Label: CEFOXITIN- cefoxitin sodium powder, for solution
- NDC Code(s): 63323-341-01, 63323-341-25, 63323-342-01, 63323-342-25
- Packager: Fresenius Kabi USA, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated September 23, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTION
451232E/ Revised: May 2021
Cefoxitin
for Injection, USP Rx only
To reduce the development of drug-resistant bacteria and maintain the effectiveness of CEFOXITIN FOR INJECTION and other antibacterial drugs, CEFOXITIN FOR INJECTION should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
-
DESCRIPTION:
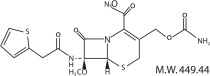
CEFOXITIN FOR INJECTION, USP contains cefoxitin sodium a semi-synthetic, broad-spectrum cephalosporin antibiotic for parenteral administration. It is derived from cephalosporin C, which is produced by Cephalosporium Acremonium. It is the sodium salt of 3-(hydroxymethyl)-7-methoxy-8-oxo-7-[2-(2-thienyl)acetamido]-5-thia-1-azabicyclo [4.2.0] oct-2-ene-2-carboxylate carbamate (ester). The molecular formula is C16H16N3NaO7S2, and the structural formula is:
CEFOXITIN FOR INJECTION, USP contains approximately 53.8 mg (2.3 milliequivalents) of sodium per gram of cefoxitin activity.
Solutions of CEFOXITIN FOR INJECTION, USP range from colorless to light amber in color.
The pH of freshly constituted solutions usually ranges from 4.2 to 7.0.
Each conventional vial contains sterile cefoxitin sodium, USP equivalent to 1 gram or 2 grams cefoxitin.
-
CLINICAL PHARMACOLOGY:
Clinical Pharmacology:
Following an intravenous dose of 1 gram, serum concentrations were 110 mcg/mL at 5 minutes, declining to less than 1 mcg/mL at 4 hours. The half-life after an intravenous dose is 41 to 59 minutes.
Approximately 85 percent of cefoxitin is excreted unchanged by the kidneys over a 6-hour period, resulting in high urinary concentrations. Probenecid slows tubular excretion and produces higher serum levels and increases the duration of measurable serum concentrations.
Cefoxitin passes into pleural and joint fluids and is detectable in antibacterial concentrations in bile.
In a published study of geriatric patients ranging in age from 64 to 88 years with normal renal function for their age (creatinine clearance ranging from 31.5 to 174.0 mL/min), the half-life for cefoxitin ranged from 51 to 90 minutes, resulting in higher plasma concentrations than in younger adults. These changes were attributed to decreased renal function associated with the aging process.
Microbiology:
Mechanism of Action:
Cefoxitin is a bactericidal agent that acts by inhibition of bacterial cell wall synthesis. Cefoxitin has activity in the presence of some beta-lactamases, both penicillinases and cephalosporinases, of Gram-negative and Gram-positive bacteria.
Mechanism of Resistance:
Resistance to cefoxitin is primarily through hydrolysis by beta-lactamase, alteration of penicillin-binding proteins (PBPs), and decreased permeability.
Cefoxitin has been shown to be active against most isolates of the following bacteria, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section:
Gram-positive bacteria
- Staphylococcus aureus (methicillin-susceptible isolates only)
- Staphylococcus epidermidis (methicillin-susceptible isolates only)
- Streptococcus agalactiae
- Streptococcus pneumoniae
- Streptococcus pyogenes
Gram-negative bacteria
- Escherichia coli
- Haemophilus influenzae
- Klebsiella spp.
- Morganella morganii
- Neisseria gonorrhoeae
- Proteus mirabilis
- Proteus vulgaris
- Providencia spp.
Anaerobic bacteria
- Clostridium spp.
- Peptococcus niger
- Peptostreptococcus spp.
- Bacteroides spp.
The following in vitro data are available, but their clinical significance is unknown. At least 90 percent of the following microorganisms exhibit an in vitro minimum inhibitory concentration (MIC) less than or equal to the susceptible breakpoint for cefoxitin. However, the efficacy of cefoxitin in treating clinical infections due to these microorganisms has not been established in adequate and well-controlled clinical trials.
Gram-negative bacteria
- Eikenella corrodens (non-β-lactamase producers)
Anaerobic bacteria
- Clostridium perfringens
- Prevotella bivia
-
INDICATIONS AND USAGE:
Treatment:
CEFOXITIN FOR INJECTION, USP is indicated for the treatment of serious infections caused by susceptible strains of the designated microorganisms in the diseases listed below.
