Label: LENALIDOMIDE capsule
-
NDC Code(s):
70710-1030-1,
70710-1030-7,
70710-1031-1,
70710-1031-7, view more70710-1032-1, 70710-1032-7, 70710-1033-1, 70710-1033-8, 70710-1034-1, 70710-1034-8, 70710-1035-1, 70710-1035-8
- Packager: Zydus Pharmaceuticals USA Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 10, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LENALIDOMIDE CAPSULES safely and effectively. See full prescribing information for LENALIDOMIDE CAPSULES.
LENALIDOMIDE capsules, for oral use
Initial U.S. Approval:2005WARNING: EMBRYO-FETAL TOXICITY, HEMATOLOGIC TOXICITY, and VENOUS and ARTERIAL THROMBOEMBOLISM
See full prescribing information for complete boxed warning.
EMBRYO-FETAL TOXICITY
- Lenalidomide, a thalidomide analogue, caused limb abnormalities in a developmental monkey study similar to birth defects caused by thalidomide in humans. If lenalidomide is used during pregnancy, it may cause birth defects or embryo-fetal death.
- Pregnancy must be excluded before start of treatment. Prevent pregnancy during treatment by the use of two reliable methods of contraception (5.1).
Lenalidomide is available only through a restricted distribution program, called the Lenalidomide REMS Program (5.2, 17).
HEMATOLOGIC TOXICITY. Lenalidomide can cause significant neutropenia and thrombocytopenia (5.3).
VENOUS AND ARTERIAL THROMBOEMBOLISM
- Significantly increased risk of deep vein thrombosis (DVT) and pulmonary embolism (PE), as well as risk of myocardial infarction and stroke in patients with multiple myeloma receiving lenalidomide with dexamethasone. Anti-thrombotic prophylaxis is recommended (5.4).
INDICATIONS AND USAGE
Lenalidomide is a thalidomide analogue indicated for the treatment of adult patients with:
- Multiple myeloma (MM), in combination with dexamethasone (1.1).
- Transfusion-dependent anemia due to low- or intermediate-1-risk myelodysplastic syndromes (MDS) associated with a deletion 5q abnormality with or without additional cytogenetic abnormalities (1.2).
Limitations of Use:
- Lenalidomide is not indicated and is not recommended for the treatment of patients with chronic lymphocytic leukemia (CLL) outside of controlled clinical trials (1.4).
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Capsules: 2.5 mg, 5 mg, 10 mg, 15 mg, 20 mg, and 25 mg (3).
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Increased Mortality: serious and fatal cardiac adverse reactions occurred in patients with CLL treated with lenalidomide (5.5).
- Second Primary Malignancies (SPM): Higher incidences of SPM were observed in controlled trials of patients with MM receiving lenalidomide (5.6).
- Increased Mortality: Observed in patients with MM when pembrolizumab was added to dexamethasone and a thalidomide analogue (5.7).
- Hepatotoxicity: Hepatic failure including fatalities; monitor liver function. Stop lenalidomide and evaluate if hepatotoxicity is suspected (5.8).
- Severe Cutaneous Reactions: Discontinue lenalidomide for severe reactions (5.9).
- Tumor lysis syndrome (TLS) including fatalities: Monitor patients at risk of TLS (i.e., those with high tumor burden) and take appropriate precautions (5.10).
- Tumor flare reaction: Serious tumor flare reactions, including fatal reactions, have occurred during investigational use of lenalidomide for chronic lymphocytic leukemia and lymphoma (5.11).
- Impaired Stem Cell mobilization: A decrease in the number of CD34+ cells collected after treatment (> 4 cycles) with lenalidomide has been reported. Consider early referral to transplant center (5.12).
- Hypersensitivity: Monitor patients for potential hypersensitivity. Discontinue lenalidomide for angioedema and anaphylaxis (5.15).
ADVERSE REACTIONS
- MM: Most common adverse reactions (≥20%) include diarrhea, fatigue, anemia, constipation, neutropenia, leukopenia, peripheral edema, insomnia, muscle cramp/spasms, abdominal pain, back pain, nausea, asthenia, pyrexia, upper respiratory tract infection, bronchitis, nasopharyngitis, gastroenteritis, cough, rash, dyspnea, dizziness, decreased appetite, thrombocytopenia, and tremor (6.1).
