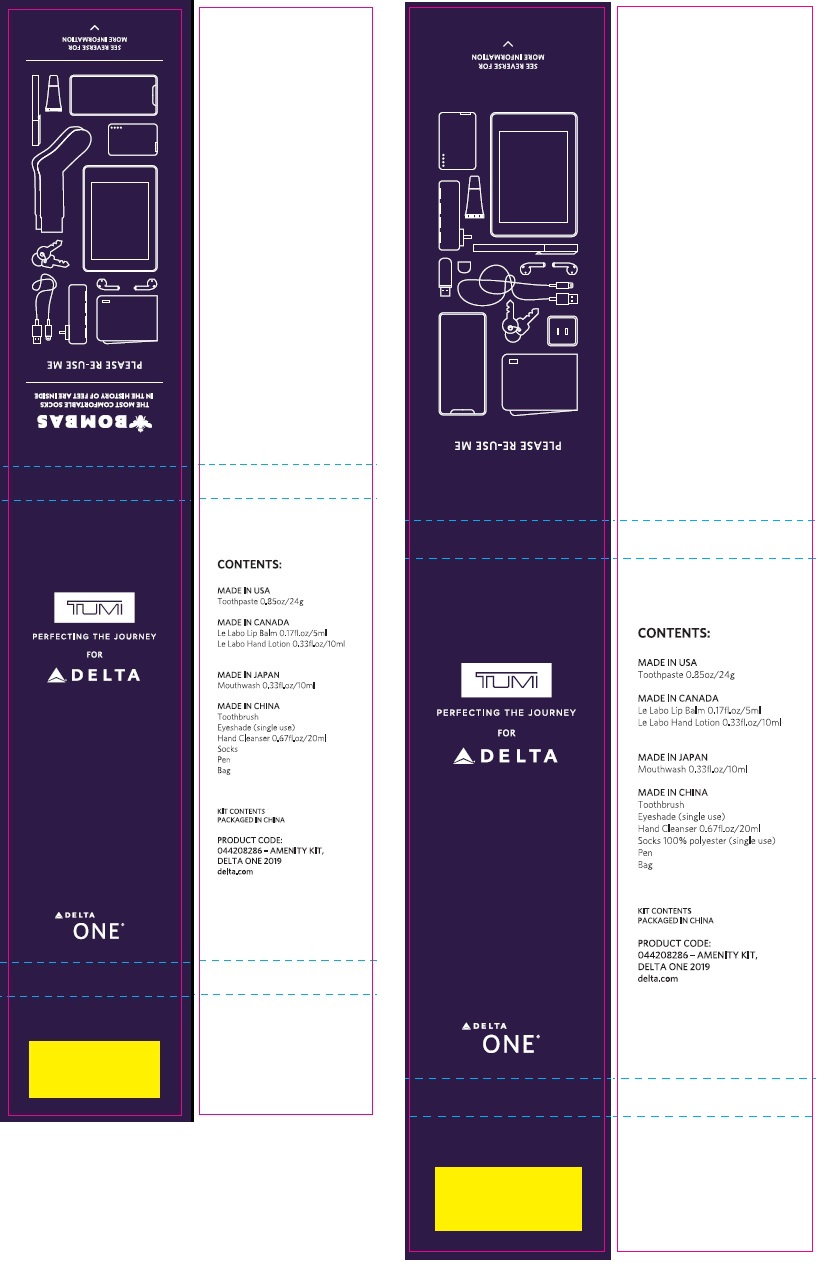
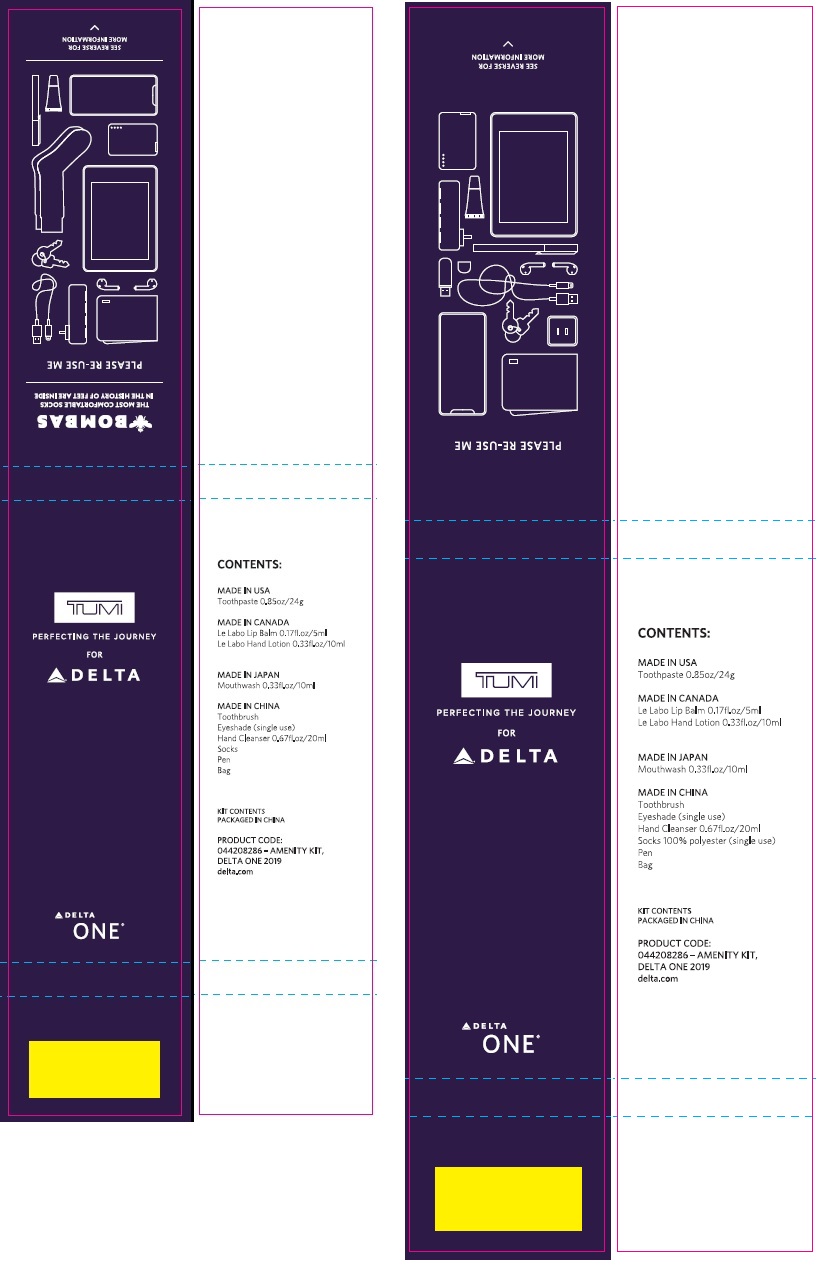
Label: DELTA ONE 2019 TUMI COLLINS AND TUMI COLLINS BOMBAS AMENITY WITH CREST 3D WHITE BRILLIANCE- sodium fluoride kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 37000-873-01, 69491-200-00 - Packager: Buzz Export Services Pty., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 27, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Use
- Warning
-
Directions
- adults and children 2 yrs. & older: brush teeth thoroughly after meals or at least twice a day or use as directed by a dentist
- do not swallow
- to minimize swallowing use a pea-sized amount in children under 6
- supervise children's brushing until good habits are established
- children under 2 yrs.: ask a dentist
- Inactive ingredients
- Questions?
- DELTA ONE 2019 - TUMI COLLINS & TUMI COLLINS BOMBAS - AMENITY KIT With Crest 3D White Brilliance (69491-200-00)
- CREST 3D WHITE BRILLIANCE 24g (37000-873-01)
-
INGREDIENTS AND APPEARANCE
DELTA ONE 2019 TUMI COLLINS AND TUMI COLLINS BOMBAS AMENITY WITH CREST 3D WHITE BRILLIANCE