- (1) Lower respiratory tract infections, including pneumonia and lung abscess, caused by Streptococcus pneumoniae, other streptococci (excluding enterococci, e.g., Enterococcus faecalis [formerly Streptococcus faecalis]), Staphylococcus aureus (including penicillinase-producing strains), Escherichia coli, Klebsiella species, Haemophilus influenzae, and Bacteroides species.
- (2) Urinary tract infections caused by Escherichia coli, Klebsiella species, Proteus mirabilis, Morganella morganii, Proteus vulgaris and Providencia species (including P. rettgeri).
- (3) Intra-abdominal infections, including peritonitis and intra-abdominal abscess, caused by Escherichia coli, Klebsiella species, Bacteroides species including Bacteroides fragilis, and Clostridium species.
- (4) Gynecological infections, including endometritis, pelvic cellulitis, and pelvic inflammatory disease caused by Escherichia coli, Neisseria gonorrhoeae (including penicillinase-producing strains), Bacteroides species including B. fragilis, Clostridium species, Peptococcus niger, Peptostreptococcus species, and Streptococcus agalactiae. CEFOXITIN FOR INJECTION, USP, like cephalosporins, has no activity against Chlamydia trachomatis. Therefore, when CEFOXITIN FOR INJECTION, USP is used in the treatment of patients with pelvic inflammatory disease and C. trachomatis is one of the suspected pathogens, appropriate anti-chlamydial coverage should be added.
- (5) Septicemia caused by Streptococcus pneumoniae, Staphylococcus aureus (including penicillinase-producing strains), Escherichia coli, Klebsiella species, and Bacteroides species including B. fragilis.
- (6) Bone and joint infections caused by Staphylococcus aureus (including penicillinase-producing strains).
- (7) Skin and skin structure infections caused by Staphylococcus aureus (including penicillinase-producing strains), Staphylococcus epidermidis, Streptococcus pyogenes and other streptococci (excluding enterococci e.g., Enterococcus faecalis [formerly Streptococcus faecalis]), Escherichia coli, Proteus mirabilis, Klebsiella species, Bacteroides species including B. fragilis, Clostridium species, Peptococcus niger, and Peptostreptococcus species.
Appropriate culture and susceptibility studies should be performed to determine the susceptibility of the causative organisms to CEFOXITIN FOR INJECTION, USP. Therapy may be started while awaiting the results of these studies.
In randomized comparative studies, CEFOXITIN FOR INJECTION, USP and cephalothin were comparably safe and effective in the management of infections caused by gram-positive cocci and gram-negative rods susceptible to the cephalosporins. CEFOXITIN FOR INJECTION, USP has a high degree of stability in the presence of bacterial beta-lactamases, both penicillinases and cephalosporinases.
Many infections caused by aerobic and anaerobic gram-negative bacteria resistant to some cephalosporins respond to CEFOXITIN FOR INJECTION, USP. Similarly, many infections caused by aerobic and anaerobic bacteria resistant to some penicillin antibiotics (ampicillin, carbenicillin, penicillin G) respond to treatment with CEFOXITIN FOR INJECTION, USP. Many infections caused by mixtures of susceptible aerobic and anaerobic bacteria respond to treatment with CEFOXITIN FOR INJECTION, USP.
Prevention:
CEFOXITIN FOR INJECTION, USP is indicated for the prophylaxis of infection in patients undergoing uncontaminated gastrointestinal surgery, vaginal hysterectomy, abdominal hysterectomy, or cesarean section.
If there are signs of infection, specimens for culture should be obtained for identification of the causative organism so that appropriate treatment may be instituted.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of CEFOXITIN FOR INJECTION, USP and other antibacterial drugs, CEFOXITIN FOR INJECTION, USP should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
- CONTRAINDICATIONS:
-
WARNINGS:
BEFORE THERAPY WITH CEFOXITIN FOR INJECTION IS INSTITUTED, CAREFUL INQUIRY SHOULD BE MADE TO DETERMINE WHETHER THE PATIENT HAS HAD PREVIOUS HYPERSENSITIVITY REACTIONS TO CEFOXITIN, CEPHALOSPORINS, PENICILLINS, OR OTHER DRUGS. THIS PRODUCT SHOULD BE GIVEN WITH CAUTION TO PENICILLIN-SENSITIVE PATIENTS. ANTIBIOTICS SHOULD BE ADMINISTERED WITH CAUTION TO ANY PATIENT WHO HAS DEMONSTRATED SOME FORM OF ALLERGY, PARTICULARLY TO DRUGS. IF AN ALLERGIC REACTION TO CEFOXITIN FOR INJECTION OCCURS, DISCONTINUE THE DRUG. SERIOUS HYPERSENSITIVITY REACTIONS MAY REQUIRE EPINEPHRINE AND OTHER EMERGENCY MEASURES.
