine are achieved 2 to 3 days after initiation of once-daily administration of AVINZA.
AVINZA 60 mg Capsules (once-daily) and 10 mg morphine oral solution (6 times daily) were equally bioavailable.
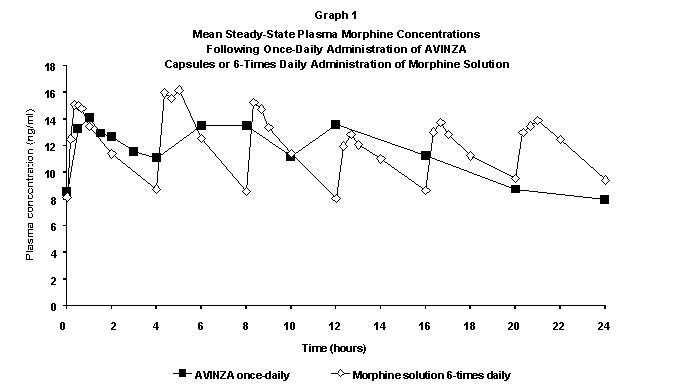
A once-daily dose of AVINZA provided similar Cmax, Cmin, and AUC values and peak-trough fluctuations (% FL, Cmax-Cmin/Cav) compared to 6-times daily administration of the same total daily dose of morphine oral solution (Table 1).
Table 1 Pharmacokinetic Data Mean ± SD Parameter AVINZA Capsules Once-Daily Morphine Oral Solution
6-Times DailyAUC (ng/ml.h) 273.26 + 81.24 279.11 + 63.00 Cmax (ng/ml) 18.65 + 7.13 19.96 + 4.82 Cmin (ng/ml) 6.98 + 2.44 6.61 + 2.15 % FL 106.38 + 78.14 116.22 + 26.67 Distribution
Once absorbed, morphine is distributed to skeletal muscle, kidneys, liver, intestinal tract, lungs, spleen and brain. Although the primary site of action is the CNS, only small quantities cross the blood-brain barrier. Morphine also crosses the placental membranes and has been found in breast milk. The volume of distribution of morphine is approximately 1 to 6 L/kg, and morphine is 20 to 35% reversibly bound to plasma proteins.
Metabolism
The major pathway of morphine detoxification is conjugation, either with D-glucuronic acid to produce glucuronides or with sulfuric acid to produce morphine-3-etheral sulfate. While a small fraction (less than 5%) of morphine is demethylated, virtually all morphine is converted by hepatic metabolism to the 3- and 6-glucuronide metabolites (M3G and M6G; about 50% and 15%, respectively). M6G has been shown to have analgesic activity but crosses the blood-brain barrier poorly, while M3G has no significant analgesic activity.
Excretion
Most of a dose of morphine is excreted in urine as M3G and M6G, with elimination of morphine occurring primarily as renal excretion of M3G. Approximately 10% of the dose is excreted unchanged in urine. A small amount of the glucuronide conjugates are excreted in bile, with minor enterohepatic recycling. Seven to 10% of administered morphine is excreted in the feces.
The mean adult plasma clearance is approximately 20 to 30 ml/min/kg. The effective terminal half-life of morphine after IV administration is reported to be approximately 2 hours. In some studies involving longer periods of plasma sampling, a longer terminal half-life of morphine of about 15 hours was reported.
In Vitro AVINZA-Alcohol Interaction
In vitro studies performed by the FDA demonstrated that when AVINZA 30 mg was mixed with 900 mL of buffer solutions containing ethanol (20% and 40%), the dose of morphine that was released was alcohol concentration-dependent, leading to a more rapid release of morphine. While the relevance of in vitro lab tests regarding AVINZA to the clinical setting remains to be determined, this acceleration of release may correlate with in vivo rapid release of the total morphine dose, which could result in the absorption of a potentially fatal dose of morphine.
Special Populations
Geriatric: Elderly patients (aged 65 years or older) may have increased sensitivity to morphine. AVINZA pharmacokinetics have not been studied specifically in elderly patients.
Nursing Mothers: Low levels of morphine sulfate have been detected in maternal milk. The milk:plasma morphine AUC ratio is about 2.5:1. The amount of morphine delivered to the infant depends on the plasma concentration of the mother, the amount of milk ingested by the infant, and the extent of first‑pass metabolism.
Pediatric: The pharmacokinetics of AVINZA have not been studied in pediatric patients below the age of 18. The range of dose strengths available may not be appropriate for treatment of very young pediatric patients. Sprinkling on applesauce is NOT a suitable alternative for these patients.
Gender: A gender analysis of pharmacokinetic data from healthy subjects taking AVINZA indicated that morphine concentrations were similar in males and females.
Race: There may be some pharmacokinetic differences associated with race. In one published study, Chinese subjects given intravenous morphine had a higher clearance when compared to Caucasian subjects (1852 +/- 116 ml/min compared to 1495 +/‑ 80 ml/min).
Hepatic Failure: Morphine pharmacokinetics have been reported to be significantly altered in patients with cirrhosis. Clearance was found to decrease with a corresponding increase in half-life. The M3G and M6G to morphine plasma AUC ratios also decreased in these subjects, indicating diminished metabolic activity.
