Label: HEMOX A- aluminum chloride solution
- NDC Code(s): 68400-203-18
- Packager: Mycone Dental Supply Co., Inc DBA Keystone Industries and Deepak Products Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 20, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DESCRIPTION:
- PRECAUTIONS:
-
BENEFITS:
Hemox-A Solution stops gingival bleeding fast. If you should inadvertantly damage the gingival margin during tooth preparation or while placing a strip, band or a cerival matrix, moisten the area with Hemox-A Solution which will control and stop bleeding quickly. Hemox-A Solution contains no epinephrine. The use of vasoconstrictors for gingival retractions and bleeding is potentially dangerous and should be avoided. A plain gingival retraction cord can be dipped in Hemox-A Solution and then placed into the gingival sulcus for its hemostatic action.
-
DIRECTIONS:
Soak a cotton tipped applicator or a retraction cord with Hemox-A Solution. Apply to bleeding area with pressure until bleeding is stopped. Should a small dark coagulum appear, flush it away with water. This will have no effect on hemostatic action. Remove the impregnated cord from sulcus. Flush with water and dry with air. If hemostatis is not achieved, repeat application, rinse with water and air dry until bleeding is no longer present.
- CONTRAINDICATIONS:
- CAUTIONS:
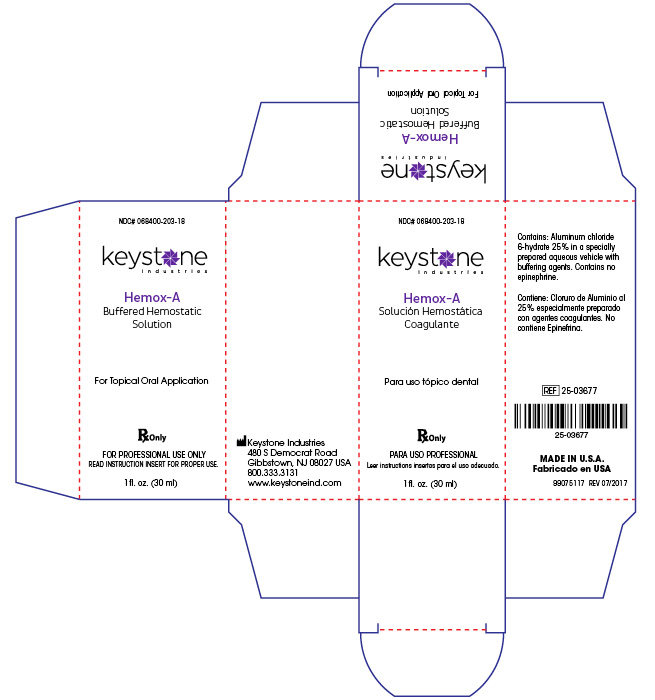
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
HEMOX A
aluminum chloride solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68400-203 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM CHLORIDE (UNII: 3CYT62D3GA) (ALUMINUM CATION - UNII:3XHB1D032B) ALUMINUM CHLORIDE 7500 mg in 30 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) POTASSIUM CHLORIDE (UNII: 660YQ98I10) DMDM HYDANTOIN (UNII: BYR0546TOW) Product Characteristics Color white (colorless) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68400-203-18 30 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 10/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 01/15/2013 Labeler - Mycone Dental Supply Co., Inc DBA Keystone Industries and Deepak Products Inc. (014769301) Registrant - Mycone Dental Supply Co., Inc DBA Keystone Industries and Deepak Products Inc. (014769301) Establishment Name Address ID/FEI Business Operations Mycone Dental Supply Co., Inc DBA Keystone Industries and Deepak Products Inc. 014769301 manufacture(68400-203) , label(68400-203)