Clostridium difficile associated diarrhea (CDAD) has been reported with the use of nearly all antibacterial agents, including cefoxitin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
-
PRECAUTIONS:
General:
The total daily dose should be reduced when CEFOXITIN FOR INJECTION is administered to patients with transient or persistent reduction of urinary output due to renal insufficiency (see DOSAGE AND ADMINISTRATION), because high and prolonged serum antibiotic concentrations can occur in such individuals from usual doses.
Antibiotics (including cephalosporins) should be prescribed with caution in individuals with a history of gastrointestinal disease, particularly colitis.
As with other antibiotics, prolonged use of CEFOXITIN FOR INJECTION may result in overgrowth of nonsusceptible organisms. Repeated evaluation of the patient's condition is essential. If superinfection occurs during therapy, appropriate measures should be taken.
Prescribing CEFOXITIN FOR INJECTION in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Information for Patients:
Patients should be counseled that antibacterial drugs including CEFOXITIN FOR INJECTION should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When CEFOXITIN FOR INJECTION is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by CEFOXITIN FOR INJECTION or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibiotics, which usually ends when the antibiotic is discontinued. Sometimes after starting the treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic.
If this occurs, patients should contact their physician as soon as possible.
Laboratory Tests:
As with any potent antibacterial agent, periodic assessment of organ system functions, including renal, hepatic, and hematopoietic, is advisable during prolonged therapy.
Drug Interactions:
Increased nephrotoxicity has been reported following concomitant administration of cephalosporins and aminoglycoside antibiotics.
Drug/Laboratory Test Interactions:
As with cephalothin, high concentrations of cefoxitin (>100 mcg/mL) may interfere with measurement of serum and urine creatinine levels by the Jaffé reaction, and produce false increases of modest degree in the levels of creatinine reported. Serum samples from patients treated with cefoxitin should not be analyzed for creatinine if withdrawn within 2 hours of drug administration.
High concentrations of cefoxitin in the urine may interfere with measurement of urinary 17-hydroxy-corticosteroids by the Porter-Silber reaction, and produce false increases of modest degree in the levels reported.
A false-positive reaction for glucose in the urine may occur. This has been observed with CLINITEST† reagent tablets.
Carcinogenesis, Mutagenesis, Impairment of Fertility: Long-term studies in animals have not been performed with cefoxitin to evaluate carcinogenic or mutagenic potential. Studies in rats treated intravenously with 400 mg/kg of cefoxitin (approximately 3 times the maximum recommended human dose) revealed no effects on fertility or mating ability.
Pregnancy:
Reproduction studies performed in rats and mice at parenteral doses of approximately one to seven and one-half times the maximum recommended human dose did not reveal teratogenic or fetal toxic effects, although a slight decrease in fetal weight was observed.
There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
In the rabbit, cefoxitin was associated with a high incidence of abortion and maternal death. This was not considered to be a teratogenic effect but an expected consequence of the rabbit's unusual sensitivity to antibiotic-induced changes in the population of the microflora of the intestine.
Nursing Mothers:
Cefoxitin is excreted in human milk in low concentrations. Caution should be exercised when CEFOXITIN FOR INJECTION is administered to a nursing woman.
Pediatric Use:
Safety and efficacy in pediatric patients from birth to 3 months of age have not yet been established. In pediatric patients 3 months of age and older, higher doses of CEFOXITIN FOR INJECTION have been associated with an increased incidence of eosinophilia and elevated SGOT.
Geriatric Use:
Of the 1,775 subjects who received cefoxitin in clinical studies, 424 (24%) were 65 and over, while 124 (7%) were 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out (see CLINICAL PHARMACOLOGY). This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see DOSAGE AND ADMINISTRATION and PRECAUTIONS).
-
ADVERSE REACTIONS:
CEFOXITIN FOR INJECTION is generally well tolerated. The most common adverse reactions have been local reactions following intravenous injection. Other adverse reactions have been encountered infrequently.
Local Reactions:
Thrombophlebitis has occurred with intravenous administration.