- MDS: Most common adverse reactions (>15%) include thrombocytopenia, neutropenia, diarrhea, pruritus, rash, fatigue, constipation, nausea, nasopharyngitis, arthralgia, pyrexia, back pain, peripheral edema, cough, dizziness, headache, muscle cramp, dyspnea,pharyngitis, and epistaxis (6.1).
To report SUSPECTED ADVERSE REACTIONS contact Zydus Pharmaceuticals (USA) Inc. at 1-877-993-8779 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 2/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: EMBRYO-FETAL TOXICITY, HEMATOLOGIC TOXICITY, and VENOUS and ARTERIAL THROMBOEMBOLISM
1 INDICATIONS AND USAGE
1.1 Multiple Myeloma
1.2 Myelodysplastic Syndromes
1.6 Limitations of Use
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage for Multiple Myeloma
2.2 Recommended Dosage for Myelodysplastic Syndromes
2.5 Dosage Modifications for Non-Hematologic Adverse Reactions
2.6 Recommended Dosage for Patients with Renal Impairment
2.7 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
4.1 Pregnancy
4.2 Severe Hypersensitivity Reactions
5 WARNINGS AND PRECAUTIONS
5.1 Embryo-Fetal Toxicity
5.2 Lenalidomide REMS Program
5.3 Hematologic Toxicity
5.4 Venous and Arterial Thromboembolism
5.5 Increased Mortality in Patients with CLL
5.6 Second Primary Malignancies
5.7 Increased Mortality in Patients with MM When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone
5.8 Hepatotoxicity
5.9 Severe Cutaneous Reactions
5.10 Tumor Lysis Syndrome
5.11 Tumor Flare Reaction
5.12 Impaired Stem Cell Mobilization
5.13 Thyroid Disorders
5.15 Hypersensitivity
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Digoxin
7.2 Concomitant Therapies That May Increase the Risk of Thrombosis
7.3 Warfarin
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Multiple Myeloma
14.2 Myelodysplastic Syndromes (MDS) with a Deletion 5q Cytogenetic Abnormality
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage
16.3 Handling and Disposal
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: EMBRYO-FETAL TOXICITY, HEMATOLOGIC TOXICITY, and VENOUS and ARTERIAL THROMBOEMBOLISM
Do not use lenalidomide during pregnancy. Lenalidomide, a thalidomide analogue, caused limb abnormalities in a developmental monkey study. Thalidomide is a known human teratogen that causes severe life-threatening human birth defects. If lenalidomide is used during pregnancy, it may cause birth defects or embryo-fetal death. In females of reproductive potential, obtain 2 negative pregnancy tests before starting lenalidomide treatment. Females of reproductive potential must use 2 forms of contraception or continuously abstain from heterosexual sex during and for 4 weeks after lenalidomide treatment [see Warnings and Precautions (5.1), and Medication Guide (17)]. To avoid embryo-fetal exposure to lenalidomide, lenalidomide is only available through a restricted distribution program, the Lenalidomide REMS Program(5.2).
Information about the Lenalidomide REMS Program is available at www.lenalidomiderems.com or by calling the toll-free number 1-888-423-5436.
Hematologic Toxicity (Neutropenia and Thrombocytopenia)
Lenalidomide can cause significant neutropenia and thrombocytopenia. Eighty percent of patients with del 5q myelodysplastic syndromes had to have a dose delay/reduction during the major study. Thirty-four percent of patients had to have a second dose delay/reduction.
Grade 3 or 4 hematologic toxicity was seen in 80% of patients enrolled in the study. Patients on therapy for del 5q myelodysplastic syndromes should have their complete blood counts monitored weekly for the first 8 weeks of therapy and at least monthly thereafter.
Patients may require dose interruption and/or reduction. Patients may require use of blood product support and/or growth factors [see Dosage and Administration (2.2)].