Drug-Drug Interactions: Known drug-drug interactions involving morphine are pharmacodynamic, not pharmacokinetic. (see PRECAUTIONS, Drug Interactions)
-
Clinical Studies
AVINZA was studied in over 140 healthy volunteers and 560 patients with chronic, moderate to severe pain who participated in 6 pharmacokinetic studies, 4 clinical studies and 3 studies which provided both pharmacokinetic and clinical data. The patient population included those who were either receiving chronic opioid therapy or had a prior sub-optimal response to acetaminophen and/or NSAID therapy, as well as patients who previously received intermittent opioid analgesic therapy. In the controlled clinical studies, patients were followed from 7 days to up to 4 weeks, and in the open label studies, patients were followed for up to 6 to 12 months.
AVINZA was studied in a double-blind, placebo-controlled, fixed-dose, parallel group trial in 295 patients with moderate to severe pain due to osteoarthritis. These patients had either a prior sub-optimal response to acetaminophen, NSAID therapy, or previously received intermittent opioid analgesic therapy. Thirty-milligrams AVINZA capsules administered once-daily, either in the morning or the evening, were more effective than placebo in reducing pain.
Table 2 Change from Baseline in WOMAC OA Index Pain VAS Subscale Score *P<0.05; REPEATED MEASURES ANALYSIS
Overall Placebo AVINZA QAM AVINZA QPM LS Mean -36.23 -75.26* -75.39* Std. Error 11.482 11.305 11.747
This study was not designed to assess the effects of AVINZA on the course of the osteoarthritis.
-
INDICATIONS & USAGE
AVINZA capsules are a modified-release formulation of morphine sulfate intended for once daily administration indicated for the relief of moderate to severe pain requiring continuous, around-the-clock opioid therapy for an extended period of time.
AVINZA is NOT intended for use as a prn analgesic.
The safety and efficacy of using AVINZA in the postoperative setting has not been evaluated. AVINZA is not indicated for postoperative use. If the patient has been receiving the drug prior to surgery, resumption of the pre-surgical dose may be appropriate once the patient is able to take the drug by mouth. Physicians should individualize treatment, moving from parenteral to oral analgesics as appropriate. (see American Pain Society guidelines)
-
CONTRAINDICATIONS
AVINZA is contraindicated in patients with known hypersensitivity to morphine, morphine salts, or any components of the product. AVINZA, like all opioids, is contraindicated in patients with respiratory depression in the absence of resuscitative equipment and in patients with acute or severe bronchial asthma.
AVINZA, like all opioids, is contraindicated in any patient who has or is suspected of having paralytic ileus.
-
WARNINGS
AVINZA must be swallowed whole (not chewed, crushed, or dissolved) or AVINZA may be opened and the entire bead contents sprinkled on a small amount of applesauce immediately prior to ingestion. THE CAPSULES MUST NOT BE CHEWED, CRUSHED, OR DISSOLVED DUE TO THE RISK OF RAPID RELEASE AND ABSORPTION OF A POTENTIALLY FATAL DOSE OF MORPHINE. (see BOX WARNING,CLINICAL PHARMACOLOGY)
Patients must not consume alcoholic beverages while on AVINZA therapy. Additionally, patients must not use prescription or non-prescription medications containing alcohol while on AVINZA therapy. Consumption of alcohol while taking AVINZA may result in the rapid release and absorption of a potentially fatal dose of morphine.
THE DAILY DOSE OF AVINZA MUST BE LIMITED TO A MAXIMUM OF 1600 MG/DAY. AVINZA DOSES OF OVER 1600 MG/DAY CONTAIN A QUANTITY OF FUMARIC ACID THAT HAS NOT BEEN DEMONSTRATED TO BE SAFE, AND WHICH MAY RESULT IN SERIOUS RENAL TOXICITY.
Misuse, Abuse and Diversion of Opioids
Morphine is an opioid agonist and a Schedule II controlled substance. Such drugs are sought by drug abusers and people with addiction disorders. Diversion of Schedule II products is an act subject to criminal penalty.
Morphine can be abused in a manner similar to other opioid agonists, legal or illicit. This should be considered when prescribing or dispensing AVINZA in situations where the physician or pharmacist is concerned about an increased risk of misuse, abuse, or diversion.
Abuse of AVINZA by crushing, chewing, snorting, or injecting the dissolved product will result in the immediate release of the entire daily dose of the opioid and pose a significant risk to the abuser that could result in overdose and death. Intravenous abuse of a water extract of AVINZA may lead to serious pulmonary complications due to the extraction of talc along with morphine sulfate. (see DRUG ABUSE AND ADDICTION)
Concerns about abuse, addiction, and diversion should not prevent the proper management of pain. Healthcare professionals should contact their State Professional Licensing Board, or State Controlled Substances Authority for information on how to prevent and detect abuse or diversion of this product.
Interactions with Alcohol and Drugs of Abuse
Morphine may be expected to have additive effects when used in conjunction with alcohol, other opioids, or illicit drugs that cause central nervous system depression. In vitro studies performed by the FDA demonstrated that when AVINZA 30 mg was mixed with 900 mL of buffer solutions containing ethanol (20% and 40%), the dose of morphine that was released was alcohol concentration-dependent, leading to a more rapid release of morphine. While the relevance of in vitro lab tests regarding AVINZA to the clinical setting remains to be determined, this acceleration of release may correlate with in vivo rapid release of the total morphine dose, which could result in the absorption of a potentially fatal dose of morphine.