Allergic Reactions:
Rash (including exfoliative dermatitis and toxic epidermal necrolysis), urticaria, flushing, pruritus, eosinophilia, fever, dyspnea, and other allergic reactions including anaphylaxis, interstitial nephritis and angioedema have been noted.
Cardiovascular:
Hypotension.
Gastrointestinal:
Diarrhea, including documented pseudomembranous colitis which can appear during or after antibiotic treatment. Nausea and vomiting have been reported rarely.
Neuromuscular:
Possible exacerbation of myasthenia gravis.
Blood:
Eosinophilia, leukopenia including granulocytopenia, neutropenia, anemia, including hemolytic anemia, thrombocytopenia, and bone marrow depression. A positive direct Coombs test may develop in some individuals, especially those with azotemia.
Liver Function:
Transient elevations in SGOT, SGPT, serum LDH, and serum alkaline phosphatase; and jaundice have been reported.
Renal Function:
Elevations in serum creatinine and/or blood urea nitrogen levels have been observed. As with the cephalosporins, acute renal failure has been reported rarely. The role of CEFOXITIN FOR INJECTION in changes in renal function tests is difficult to assess, since factors predisposing to prerenal azotemia or to impaired renal function usually have been present.
In addition to the adverse reactions listed above which have been observed in patients treated with CEFOXITIN FOR INJECTION, the following adverse reactions and altered laboratory test results have been reported for cephalosporin class antibiotics:
Urticaria, erythema multiforme, Stevens-Johnson syndrome, serum sickness-like reactions, abdominal pain, colitis, renal dysfunction, toxic nephropathy, false-positive test for urinary glucose, hepatic dysfunction including cholestasis, elevated bilirubin, aplastic anemia, hemorrhage, prolonged prothrombin time, pancytopenia, agranulocytosis, superinfection, vaginitis including vaginal candidiasis.
Several cephalosporins have been implicated in triggering seizures, particularly in patients with renal impairment when the dosage was not reduced (see DOSAGE AND ADMINISTRATION). If seizures associated with drug therapy occur, the drug should be discontinued. Anticonvulsant therapy can be given if clinically indicated.
To report SUSPECTED ADVERSE EVENTS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov.
- OVERDOSAGE:
-
DOSAGE AND ADMINISTRATION:
Treatment:
Adults:
The usual adult dosage range is 1 gram to 2 grams every 6 to 8 hours. Dosage should be determined by susceptibility of the causative organisms, severity of infection, and the condition of the patient (see Table 1 for dosage guidelines).
If C. trachomatis is a suspected pathogen, appropriate anti-chlamydial coverage should be added, because cefoxitin sodium has no activity against this organism.
CEFOXITIN FOR INJECTION may be used in patients with reduced renal function with the following dosage adjustments:
In adults with renal insufficiency, an initial loading dose of 1 gram to 2 grams may be given. After a loading dose, the recommendations for maintenance dosage (Table 2) may be used as a guide.
When only the serum creatinine level is available, the following formula (based on sex, weight, and age of the patient) may be used to convert this value into creatinine clearance. The serum creatinine should represent a steady state of renal function.
Males: Weight (kg) x (140-age)
72 x serum creatinine (mg/100 mL)Females: 0.85 x above value
In patients undergoing hemodialysis, the loading dose of 1 gram to 2 grams should be given after each hemodialysis, and the maintenance dose should be given as indicated in Table 2.
Antibiotic therapy for group A beta-hemolytic streptococcal infections should be maintained for at least 10 days to guard against the risk of rheumatic fever or glomerulonephritis. In staphylococcal and other infections involving a collection of pus, surgical drainage should be carried out where indicated.
Pediatric Patients:
The recommended dosage in pediatric patients 3 months of age and older is 80 to 160 mg/kg of body weight per day divided into four to six equal doses.
The higher dosages should be used for more severe or serious infections.
The total daily dosage should not exceed 12 grams.
At this time no recommendation is made for pediatric patients from birth to 3 months of age (see PRECAUTIONS).
In pediatric patients with renal insufficiency, the dosage and frequency of dosage should be modified consistent with the recommendations for adults (see Table 2).
Prevention:
Effective prophylactic use depends on the time of administration. CEFOXITIN FOR INJECTION usually should be given one-half to one hour before the operation, which is sufficient time to achieve effective levels in the wound during the procedure. Prophylactic administration should usually be stopped within 24 hours since continuing administration of any antibiotic increases the possibility of adverse reactions but, in the majority of surgical procedures, does not reduce the incidence of subsequent infection.