Venous and Arterial Thromboembolism
Lenalidomide has demonstrated a significantly increased risk of deep vein thrombosis (DVT) and pulmonary embolism (PE), as well as risk of myocardial infarction and stroke in patients with multiple myeloma who were treated with lenalidomide and dexamethasone therapy. Monitor for and advise patients about signs and symptoms of thromboembolism. Advise patients to seek immediate medical care if they develop symptoms such as shortness of breath, chest pain, or arm or leg swelling. Thromboprophylaxis is recommended and the choice of regimen should be based on an assessment of the patient's underlying risks [see Warnings and Precautions (5.4)].
-
1 INDICATIONS AND USAGE
1.1 Multiple Myeloma
Lenalidomide in combination with dexamethasone is indicated for the treatment of adult patients with multiple myeloma (MM).
1.2 Myelodysplastic Syndromes
Lenalidomide is indicated for the treatment of adult patients with transfusion-dependent anemia due to low- or intermediate-1-risk myelodysplastic syndromes (MDS) associated with a deletion 5q cytogenetic abnormality with or without additional cytogenetic abnormalities.
1.6 Limitations of Use
Lenalidomide is not indicated and is not recommended for the treatment of patients with CLL outside of controlled clinical trials [see Warnings and Precautions (5.5)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage for Multiple Myeloma
Lenalidomide Combination Therapy
The recommended starting dose of lenalidomide is 25 mg orally once daily on Days 1 to 21 of repeated 28-day cycles in combination with dexamethasone. Refer to Section 14.1 for specific dexamethasone dosing. For patients greater than 75 years old, the starting dose of dexamethasone may be reduced [see Clinical Studies (14.1)]. Treatment should be continued until disease progression or unacceptable toxicity.
Dose Adjustments for Hematologic Toxicities During MM Treatment
Dose modification guidelines, as summarized in Table 1 below, are recommended to manage Grade 3 or 4 neutropenia or thrombocytopenia or other Grade 3 or 4 toxicity judged to be related to lenalidomide.
Table 1 Dose Adjustments for Hematologic Toxicities for MM Platelet counts
Thrombocytopenia in MM
When Platelets
Recommended Course
Days 1-21 of repeated 28-day cycle
Fall below 30,000/mcL
Interrupt lenalidomide treatment, follow CBC weekly
Return to at least 30,000/mcL
Resume lenalidomide at next lower dose. Do not dose below 2.5 mg daily
For each subsequent drop below 30,000/mcL
Interrupt lenalidomide treatment
Return to at least 30,000/mcL
Resume lenalidomide at next lower dose. Do not dose below 2.5 mg daily
Absolute Neutrophil counts (ANC)
Neutropenia in MM
When Neutrophils
Recommended Course
Days 1-21 of repeated 28-day cycle
Fall below 1000/mcL
Interrupt lenalidomide treatment, follow CBC weekly
Return to at least 1,000/mcL and neutropenia is the only toxicity
Resume lenalidomide at 25 mg daily or initial starting dose
Return to at least 1,000/mcL and if other toxicity
Resume lenalidomide at next lower dose. Do not dose below 2.5 mg daily
For each subsequent drop below 1,000/mcL
Interrupt lenalidomide treatment
Return to at least 1,000/mcL
Resume lenalidomide at next lower dose. Do not dose below 2.5 mg daily
2.2 Recommended Dosage for Myelodysplastic Syndromes
The recommended starting dose of lenalidomide is 10 mg daily. Treatment is continued or modified based upon clinical and laboratory findings. Continue treatment until disease progression or unacceptable toxicity.