Impaired Respiration
Respiratory depression is the chief hazard of all morphine preparations. Respiratory depression occurs more frequently in elderly or debilitated patients and in those suffering from conditions accompanied by hypoxia, hypercapnia, or upper airway obstruction, in whom even moderate therapeutic doses may significantly decrease pulmonary ventilation.
Morphine should be used with extreme caution in patients with chronic obstructive pulmonary disease or cor pulmonale and in patients having a substantially decreased respiratory reserve (e.g., severe kyphoscoliosis), hypoxia, hypercapnia, or pre-existing respiratory depression. In such patients, even usual therapeutic doses of morphine may increase airway resistance and decrease respiratory drive to the point of apnea.
Head Injury and Increased Intracranial Pressure
The respiratory depressant effects of morphine with carbon dioxide retention and secondary elevation of cerebrospinal fluid pressure may be markedly exaggerated in the presence of head injury, other intracranial lesions, or a pre-existing increase in intracranial pressure. Morphine produces effects which may obscure neurologic signs of further increases in intracranial pressure in patients with head injuries. Morphine should only be administered under such circumstances when considered essential and then with extreme care.
Hypotensive Effect
AVINZA, like all morphine products, may cause severe hypotension in an individual whose ability to maintain blood pressure has already been compromised by a depleted blood volume or concurrent administration of drugs such as phenothiazines or general anesthetics. (see also PRECAUTIONS, Drug Interactions) AVINZA may produce orthostatic hypotension and syncope in ambulatory patients.
AVINZA is an opioid analgesic which should be administered with caution to patients in circulatory shock, as vasodilation produced by the drug may further reduce cardiac output and blood pressure.
-
PRECAUTIONS
General
AVINZA is intended for use in patients requiring continuous around-the-clock treatment with an opioid analgesic. It is not appropriate as a prn treatment for pain. As with any opioid, it is critical to adjust the dose of AVINZA for each individual patient, taking into account the patient’s prior experience with analgesics. (see DOSAGE AND ADMINISTRATION)
Use in Pancreatic/Biliary Tract Disease
AVINZA should be used with caution in patients with biliary tract disease, including acute pancreatitis, as morphine may cause spasm of the sphincter of Oddi and diminish biliary and pancreatic secretions.
Special Risk Groups
AVINZA should be administered cautiously and in reduced dosages in patients with severe renal or hepatic insufficiency, Addison's disease, hypothyroidism, prostatic hypertrophy, or urethral stricture, and in elderly or debilitated patients. (see Geriatric Use and CLINICAL PHARMACOLOGY, Special Populations)
Caution should be exercised in the administration of morphine to patients with CNS depression, toxic psychosis, acute alcoholism and delirium tremens, and seizure disorders.
Driving and Operating Machinery
Patients should be cautioned that AVINZA could impair the mental and/or physical abilities needed to perform potentially hazardous activities such as driving a car or operating machinery.
Patients should also be cautioned about the potential combined effects of AVINZA with other CNS depressants, including other opioids, phenothiazines, sedative/hypnotics and alcohol. (see PRECAUTIONS, Drug Interactions)
Tolerance and Physical Dependence
Tolerance is the need for increasing doses of opioids to maintain a defined effect such as analgesia (in the absence of disease progression or other external factors). Physical dependence is manifested by withdrawal symptoms after abrupt discontinuation of a drug or upon administration of an antagonist. Physical dependence and tolerance are not unusual during chronic opioid therapy.
The opioid abstinence or withdrawal syndrome is characterized by some or all of the following: restlessness, lacrimation, rhinorrhea, yawning, perspiration, chills, myalgia, and mydriasis. Other symptoms also may develop, including irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate.
In general, opioids should not be abruptly discontinued. (see DOSAGE AND ADMINISTRATION, Cessation of Therapy)
Information for Patients
Patients receiving AVINZA (morphine sulfate extended-release capsules) should be given the following instructions by the physician:
- Patients should be advised that AVINZA capsules contain morphine and should be taken once daily.
- AVINZA must be swallowed whole (not chewed, crushed, or dissolved) or AVINZA may be opened and the entire bead contents sprinkled on a small amount of applesauce immediately prior to ingestion. The beads must NOT be chewed, crushed, or dissolved due to the risk of exposure to a potentially toxic dose of morphine.
- Patients should be informed that they must not consume alcoholic beverages while on AVINZA therapy. Additionally, patients should be informed that they must not use prescription or non-prescription medication containing alcohol while on AVINZA therapy. Consumption of alcohol while taking AVINZA may result in the rapid release and absorption of a potentially fatal dose of morphine.
- The dose of AVINZA should not be adjusted without consulting with a physician or other healthcare professional.
- Patients should be advised that AVINZA may impair mental and/or physical ability required for the performance of potentially hazardous tasks (e.g., driving, operating machinery). Patients started on AVINZA or patients whose dose has been adjusted should refrain from any potentially dangerous activity until it is established that they are not adversely affected.
- Patients should be advised that AVINZA should not be combined with alcohol or other CNS depressants (e.g., sleep medications, tranquilizers). A physician should be consulted if other medications are currently being used or are added in the future.