For prophylactic use in uncontaminated gastrointestinal surgery, vaginal hysterectomy, or abdominal hysterectomy, the following doses are recommended:
Adults:
2 grams administered intravenously just prior to surgery (approximately one-half to one hour before the initial incision) followed by 2 grams every 6 hours after the first dose for no more than 24 hours.
Pediatric Patients (3 Months and Older):
30 to 40 mg/kg doses may be given at the times designated above.
Cesarean Section Patients:
For patients undergoing cesarean section, either a single 2 gram dose administered intravenously as soon as the umbilical cord is clamped OR a 3-dose regimen consisting of 2 grams given intravenously as soon as the umbilical cord is clamped followed by 2 grams 4 and 8 hours after the initial dose is recommended (see CLINICAL STUDIES).
Table 1. Guidelines for Dosage of CEFOXITIN FOR INJECTION * Including patients in whom bacteremia is absent or unlikely.
Type of Infection Daily Dosage Frequency and Route Uncomplicated forms* of infections such as pneumonia, urinary tract infection, cutaneous infection 3 to 4 grams 1 gram every 6 to 8 hours I.V. Moderately severe or severe infections 6 to 8 grams 1 gram every 4 hours or 2 grams every 6 to 8 hours I.V. Infections commonly needing antibiotics in higher dosage (e.g., gas gangrene) 12 grams 2 grams every 4 hours or 3 grams every 6 hours I.V. Table 2. Maintenance Dosage of CEFOXITIN FOR INJECTION in Adults with Reduced Renal Function Renal Function Creatinine
Clearance
(mL/min)Dose (grams) Frequency Mild impairment 50 to 30 1 to 2 every 8 to 12 hours Moderate impairment 29 to 10 1 to 2 every 12 to 24 hours Severe impairment 9 to 5 0.5 to 1 every 12 to 24 hours Essentially no function < 5 0.5 to 1 every 24 to 48 hours Table 3. Preparation of Solution for Intravenous Administration ** Shake to dissolve and let stand until clear.
Strength Amount of
Diluent to be
Added (mL)**Approximate
Withdrawable
Volume (mL)Approximate Average
Concentration
(mg/mL)1 gram Vial 10 10.5 95 2 gram Vial 10 or 20 11.1 or 21 180 or 95 Preparation of Solution:
Table 3 is provided for convenience in constituting CEFOXITIN FOR INJECTION for intravenous administration.
For Vials: One gram should be constituted with at least 10 mL, and 2 grams with 10 mL or 20 mL, of Sterile Water for Injection, Bacteriostatic Water for Injection, 0.9 percent Sodium Chloride Injection, or 5 percent Dextrose Injection. These primary solutions may be further diluted in 50 mL to 1000 mL of the diluents listed under the Vials portion of the Compatibility and Stability section.
Benzyl alcohol as a preservative has been associated with toxicity in neonates. While toxicity has not been demonstrated in pediatric patients greater than 3 months of age, in whom use of CEFOXITIN FOR INJECTION may be indicated, small pediatric patients in this age range may also be at risk for benzyl alcohol toxicity. Therefore, diluent containing benzyl alcohol should not be used when CEFOXITIN FOR INJECTION is constituted for administration to pediatric patients in this age range.
Administration:
CEFOXITIN FOR INJECTION may be administered intravenously after constitution.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Intravenous Administration:
The intravenous route is preferable for patients with bacteremia, bacterial septicemia, or other severe or life-threatening infections, or for patients who may be poor risks because of lowered resistance resulting from such debilitating conditions as malnutrition, trauma, surgery, diabetes, heart failure, or malignancy, particularly if shock is present or impending.
For intermittent intravenous administration:
A solution containing 1 gram or 2 grams in 10 mL of Sterile Water for Injection can be injected over a period of 3 to 5 minutes. Using an infusion system, it may also be given over a longer period of time through the tubing system by which the patient may be receiving other intravenous solutions. However, during infusion of the solution containing CEFOXITIN FOR INJECTION, it is advisable to temporarily discontinue administration of any other solutions at the same site.
For the administration of higher doses by continuous intravenous infusion, a solution of CEFOXITIN FOR INJECTION may be added to an intravenous bottle containing 5 percent Dextrose Injection, 0.9 percent Sodium Chloride Injection, or 5 percent Dextrose and 0.9 percent Sodium Chloride Injection. BUTTERFLY®†† or scalp vein-type needles are preferred for this type of infusion.