Dose Adjustments for Hematologic Toxicities During MDS Treatment
Patients who are dosed initially at 10 mg and who experience thrombocytopenia should have their dosage adjusted as follows:
Platelet counts
If thrombocytopenia develops WITHIN 4 weeks of starting treatment at 10 mg daily in MDS
If baseline is at least 100,000/mcL
When Platelets
Recommended Course
Fall below 50,000/mcL
Interrupt lenalidomide treatment
Return to at least 50,000/mcL
Resume lenalidomide at 5 mg daily
If baseline is below 100,000/mcL
When Platelets
Recommended Course
Fall to 50% of the baseline value
Interrupt lenalidomide treatment
If baseline is at least 60,000/mcL and returns to at least 50,000/mcL
Resume lenalidomide at 5 mg daily
If baseline is below 60,000/mcL and returns to at least 30,000/mcL
Resume lenalidomide at 5 mg daily
If thrombocytopenia develops AFTER 4 weeks of starting treatment at 10 mg daily in MDS
When Platelets
Recommended Course
Fall below 30,000/mcL or below 50,000/mcL
with platelet transfusions
Interrupt lenalidomide treatment
Return to at least 30,000/mcL
(without hemostatic failure)
Resume lenalidomide at 5 mg daily
Patients who experience thrombocytopenia at 5 mg daily should have their dosage adjusted as follows:
If thrombocytopenia develops during treatment at 5 mg daily in MDS
When Platelets
Recommended Course
Fall below 30,000/mcL or below 50,000/mcL
with platelet transfusions
Interrupt lenalidomide treatment
Return to at least 30,000/mcL
(without hemostatic failure)
Resume lenalidomide at 2.5 mg daily
Patients who are dosed initially at 10 mg and experience neutropenia should have their dosage adjusted as follows:
Absolute Neutrophil counts (ANC)
If neutropenia develops WITHIN 4 weeks of starting treatment at 10 mg daily in MDS
If baseline ANC is at least 1,000/mcL
When Neutrophils
Recommended Course
Fall below 750/mcL
Interrupt lenalidomide treatment
Return to at least 1,000/mcL
Resume lenalidomide at 5 mg daily
If baseline ANC is below 1,000/mcL
When Neutrophils
Recommended Course
Fall below 500/mcL
Interrupt lenalidomide treatment
Return to at least 500/mcL
Resume lenalidomide at 5 mg daily
If neutropenia develops AFTER 4 weeks of starting treatment at 10 mg daily in MDS
When Neutrophils
Recommended Course
Fall below 500/mcL for at least 7 days or below 500/mcL
associated with fever (at least 38.5°C)
Interrupt lenalidomide treatment
Return to at least 500/mcL
Resume lenalidomide at 5 mg daily
Patients who experience neutropenia at 5 mg daily should have their dosage adjusted as follows:
If neutropenia develops during treatment at 5 mg daily in MDS
When Neutrophils
Recommended Course
Fall below 500/mcL for at least 7 days or below 500/mcL associated with fever (at least 38.5°C)
Interrupt lenalidomide treatment
Return to at least 500/mcL
Resume lenalidomide at 2.5 mg daily
2.5 Dosage Modifications for Non-Hematologic Adverse Reactions
For non-hematologic Grade 3/4 toxicities judged to be related to lenalidomide, hold treatment and restart at the physician's discretion at next lower dose level when toxicity has resolved to Grade 2 or below.
Permanently discontinue lenalidomide capsules for angioedema, anaphylaxis, Grade 4 rash, skin exfoliation, bullae, or any other severe dermatologic reactions [see Warnings and Precautions (5.9, 5.15)].
2.6 Recommended Dosage for Patients with Renal Impairment
The recommendations for dosing patients with renal impairment are shown in the following table [see Clinical Pharmacology (12.3)].
Table 3 Dose Adjustments for Patients with Renal Impairment Renal Function (Cockcroft-Gault)
Dose in Lenalidomide Combination Therapy for MM
CLcr 30 to 60 mL/min
10 mg once daily
CLcr below 30 mL/min (not requiring dialysis)
15 mg every other day
CLcr below 30 mL/min (requiring dialysis)
5 mg once daily. On dialysis days, administer the dose following dialysis.
Lenalidomide Combination Therapy for MM: For CLcr of 30 to 60 mL/min, consider escalating the dose to 15 mg after 2 cycles if the patient tolerates the 10 mg dose of lenalidomide without dose-limiting toxicity.
-
3 DOSAGE FORMS AND STRENGTHS
Lenalidomide capsules, 2.5 mg, 5 mg, 10 mg, 15 mg, 20 mg and 25 mg will be supplied through the Lenalidomide REMS Program.
Lenalidomide capsules are available in the following strengths:
2.5 mg: White to off-white powder filled in size '5' hard gelatin capsule with white opaque colored cap imprinted with "1030" in black ink and blue opaque color body.
5 mg: White to off-white powder filled in size '5' hard gelatin capsule with white opaque colored cap imprinted with "1031" in black ink and white opaque color body.