- Women of childbearing potential who become or are planning to become pregnant should consult a physician prior to initiating or continuing therapy with AVINZA.
- If patients have been receiving treatment with AVINZA for more than a few weeks and cessation of therapy is indicated, they should be counseled on the importance of safely tapering the dose and that abruptly discontinuing the medication could precipitate withdrawal symptoms. The physician should provide a dose schedule to accomplish a gradual discontinuation of the medication
- Patients should be advised that AVINZA is a potential drug of abuse. They should protect it from theft. It should never be given to anyone other than the individual for whom it was prescribed.
- Patients should be instructed to keep AVINZA in a secure place out of the reach of children. When AVINZA is no longer needed, the unused capsules should be destroyed by flushing down the toilet.
As with other opioids, patients taking AVINZA should be advised of the potential for severe constipation; appropriate laxatives, and/or stool softeners as well as other appropriate treatments should be initiated from the onset of opioid therapy.
Drug Interactions
CNS Depressants: The concurrent use of other central nervous system (CNS) depressants including sedatives, hypnotics, general anesthetics, antiemetics, phenothiazines, or other tranquilizers or alcohol increases the risk of respiratory depression, hypotension, profound sedation, or coma. Use with caution and in reduced dosages in patients taking these agents.
Muscle Relaxants: Morphine may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression.
Mixed Agonist/Antagonist Opioid Analgesics: Mixed agonist/antagonist analgesics (i.e., pentazocine, nalbuphine and butorphanol) should NOT be administered to patients who have received or are receiving a course of therapy with a pure opioid agonist analgesic. In these patients, mixed agonist/antagonist analgesics may reduce the analgesic effect and/or may precipitate withdrawal symptoms.
Monoamine Oxidase Inhibitors (MAOIs): MAOIs markedly potentiate the action of morphine. AVINZA should not be used in patients taking MAOIs or within 14 days of stopping such treatment.
Carcinogenicity/Mutagenicity/Impairment of Fertility
Studies in animals to evaluate the carcinogenic potential of morphine sulfate have not been conducted. No formal studies to assess the mutagenic potential of morphine have been conducted. In the published literature, the results of in vitro studies showed that morphine is non‑mutagenic in the Drosophila melanogaster lethal mutation assay and produced no evidence of chromosomal aberrations when incubated with murine splenocytes. Contrary to these results, morphine was found to increase DNA fragmentation when incubated in vitro with a human lymphoma cell line. In vivo, morphine has been reported to produce an increase in the frequency of micronuclei in bone marrow cells and immature red blood cells in the mouse micronucleus test and to induce chromosomal aberrations in murine lymphocytes and spermatids. Some of the in vivo clastogenic effects reported with morphine in mice may be directly related to increases in glucocorticoid levels produced by morphine in this species.
Pregnancy
Teratogenic Effects (Pregnancy Category C)
No formal studies to assess the teratogenic effects of morphine in animals have been performed. Several literature reports indicate that morphine administered subcutaneously during the early gestational period in mice and hamsters produced neurological, soft tissue and skeletal abnormalities. With one exception, the effects that have been reported were following doses that were maternally toxic and the abnormalities noted were characteristic of those observed when maternal toxicity is present. In one study, following subcutaneous infusion of doses greater than or equal to 0.15 mg/kg to mice, exencephaly, hydronephrosis, intestinal hemorrhage, split supraoccipital, malformed sternebrae, and malformed xiphoid were noted in the absence of maternal toxicity. In the hamster, morphine sulfate given subcutaneously on gestation day 8 produced exencephaly and cranioschisis. Morphine was not a significant teratogen in the rat at exposure levels significantly beyond that normally encountered in clinical practice. In one study however, decreased litter size and viability were observed in the offspring of male rats administered morphine at doses approximately 3-fold the maximum recommended human daily dose (MRHDD) for 10 days prior to mating. In two studies performed in the rabbit, no evidence of teratogenicity was reported at subcutaneous doses up to 100 mg/kg.
In humans, the frequency of congenital anomalies has been reported to be no greater than expected among the children of 70 women who were treated with morphine during the first four months of pregnancy or in 448 women treated with this drug anytime during pregnancy. Furthermore, no malformations were observed in the infant of a woman who attempted suicide by taking an overdose of morphine and other medication during the first trimester of pregnancy.
Nonteratogenic Effects
Published literature has reported that exposure to morphine during pregnancy is associated with reduction in growth and a host of behavioral abnormalities in the offspring of animals. Morphine treatment during gestational periods of organogenesis in rats, hamsters, guinea pigs and rabbits resulted in the following treatment-related embryotoxicity and neonatal toxicity in one or more studies: decreased litter size, embryo-fetal viability, fetal and neonatal body weights, absolute brain and cerebellar weights, lengths or widths at birth and during the neonatal period, delayed motor and sexual maturation, and increased neonatal mortality, cyanosis and hypothermia. Decreased fertility in female offspring, and decreased plasma and testicular levels of luteinizing hormone and testosterone, decreased testes weights, seminiferous tubule shrinkage, germinal cell aplasia, and decreased spermatogenesis in male offspring were also observed. Behavioral abnormalities resulting from chronic morphine exposure of fetal animals included altered reflex and motor skill development, mild withdrawal, and altered responsiveness to morphine persisting into adulthood.