Solutions of CEFOXITIN FOR INJECTION, like those of most beta-lactam antibiotics, should not be added to aminoglycoside solutions (e.g., gentamicin sulfate, tobramycin sulfate, amikacin sulfate) because of potential interaction. However, CEFOXITIN FOR INJECTION and aminoglycosides may be administered separately to the same patient.
Compatibility and Stability:
Vials:
CEFOXITIN FOR INJECTION as supplied in vials and constituted to 1 gram/10 mL with Sterile Water for Injection, Bacteriostatic Water for Injection, (see Preparation of Solution), 0.9 percent Sodium Chloride Injection, or 5 percent Dextrose Injection maintains satisfactory potency for 6 hours at room temperature or for one week under refrigeration (below 5°C).
These primary solutions may be further diluted in 50 mL to 1000 mL of the following diluents and maintain potency for an additional 18 hours at room temperature or an additional 48 hours under refrigeration:
- 0.9 percent Sodium Chloride Injection
- 5 percent or 10 percent Dextrose Injection
- 5 percent Dextrose and 0.9 percent Sodium Chloride Injection
- 5 percent Dextrose Injection with 0.2 percent or
- 0.45 percent saline solution
- Lactated Ringer's Injection
- 5 percent Dextrose in Lactated Ringer's Injection
- 5 percent Sodium Bicarbonate Injection
- M/6 sodium lactate solution
- Mannitol 5% and 10%
After the periods mentioned above, any unused solutions should be discarded.
-
HOW SUPPLIED:
CEFOXITIN FOR INJECTION, USP is a dry white to off-white powder supplied in conventional vials containing cefoxitin sodium as follows:
Product Code Unit of Sale Strength Each vial 304125 NDC 63323-341-25
Unit of 25Sterile cefoxitin sodium, USP equivalent to 1 gram cefoxitin NDC 63323-341-01 304225 NDC 63323-342-25
Unit of 25Sterile cefoxitin sodium, USP equivalent to 2 grams cefoxitin NDC 63323-342-01 The container closure is not made with natural rubber latex.
Special storage instructions:
CEFOXITIN FOR INJECTION, USP in the dry state should be stored between 2° to 25°C (36° to 77°F). Avoid exposure to temperatures above 50°C. The dry material as well as solutions tend to darken, depending on storage conditions; product potency, however, is not adversely affected.
-
CLINICAL STUDIES:
A prospective, randomized, double-blind, placebo-controlled clinical trial was conducted to determine the efficacy of short-term prophylaxis with CEFOXITIN FOR INJECTION in patients undergoing cesarean section who were at high risk for subsequent endometritis because of ruptured membranes. Patients were randomized to receive either three doses of placebo (n=58), a single dose of CEFOXITIN FOR INJECTION (2 g) followed by two doses of placebo (n=64), or a three-dose regimen of CEFOXITIN FOR INJECTION (each dose consisting of 2 g) (n=60), given intravenously, usually beginning at the time of clamping of the umbilical cord, with the second and third doses given 4 and 8 hours post-operatively. Endometritis occurred in 16/58 (27.6%) patients given placebo, 5/63 (7.9%) patients given a single dose of CEFOXITIN FOR INJECTION and 3/58 (5.2%) patients given three doses of CEFOXITIN FOR INJECTION. The differences between the two groups treated with CEFOXITIN FOR INJECTION and placebo with respect to endometritis were statistically significant (p<0.01) in favor of CEFOXITIN FOR INJECTION. The differences between the one-dose and three-dose regimens of CEFOXITIN FOR INJECTION were not statistically significant.
Two double-blind, randomized studies compared the efficacy of a single 2 gram intravenous dose of CEFOXITIN FOR INJECTION to a single 2 gram intravenous dose of cefotetan in the prevention of surgical site-related infection (major morbidity) and non-site-related infections (minor morbidity) in patients following cesarean section. In the first study, 82/98 (83.7%) patients treated with CEFOXITIN FOR INJECTION and 71/95 (74.7%) patients treated with cefotetan experienced no major or minor morbidity. The difference in the outcomes in this study (95% CI: –0.03, +0.21) was not statistically significant. In the second study, 65/75 (86.7%) patients treated with CEFOXITIN FOR INJECTION and 62/76 (81.6%) patients treated with cefotetan experienced no major or minor morbidity. The difference in the outcomes in this study (95% CI: –0.08, +0.18) was not statistically significant.
In clinical trials of patients with intra-abdominal infections due to Bacteroides fragilis group microorganisms, eradication rates at 1 to 2 weeks posttreatment for isolates were in the range of 70% to 80%.