10 mg: White to off-white powder filled in size '3' hard gelatin capsule with white opaque colored cap imprinted with "1032" in black ink and turquoise blue opaque color body.
15 mg: White to off-white powder filled in size '2' hard gelatin capsule with white opaque colored cap imprinted with "1033" in black ink and light blue opaque color body.
20 mg: White to off-white powder filled in size '1' hard gelatin capsule with light blue opaque colored cap imprinted with "1034" in black ink and turquoise blue opaque color body.
25 mg: White to off-white powder filled in size '0' hard gelatin capsule with white opaque colored cap imprinted with "1035" in black ink and light blue opaque color body.
-
4 CONTRAINDICATIONS
4.1 Pregnancy
Lenalidomide can cause fetal harm when administered to a pregnant female. Limb abnormalities were seen in the offspring of monkeys that were dosed with lenalidomide during organogenesis. This effect was seen at all doses tested. Due to the results of this developmental monkey study, and lenalidomide's structural similarities to thalidomide, a known human teratogen, lenalidomide is contraindicated in females who are pregnant [see Boxed Warning]. If this drug is used during pregnancy or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential risk to a fetus [see Warnings and Precautions (5.1, 5.2), Use in Special Populations (8.1, 8.3)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Embryo-Fetal Toxicity
Lenalidomide is a thalidomide analogue and is contraindicated for use during pregnancy. Thalidomide is a known human teratogen that causes life-threatening human birth defects or embryo-fetal death [see Use in Specific Populations (8.1)]. An embryo-fetal development study in monkeys indicates that lenalidomide produced malformations in the offspring of female monkeys who received the drug during pregnancy, similar to birth defects observed in humans following exposure to thalidomide during pregnancy.
Lenalidomide is only available through the Lenalidomide REMS program [see Warnings and Precautions (5.2)].
Females of Reproductive Potential
Females of reproductive potential must avoid pregnancy for at least 4 weeks before beginning lenalidomide therapy, during therapy, during dose interruptions and for at least 4 weeks after completing therapy.
Females must commit either to abstain continuously from heterosexual sexual intercourse or to use two methods of reliable birth control, beginning 4 weeks prior to initiating treatment with lenalidomide, during therapy, during dose interruptions and continuing for 4 weeks following discontinuation of lenalidomide therapy.
Two negative pregnancy tests must be obtained prior to initiating therapy. The first test should be performed within 10 to 14 days and the second test within 24 hours prior to prescribing lenalidomide therapy and then weekly during the first month, then monthly thereafter in females with regular menstrual cycles or every 2 weeks in females with irregular menstrual cycles [see Use in Specific Populations (8.3)].
Males
Lenalidomide is present in the semen of patients receiving the drug. Therefore, males must always use a latex or synthetic condom during any sexual contact with females of reproductive potential while taking lenalidomide and for up to 4 weeks after discontinuing lenalidomide, even if they have undergone a successful vasectomy. Male patients taking lenalidomide must not donate sperm and for up to 4 weeks after discontinuing lenalidomide [see Use in Specific Populations (8.3)].
5.2 Lenalidomide REMS Program
Because of the embryo-fetal risk [see Warnings and Precautions (5.1)], lenalidomide is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS), the Lenalidomide REMS Program.
Required components of the Lenalidomide REMS program include the following:
- Prescribers must be certified with the Lenalidomide REMS program by enrolling and complying with the REMS requirements.
- Patients must sign a Patient-Physician agreement form and comply with the REMS requirements. In particular, female patients of reproductive potential who are not pregnant must comply with the pregnancy testing and contraception requirements [see Use in Specific Populations (8.3)] and males must comply with contraception requirements [see Use in Specific Populations (8.3)].
- Pharmacies must be certified with the Lenalidomide REMS program, must only dispense to patients who are authorized to receive lenalidomide and comply with REMS requirements.
Further information about the Lenalidomide REMS program is available at www.lenalidomiderems.com or by telephone at 1-888-423-5436.
5.3 Hematologic Toxicity
Lenalidomide can cause significant neutropenia and thrombocytopenia. Monitor patients with neutropenia for signs of infection. Advise patients to observe for bleeding or bruising, especially with use of concomitant medication that may increase risk of bleeding. Patients taking lenalidomide should have their complete blood counts assessed periodically as described below [see Dosage and Administration (2.1)].