Controlled studies of chronic in utero morphine exposure in pregnant women have not been conducted. Infants born to mothers who have taken opioids chronically may exhibit withdrawal symptoms, reversible reduction in brain volume, small size, decreased ventilatory response to CO2 and increased risk of sudden infant death syndrome. Morphine sulfate should be used by a pregnant woman only if the need for opioid analgesia clearly outweighs the potential risks to the fetus.
Labor and Delivery
Opioids cross the placenta and may produce respiratory depression and psycho-physiologic effects in neonates. AVINZA is not recommended for use in women during and immediately prior to labor, when use of shorter acting analgesics or other analgesic techniques are more appropriate. Occasionally, opioid analgesics may prolong labor through actions which temporarily reduce the strength, duration and frequency of uterine contractions. However this effect is not consistent and may be offset by an increased rate of cervical dilatation, which tends to shorten labor. Neonates whose mothers received opioid analgesics during labor should be observed closely for signs of respiratory depression. A specific opioid antagonist, such as naloxone or nalmefene, should be available for reversal of opioid-induced respiratory depression in the neonate.
Neonatal Withdrawal Syndrome
Chronic maternal use of opioids during pregnancy may cause newborns to suffer from neonatal withdrawal syndrome (NWS) following birth. Manifestations of this syndrome include irritability, hyperactivity, abnormal sleep pattern, high-pitched cry, tremor, vomiting, diarrhea, weight loss, and failure to gain weight. The time and amount of the mother’s last dose, and the rate of elimination of the drug from the newborn may affect the onset, duration, and severity of the disorder. When severe symptoms occur, pharmacologic intervention may be required.
Nursing Mothers
Low levels of morphine sulfate have been detected in human milk. Breast-feeding infants might experience withdrawal symptoms upon cessation of AVINZA administration to the mother. Because of the potential for nursing infants to experience adverse reactions, a decision should be made whether to discontinue nursing or discontinue AVINZA, taking into account the benefit of the drug to the mother.
Pediatric Use
Safety and effectiveness of AVINZA in pediatric patients below the age of 18 have not been established. The range of dose strengths available may not be appropriate for treatment of very young pediatric patients. Sprinkling on applesauce is NOT a suitable alternative for these patients.
Geriatric Use
Of the total number of subjects in clinical studies of AVINZA, there were 168 patients age 65 and over, including 64 patients over the age of 74, 100 of whom were treated with AVINZA. Subgroup analyses comparing efficacy were not possible given the small number of subjects in each treatment group. No overall differences in safety were observed between these subjects and younger subjects. In general, caution should be exercised in the selection of the starting dose of AVINZA for an elderly patient, usually starting at the low end of the dosing range. As with all opioids, the starting dose should be reduced in debilitated and non-tolerant patients. (see CLINICAL PHARMACOLOGY, Special Populations, Geriatric and PRECAUTIONS, Special Risk Groups)
-
ADVERSE REACTIONS
In controlled and open label clinical studies, 560 patients with chronic malignant or non-malignant pain were treated with AVINZA. The most common serious adverse events reported with administration of AVINZA were vomiting, nausea, death, dehydration, dyspnea, and sepsis. (Deaths occurred in patients treated for pain due to underlying malignancy.) Serious adverse events caused by morphine include respiratory depression, apnea, and to a lesser degree, circulatory depression, respiratory arrest, shock and cardiac arrest.
Adverse Events
The common adverse events seen on initiation of therapy with morphine are dose-dependent and are typical opioid‑related side effects. The most frequent of these include constipation, nausea and somnolence. The frequency of these events depends upon several factors including the clinical setting, the patient’s level of opioid tolerance, and host factors specific to the individual. These events should be anticipated and managed as part of opioid analgesia therapy.
The most common adverse events (seen in greater than 10%) reported by patients treated with AVINZA during the clinical trials at least once during therapy were constipation, nausea, somnolence, vomiting, and headache. Adverse events occurring in 5‑10% of study patients were peripheral edema, diarrhea, abdominal pain, infection, urinary tract infection, accidental injury, flu syndrome, back pain, rash, sweating, fever, insomnia, depression, paresthesia, anorexia, dry mouth, asthenia and dyspnea. Other less common side effects expected from opioid analgesics, including morphine, or seen in fewer than 5% of patients taking AVINZA in the clinical trials were:
Body as a Whole: malaise, withdrawal syndrome.
Cardiovascular System: bradycardia, hypertension, hypotension, palpitations, syncope, tachycardia.
Digestive System: biliary pain, dyspepsia, dysphagia, gastroenteritis, abnormal liver function tests, rectal disorder, thirst.
Hemic and Lymphatic System: anemia, thrombocytopenia.
Metabolic and Nutritional Disorders: edema, weight loss.
Musculoskeletal: skeletal muscle rigidity.
Nervous System: abnormal dreams, abnormal gait, agitation, amnesia, anxiety, ataxia, confusion, convulsions, coma, delirium, euphoria, hallucinations, lethargy, nervousness, abnormal thinking, tremor, vasodilation, vertigo.
Respiratory System: hiccup, hypoventilation, voice alteration.
Skin and Appendages: dry skin, urticaria.
Special Senses: amblyopia, eye pain, taste perversion.