sodium fluoride kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69491-200 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69491-200-00 1 in 1 KIT 04/01/2019 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 24 g Part 1 of 1 CREST 3D WHITE BRILLIANCE
sodium fluoride paste, dentifriceProduct Information Item Code (Source) NDC:37000-873 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 1.6 mg in 1 g Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) HYDRATED SILICA (UNII: Y6O7T4G8P9) HEXASODIUM HEXAMETAPHOSPHATE (UNII: N40N91DW96) WATER (UNII: 059QF0KO0R) POLYETHYLENE GLYCOL 300 (UNII: 5655G9Y8AQ) METHYL SALICYLATE (UNII: LAV5U5022Y) SODIUM LAURYL SULFATE (UNII: 368GB5141J) CARRAGEENAN (UNII: 5C69YCD2YJ) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) SACCHARIN SODIUM (UNII: SB8ZUX40TY) POLYETHYLENE OXIDE 900000 (UNII: 16P9295IIL) XANTHAN GUM (UNII: TTV12P4NEE) SUCRALOSE (UNII: 96K6UQ3ZD4) MICA (UNII: V8A1AW0880) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white Score Shape Size Flavor PEPPERMINT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:37000-873-01 24 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part355 04/01/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part355 04/01/2019 Labeler - Buzz Export Services Pty., Ltd. (747317902)