Monitor complete blood counts (CBC) in patients taking lenalidomide in combination with dexamethasone for MM every 7 days (weekly) for the first 2 cycles, on Days 1 and 15 of Cycle 3, and every 28 days (4 weeks) thereafter. A dose interruption and/or dose reduction may be required [see Dosage and Administration (2.1, 2.2)]. In trials for another indication, Grade 3 or 4 neutropenia was reported in up to 59% of lenalidomide-treated patients and Grade 3 or 4 thrombocytopenia in up to 38% of lenalidomide-treated patients.
Monitor complete blood counts (CBC) in patients taking lenalidomide for MDS weekly for the first 8 weeks and at least monthly thereafter. Grade 3 or 4 hematologic toxicity was seen in 80% of patients enrolled in the MDS study. In the 48% of patients who developed Grade 3 or 4 neutropenia, the median time to onset was 42 days (range, 14-411 days), and the median time to documented recovery was 17 days (range, 2-170 days). In the 54% of patients who developed Grade 3 or 4 thrombocytopenia, the median time to onset was 28 days (range, 8-290 days), and the median time to documented recovery was 22 days (range, 5-224 days) [see Boxed Warning and Dosage and Administration (2.2)].
5.4 Venous and Arterial Thromboembolism
Venous thromboembolic events (VTE [DVT and PE]) and arterial thromboembolic events (ATE, myocardial infarction and stroke) are increased in patients treated with lenalidomide.
A significantly increased risk of DVT (7.4%) and of PE (3.7%) occurred in patients with MM after at least one prior therapy who were treated with lenalidomide and dexamethasone therapy compared to patients treated in the placebo and dexamethasone group (3.1% and 0.9%) in clinical trials with varying use of anticoagulant therapies. In the newly diagnosed multiple myeloma (NDMM) study in which nearly all patients received antithrombotic prophylaxis, DVT was reported as a serious adverse reaction (3.6%, 2.0%, and 1.7%) in the Rd Continuous, Rd18, and MPT Arms, respectively. The frequency of serious adverse reactions of PE was similar between the Rd Continuous, Rd18, and MPT Arms (3.8%, 2.8%, and 3.7%, respectively) [see Boxed Warning and Adverse Reactions (6.1)].
Myocardial infarction (1.7%) and stroke (CVA) (2.3%) are increased in patients with MM after at least one prior therapy who were treated with lenalidomide and dexamethasone therapy compared to patients treated with placebo and dexamethasone (0.6%, and 0.9%) in clinical trials. In the NDMM study, myocardial infarction (including acute) was reported as a serious adverse reaction (2.3%, 0.6%, and 1.1%) in the Rd Continuous, Rd18, and MPT Arms, respectively. The frequency of serious adverse reactions of CVA was similar between the Rd Continuous, Rd18, and MPT Arms (0.8%, 0.6 %, and 0.6%, respectively) [see Adverse Reactions (6.1)].
Patients with known risk factors, including prior thrombosis, may be at greater risk and actions should be taken to try to minimize all modifiable factors (e.g. hyperlipidemia, hypertension, smoking).
In controlled clinical trials that did not use concomitant thromboprophylaxis, 21.5% overall thrombotic events (Standardized MedDRA Query Embolic and Thrombotic events) occurred in patients with refractory and relapsed MM who were treated with lenalidomide and dexamethasone compared to 8.3% thrombosis in patients treated with placebo and dexamethasone. The median time to first thrombosis event was 2.8 months. In the NDMM study in which nearly all patients received antithrombotic prophylaxis, the overall frequency of thrombotic events was 17.4% in patients in the combined Rd Continuous and Rd18 Arms, and was 11.6% in the MPT Arm. The median time to first thrombosis event was 4.3 months in the combined Rd Continuous and Rd18 Arms.
Thromboprophylaxis is recommended. The regimen of thromboprophylaxis should be based on an assessment of the patient's underlying risks. Instruct patients to report immediately any signs and symptoms suggestive of thrombotic events. ESAs and estrogens may further increase the risk of thrombosis and their use should be based on a benefit-risk decision in patients receiving lenalidomide [see Drug Interactions (7.2)].