Eradication rates for individual species are listed below:
NOTES:
† Clinitest is a trademark of Siemens Healthcare Diagnostics Inc.
†† Registered trademark of Abbott Laboratories, Inc.
Bacteroides distasonis 7/10 (70%) Bacteroides fragilis 26/33 (79%) Bacteroides ovatus 10/13 (77%) B. thetaiotaomicron 13/18 (72%) Manufactured for:
Lake Zurich, IL 60047
Made in Italy
www.fresenius-kabi.com/us
451232E
Revised: May 2021
- PRINCIPAL DISPLAY PANEL
-
PRINCIPAL DISPLAY PANEL
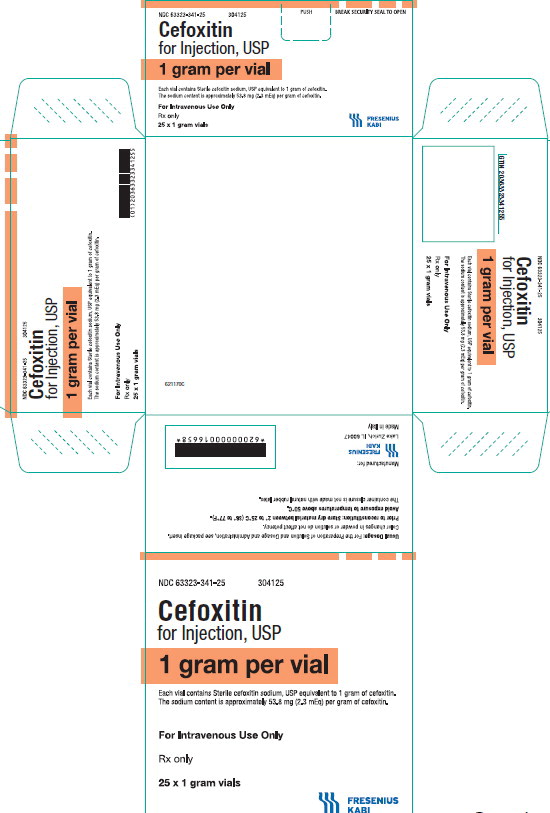
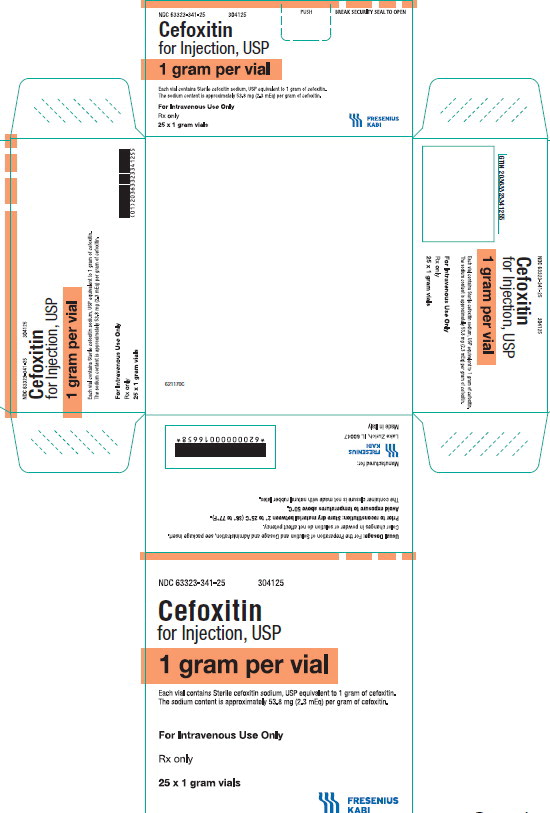
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - Cefoxitin 1 gram Vial Carton Panel
NDC 63323-341-25
304125Cefoxitin
for Injection, USP
1 gram per vial
Each vial contains Sterile cefoxitin sodium, USP equivalent to 1 gram of cefoxitin. The sodium content is approximately 53.8 mg (2.3 mEq) per gram of cefoxitin.