Urogenital System: abnormal ejaculation, dysuria, impotence, decreased libido, oliguria, urinary retention.
-
DRUG ABUSE AND ADDICTION
AVINZA is a mu-agonist opioid and is a Schedule II controlled substance. Morphine, like other opioids used in analgesia, can be abused and is subject to criminal diversion.
Drug addiction is characterized by compulsive use, use for non-medical purposes, and continued use despite harm or risk of harm. Drug addiction is a treatable disease, utilizing a multi-disciplinary approach, but relapse is common.
“Drug‑seeking” behavior is very common in addicts and drug abusers. Drug-seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing or referral, repeated “loss” of prescriptions, tampering with prescriptions and reluctance to provide prior medical records or contact information for other treating physician(s). “Doctor shopping” to obtain additional prescriptions is common among drug abusers and people suffering from untreated addiction.
Abuse and addiction are separate and distinct from physical dependence and tolerance. Physicians should be aware that addiction may not be accompanied by concurrent tolerance and symptoms of physical dependence. The converse is also true. In addition, abuse of opioids can occur in the absence of true addiction and is characterized by misuse for non-medical purposes, often in combination with other psychoactive substances. Careful record-keeping of prescribing information, including quantity, frequency, and renewal requests is strongly advised.
Proper assessment of the patient, proper prescribing practices, periodic re-evaluation of therapy, and proper dispensing and storage are appropriate measures that help to limit abuse of opioid drugs.
AVINZA is intended for oral use only. Abuse of the crushed capsule poses a hazard of overdose and death. This risk is increased with concurrent abuse of alcohol and other substances. With parenteral abuse, the capsule excipients, especially talc, can be expected to result in local tissue necrosis, infection, pulmonary granulomas, and increased risk of endocarditis and valvular heart injury. Parenteral drug abuse is commonly associated with transmission of infectious diseases such as hepatitis and HIV.
-
AVINZA OVERDOSAGE
Symptoms
Acute overdosage with morphine is manifested by respiratory depression, somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, constricted pupils, and, in some cases, pulmonary edema, bradycardia, hypotension, and death.
Treatment
Primary attention should be given to re-establishment of a patent airway and institution of assisted or controlled ventilation when overdose of an extended‑release formulation such as AVINZA has been ingested. Elimination or evacuation of gastric contents may be necessary in order to eliminate unabsorbed drug. Before attempting treatment by gastric emptying or activated charcoal, care should be taken to secure the airway. Pure opioid antagonists, naloxone or nalmefene, are specific antidotes to respiratory depression resulting from opioid overdose. Since the duration of reversal is expected to be less than the duration of action of AVINZA, the patient must be carefully monitored until spontaneous respiration is reliably re-established. AVINZA, as with other controlled delivery preparations in overdose situations, may continue to release morphine for 36 to 48 hours or longer following ingestion, and management of an overdose should be monitored accordingly. If the response to opioid antagonists is sub-optimal or only brief in nature, additional antagonist should be administered as directed by the manufacturer of the product.
Opioid antagonists should not be administered in the absence of clinically significant respiratory or circulatory depression secondary to morphine overdose. Such agents should be administered cautiously to persons who are known, or suspected to be physically dependent on AVINZA. In such cases, an abrupt or complete reversal of opioid effects may precipitate an acute abstinence syndrome.
Opioid-Tolerant Individuals: In an individual physically dependent on opioids, administration of the usual dose of the antagonist will precipitate an acute withdrawal syndrome. The severity of the withdrawal symptoms experienced will depend on the degree of physical dependence and the dose of the antagonist administered. Use of an opioid antagonist should be reserved for cases where such treatment is clearly needed. If it is necessary to treat serious respiratory depression in the physically dependent patient, administration of the antagonist should be initiated with care and titrated with smaller than usual doses.
Supportive measures (including oxygen, vasopressors) should be employed in the management of circulatory shock and pulmonary edema as indicated. Cardiac arrest or arrhythmias may require cardiac massage or defibrillation.
-
DOSAGE AND ADMINISTRATION
AVINZA MUST BE SWALLOWED WHOLE (NOT CHEWED, CRUSHED, OR DISSOLVED) OR AVINZA MAY BE OPENED AND THE ENTIRE BEAD CONTENTS SPRINKLED ON A SMALL AMOUNT OF APPLESAUCE IMMEDIATELY PRIOR TO INGESTION. THE BEADS MUST NOT BE CHEWED, CRUSHED, OR DISSOLVED DUE TO RISK OF ACUTE OVERDOSE. INGESTING CHEWED OR CRUSHED AVINZA BEADS WILL LEAD TO THE RAPID RELEASE AND ABSORPTION OF A POTENTIALLY TOXIC DOSE OF MORPHINE.
Patients must not consume alcoholic beverages while on AVINZA therapy. Additionally, patients must not use prescription or non-prescription medicine containing alcohol while on AVINZA therapy. Consumption of alcohol while taking AVINZA may result in the rapid release and absorption of a potentially fatal dose of morphine.
The daily dose of AVINZA must be limited to a maximum of 1600 mg/day. AVINZA doses of over 1600 mg/day contain a quantity of fumaric acid that has not been demonstrated to be safe, and which may result in serious renal toxicity. (see WARNINGS)
The 60, 90, and 120 mg capsules are for use only in opioid‑tolerant patients.