5.5 Increased Mortality in Patients with CLL
In a prospective randomized (1:1) clinical trial in the first line treatment of patients with chronic lymphocytic leukemia, single agent lenalidomide therapy increased the risk of death as compared to single agent chlorambucil. In an interim analysis, there were 34 deaths among 210 patients on the lenalidomide treatment arm compared to 18 deaths among 211 patients in the chlorambucil treatment arm, and hazard ratio for overall survival was 1.92 [95% CI: 1.08 to 3.41], consistent with a 92% increase in the risk of death. The trial was halted for safety in July 2013.
Serious adverse cardiovascular reactions, including atrial fibrillation, myocardial infarction, and cardiac failure occurred more frequently in the lenalidomide treatment arm. Lenalidomide is not indicated and not recommended for use in CLL outside of controlled clinical trials.
5.6 Second Primary Malignancies
In clinical trials in patients with MM receiving lenalidomide, an increase of hematologic plus solid tumor second primary malignancies (SPM) notably AML and MDS have been observed. An increase in hematologic SPM including AML and MDS occurred in 5.3% of patients with NDMM receiving lenalidomide in combination with oral melphalan compared with 1.3% of patients receiving melphalan without lenalidomide. The frequency of AML and MDS cases in patients with NDMM treated with lenalidomide in combination with dexamethasone without melphalan was 0.4%.
In a study for another indication, hematologic SPM occurred in 7.5% of patients compared to 3.3% in patients receiving placebo. The incidence of hematologic plus solid tumor (excluding squamous cell carcinoma and basal cell carcinoma) SPM was 14.9%, compared to 8.8% in patients receiving placebo with a median follow-up of 91.5 months. Non-melanoma skin cancer SPM, including squamous cell carcinoma and basal cell carcinoma, occurred in 3.9% of patients receiving lenalidomide maintenance, compared to 2.6% in the placebo arm.
In patients with relapsed or refractory MM treated with lenalidomide/dexamethasone, the incidence of hematologic plus solid tumor (excluding squamous cell carcinoma and basal cell carcinoma) SPM was 2.3% versus 0.6% in the dexamethasone alone arm. Non-melanoma skin cancer SPM, including squamous cell carcinoma and basal cell carcinoma, occurred in 3.1% of patients receiving lenalidomide/dexamethasone, compared to 0.6% in the dexamethasone alone arm.
Patients who received lenalidomide-containing therapy until disease progression did not show a higher incidence of invasive SPM than patients treated in the fixed duration lenalidomide-containing arms. Monitor patients for the development of second primary malignancies. Take into account both the potential benefit of lenalidomide and the risk of second primary malignancies when considering treatment with lenalidomide.
5.7 Increased Mortality in Patients with MM When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone
In two randomized clinical trials in patients with MM, the addition of pembrolizumab to a thalidomide analogue plus dexamethasone, a use for which no PD-1 or PD-L1 blocking antibody is indicated, resulted in increased mortality. Treatment of patients with MM with a PD-1 or PD-L1 blocking antibody in combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled clinical trials.
5.8 Hepatotoxicity
Hepatic failure, including fatal cases, has occurred in patients treated with lenalidomide in combination with dexamethasone. In clinical trials, 15% of patients experienced hepatotoxicity (with hepatocellular, cholestatic and mixed characteristics); 2% of patients with MM had serious hepatotoxicity events. The mechanism of drug-induced hepatotoxicity is unknown. Pre-existing viral liver disease, elevated baseline liver enzymes, and concomitant medications may be risk factors. Monitor liver enzymes periodically. Stop lenalidomide upon elevation of liver enzymes. After return to baseline values, treatment at a lower dose may be considered.
5.9 Severe Cutaneous Reactions
Severe cutaneous reactions including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS) have been reported. DRESS may present with a cutaneous reaction (such as rash or exfoliative dermatitis), eosinophilia, fever, and/or lymphadenopathy with systemic complications such as hepatitis, nephritis, pneumonitis, myocarditis, and/or pericarditis. These events can be fatal. Patients with a prior history of Grade 4 rash associated with thalidomide treatment should not receive lenalidomide. Consider lenalidomide interruption or discontinuation for Grade 2-3 skin rash. Permanently discontinue lenalidomide for Grade 4 rash, exfoliative or bullous rash, or for other severe cutaneous reactions such as SJS, TEN or DRESS [see Dosage and Administration (2.5)].