For Intravenous Use Only
Rx only
25 x 1 gram vials
- PRINCIPAL DISPLAY PANEL
-
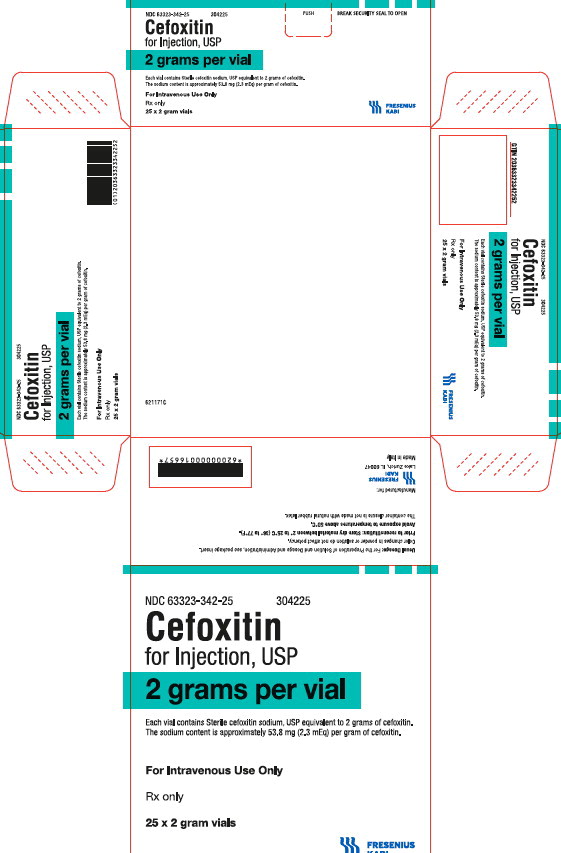
PRINCIPAL DISPLAY PANEL
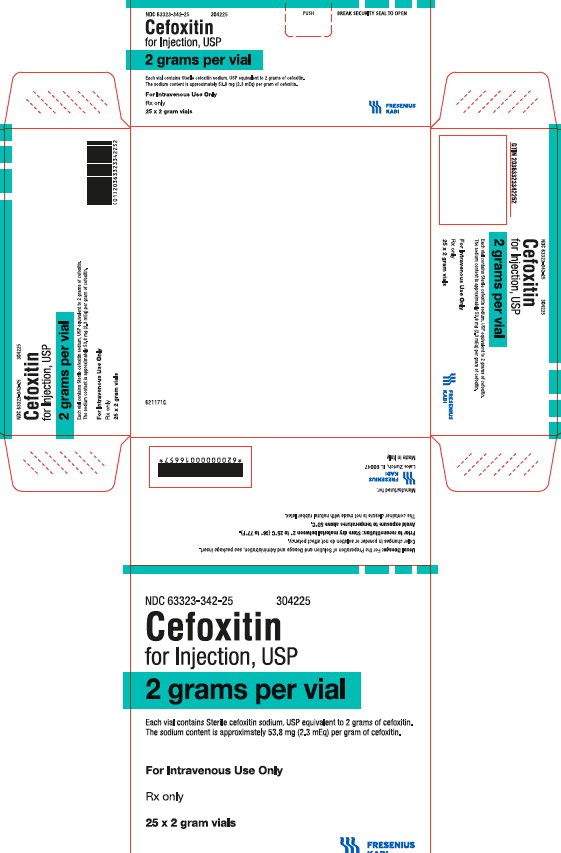
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL - Cefoxitin 2 gram Vial Carton Panel
NDC 63323-342-25
304225Cefoxitin
for Injection, USP
2 grams per vial
Each vial contains Sterile cefoxitin sodium, USP equivalent to 2 grams of cefoxitin. The sodium content is approximately 53.8 mg (2.3 mEq) per gram of cefoxitin.
For Intravenous Use Only
Rx only
25 x 2 gram vials
-
INGREDIENTS AND APPEARANCE
CEFOXITIN
cefoxitin sodium powder, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:63323-341 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CEFOXITIN SODIUM (UNII: Q68050H03T) (CEFOXITIN - UNII:6OEV9DX57Y) CEFOXITIN 1 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63323-341-25 25 in 1 CARTON 07/06/2011 1 NDC:63323-341-01 1 in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA065414 07/06/2011 CEFOXITIN
cefoxitin sodium powder, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:63323-342 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CEFOXITIN SODIUM (UNII: Q68050H03T) (CEFOXITIN - UNII:6OEV9DX57Y) CEFOXITIN 2 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63323-342-25 25 in 1 CARTON 07/06/2011 1 NDC:63323-342-01 1 in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA065414 07/06/2011 Labeler - Fresenius Kabi USA, LLC (608775388) Establishment Name Address ID/FEI Business Operations ACS Dobfar SpA 440886261 ANALYSIS(63323-341, 63323-342) , MANUFACTURE(63323-341, 63323-342) , PACK(63323-341, 63323-342)