All doses are intended to be administered once daily. As with any opioid drug product, it is necessary to adjust the dosing regimen for each patient individually, taking into account the patient's prior analgesic treatment experience. In the selection of the initial dose of AVINZA, attention should be given to the following:
- the total daily dose, potency and specific characteristics of the opioid the patient has been taking previously;
- the reliability of the relative potency estimate used to calculate the equivalent morphine dose needed;
- the patient’s degree of opioid tolerance;
- the general condition and medical status of the patient;
- concurrent medications;
- the type and severity of the patient's pain.
The following dosing recommendations, therefore, can only be considered suggested approaches to what is actually a series of clinical decisions over time in the management of the pain of each individual patient.
Conversion from Other Oral Morphine Formulations to AVINZA
Patients receiving other oral morphine formulations may be converted to AVINZA by administering the patient's total daily oral morphine dose as AVINZA once-daily. AVINZA should not be given more frequently than every 24 hours. As with conversion from any oral morphine formulation to another, supplemental pain medication may be required until the response to the patient's daily AVINZA dosage has stabilized (up to 4 days).
Conversion from Parenteral Morphine or Other Non-Morphine Opioids (Parenteral or Oral) to AVINZA
There is inter-patient variability in the potency of opioid drugs and opioid formulations. Therefore, a conservative approach is advised when determining the total daily dose of AVINZA. It is better to underestimate a patient’s 24-hour oral morphine dose and make available rescue medication than to overestimate the 24-hour oral morphine dose and manage an adverse experience or overdose. The following general points should be considered regarding opioid conversions.
Parenteral to oral morphine ratio: Anywhere from 3 to 6 mg of oral morphine may be required to provide pain relief equivalent to 1 mg of parenteral morphine. Based on this rationale, a reasonable starting dose of AVINZA would be approximately three times the previous daily parenteral morphine requirement.
Other parenteral or oral non-morphine opioids to oral morphine sulfate: Physicians and other healthcare professionals are advised to refer to published relative potency information, keeping in mind that conversion ratios are only approximate. In general, it is safest to administer half of the estimated daily morphine requirement as the initial AVINZA dose once per day and then manage insufficient pain relief by supplementation with immediate‑release morphine or other short-acting analgesics. (see Individualization of Dosage)
Individualization of Dosage
Physicians should individualize treatment using a progressive plan of pain management such as outlined by the World Health Organization, the American Pain Society and the Federation of State Medical Boards Model Guidelines. Healthcare professionals should follow appropriate pain management principles of careful assessment and ongoing monitoring. AVINZA (morphine sulfate) is on the third step of the WHO three step analgesic ladder and is of most benefit when a constant level of opioid analgesia is used as a platform from which break-through pain is managed. Once acceptable pain relief is no longer achieved from combinations of non-opioid medications (NSAIDs and acetaminophen) and intermittent usage of moderate or strong opioids, conversion to a 24-hour oral morphine equivalent is warranted.
The dose may be titrated as frequently as every other day to control analgesia. In the event that break-through pain occurs, AVINZA may be supplemented with a small dose (5-15% of the total daily dose of morphine) of a short-acting analgesic.
When AVINZA is chosen as the initial opioid for patients who do not have a proven tolerance to opioids, patients should be treated initially at a dose of 30 mg once-daily (at 24-hour intervals). For opioid-naïve patients, the dose should be increased conservatively. For such patients, it is recommended that the dose of AVINZA be adjusted in increments not greater than 30 mg every 4 days. Some degree of tolerance may occur, requiring dosage adjustment until the achievement of a balance between analgesia and opioid side effects. When necessary, the total dose of AVINZA should be increased until pain relief is reached or clinically significant opioid-related adverse reactions occur.
Alternative Methods of Administration
AVINZA beads sprinkled over applesauce were found to be bioequivalent to AVINZA capsules swallowed whole under fasting conditions in a study of healthy volunteers. Absorption of the beads sprinkled on other foods has not been tested. This method of administration may be beneficial for patients who have difficulty swallowing whole capsules or tablets.
- Sprinkle the entire contents of the capsule(s) onto a small amount of applesauce. The applesauce should be at room temperature or cooler. Use immediately. (see also CLINICAL PHARMACOLOGY, Food Effects)
- Swallow mixture without chewing or crushing beads.
- Rinse mouth and swallow to ensure all beads have been ingested.
- Patients should consume the entire portion and should not divide applesauce into separate doses.
Conversion from AVINZA to Other Pain Control Therapies
It is important to remember that the persistence of AVINZA-derived plasma morphine concentrations may be in excess of 36 hours when making a conversion to other pain control therapies.
Conversion from AVINZA to Other Controlled-Release Oral Morphine Formulations
For a given dose, the same total amount of morphine is available from AVINZA as from oral morphine solution or controlled-release morphine tablets. The extended duration of release of morphine from AVINZA results in reduced maximum and increased minimum plasma morphine concentrations than with shorter acting morphine products. Conversion from AVINZA to the same total daily dose of another controlled-release morphine formulation could lead to either excessive sedation at peak serum levels or inadequate analgesia at trough serum levels. Dosage adjustment with close observation is recommended.
Conversion from AVINZA to Parenteral Opioids
When converting from AVINZA to parenteral opioids, it is best to calculate an equivalent parenteral dose and then initiate treatment at half of this calculated value. As an example, an estimated total 24-hour parenteral morphine requirement of a patient receiving AVINZA is one-third of the dose of AVINZA. This is because the oral bioavailability of morphine is one-third that of parenteral morphine. This estimated dose should then be divided in half, and this last calculated dose is the total daily dose. This value should be further divided by six if the desire is to dose with parenteral morphine every four hours.
Consider a patient taking 360 mg of AVINZA daily. First, divide by 3, to account for differences in bioavailability between oral and parenteral morphine. This new figure, 120 mg, is the estimated total 24-hour requirement of parenteral morphine. Dividing by 2, the result gives the total daily dose of 60 mg. If it is decided to administer the drug at four-hour intervals, then administer 10 mg (60 divided by 6) every four hours.
Although this approach may require a dosage increase in the first 24 hours for many patients, this method is recommended, as it is less likely to result in overdose. Overdose is more likely to occur when administering an equivalent dose of parenteral morphine without titration. Provision for break-through pain should be made.
- SAFETY AND HANDLING
-
HOW SUPPLIED
30 mg Capsule: size 3 capsule, yellow cap imprinted AVINZA and white, opaque body imprinted 30 mg and 505.
NDC 64365-505-03: Bottles of 100 capsules.
60 mg Capsule: size 3 capsule, bluish‑green cap imprinted AVINZA and white, opaque body imprinted 60 mg and 506.
NDC 64365-506-03: Bottles of 100 capsules.
90 mg Capsule: size 1 capsule, red cap imprinted AVINZA and white, opaque body imprinted 90 mg and 507.
NDC 64365-507-02: Bottles of 100 capsules.
120 mg Capsule: size 1 capsule, blue‑violet cap imprinted AVINZA and white, opaque body imprinted 120 mg and 508.
NDC 64365-508-02: Bottles of 100 capsules.
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F). [see USP Controlled Room Temperature]
Protect from light and moisture.
Dispense in a tight, light-resistant container as defined in USP.
CAUTION: DEA Order Form Required.
Rx Only.
Manufactured for:
Ligand Pharmaceuticals Incorporated
San Diego, CA 92121
AVINZA® Information Service: 1-888-8-AVINZA
Utilizing technology developed by:
Elan Corporation plc.
Monksland, Athlone
Co Westmeath, Ireland
Rev. 12/05
AVINZA® is a registered trademark of Ligand® Pharmaceuticals Inc.
SODAS® is a registered trademark of Elan Corporation, plc.
U.S. Patent No.: 6,066,339
-
INGREDIENTS AND APPEARANCE
AVINZA
morphine sulfate capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:64365-505 Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength morphine sulfate (UNII: X3P646A2J0) (morphine - UNII:76I7G6D29C) 30 mg Inactive Ingredients Ingredient Name Strength ammonio-methacrylate copolymers () fumaric acid () povidone () sodium lauryl sulfate (UNII: 368GB5141J) sugar starch spheres () talc (UNII: 7SEV7J4R1U) Product Characteristics Color YELLOW (YELLOW) , WHITE (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 16mm Flavor Imprint Code AVINZA;30;mg;505 Contains Coating false Symbol false Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64365-505-03 100 in 1 BOTTLE AVINZA
morphine sulfate capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:64365-506 Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength morphine sulfate (UNII: X3P646A2J0) (morphine - UNII:76I7G6D29C) 60 mg Inactive Ingredients Ingredient Name Strength ammonio-methacrylate copolymers () fumaric acid () povidone () sodium lauryl sulfate (UNII: 368GB5141J) sugar starch spheres () talc (UNII: 7SEV7J4R1U) Product Characteristics Color GREEN (BLUISH-GREEN) , BLUE (BLUISH-GREEN) , WHITE (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 16mm Flavor Imprint Code AVINZA;60;mg;506 Contains Coating false Symbol false Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64365-506-03 100 in 1 BOTTLE AVINZA
morphine sulfate capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:64365-507 Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength morphine sulfate (UNII: X3P646A2J0) (morphine - UNII:76I7G6D29C) 90 mg Inactive Ingredients Ingredient Name Strength sugar starch spheres () fumaric acid () povidone () sodium lauryl sulfate (UNII: 368GB5141J) ammonio-methacrylate copolymers () talc (UNII: 7SEV7J4R1U) Product Characteristics Color RED (RED) , WHITE (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 19mm Flavor Imprint Code AVINZA;90;mg;507 Contains Coating false Symbol false Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64365-507-02 100 in 1 BOTTLE AVINZA
morphine sulfate capsule, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:64365-508 Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength morphine sulfate (UNII: X3P646A2J0) (morphine - UNII:76I7G6D29C) 120 mg Inactive Ingredients Ingredient Name Strength ammonio-methacrylate copolymers () fumaric acid () povidone () sodium lauryl sulfate (UNII: 368GB5141J) sugar starch spheres () talc (UNII: 7SEV7J4R1U) Product Characteristics Color BLUE (BLUE) , PURPLE (VIOLET) , WHITE (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 19mm Flavor Imprint Code AVINZA;120;mg;508 Contains Coating false Symbol false Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64365-508-02 100 in 1 BOTTLE Labeler - Ligand Pharmaceuticals Incorporated