5.10 Tumor Lysis Syndrome
Fatal instances of tumor lysis syndrome (TLS) have been reported during treatment with lenalidomide. The patients at risk of TLS are those with high tumor burden prior to treatment. Monitor patients at risk closely and take appropriate preventive approaches.
5.11 Tumor Flare Reaction
Tumor flare reaction (TFR), including fatal reactions, have occurred during investigational use of lenalidomide for CLL and lymphoma, and is characterized by tender lymph node swelling, low grade fever, pain and rash. Lenalidomide is not indicated and not recommended for use in CLL outside of controlled clinical trials.
Tumor flare reaction may mimic progression of disease (PD).
Lenalidomide capsules may be continued in patients with Grade 1 and 2 TFR without interruption or modification, at the physician's discretion. Patients with Grade 1 and 2 TFR may also be treated with corticosteroids, non-steroidal anti-inflammatory drugs (NSAIDs) and/or narcotic analgesics for management of TFR symptoms. In patients with Grade 3 or 4 TFR, it is recommended to withhold treatment with lenalidomide capsules until TFR resolves to ≤ Grade 1. Patients with Grade 3 or 4 TFR may be treated for management of symptoms per the guidance for treatment of Grade 1 and 2 TFR.
5.12 Impaired Stem Cell Mobilization
A decrease in the number of CD34+ cells collected after treatment (> 4 cycles) with lenalidomide has been reported. In patients who received more than 4 cycles of a lenalidomide-containing treatment or for whom inadequate numbers of CD 34+ cells have been collected with G-CSF alone, G-CSF with cyclophosphamide or the combination of G-CSF with a CXCR4 inhibitor may be considered.
5.13 Thyroid Disorders
Both hypothyroidism and hyperthyroidism have been reported [see Adverse Reactions (6.2)]. Measure thyroid function before start of lenalidomide treatment and during therapy.
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described in detail in other sections of the prescribing information:
- Embryo-Fetal Toxicity [see Boxed Warning, Warnings and Precautions (5.1, 5.2)]
- Hematologic Toxicity [see Boxed Warning, Warnings and Precautions (5.3)]
- Venous and Arterial Thromboembolism [see Boxed Warning, Warnings and Precautions (5.4)]
- Increased Mortality in Patients with CLL [see Warnings and Precautions (5.5)]
- Second Primary Malignancies [see Warnings and Precautions (5.6)]
- Increased Mortality in Patients with MM When Pembrolizumab Is Added to a Thalidomide Analogue and Dexamethasone [see Warnings and Precautions (5.7)]
- Hepatotoxicity [see Warnings and Precautions (5.8)]
- Severe Cutaneous Reactions [see Warnings and Precautions (5.9)]
- Tumor Lysis Syndrome [see Warnings and Precautions (5.10)]
- Tumor Flare Reactions [see Warnings and Precautions (5.11)]
- Impaired Stem Cell Mobilization [see Warnings and Precautions (5.12)]
- Thyroid Disorders [see Warnings and Precautions (5.13)]
- Hypersensitivity [see Warnings and Precautions (5.15)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Newly Diagnosed MM - Lenalidomide Combination Therapy:
Data were evaluated from 1613 patients in a large phase 3 study who received at least one dose of lenalidomide with low dose dexamethasone (Rd) given for 2 different durations of time (i.e., until progressive disease [Arm Rd Continuous; N=532] or for up to eighteen 28-day cycles [72 weeks, Arm Rd18; N=540] or who received melphalan, prednisone and thalidomide (Arm MPT; N=541) for a maximum of twelve 42-day cycles (72 weeks). The median treatment duration in the Rd Continuous arm was 80.2 weeks (range 0.7 to 246.7) or 18.4 months (range 0.16 to 56.7).
In general, the most frequently reported adverse reactions were comparable in Arm Rd Continuous and Arm Rd18, and included diarrhea, anemia, constipation, peripheral edema, neutropenia, fatigue, back pain, nausea, asthenia, and insomnia. The most frequently reported Grade 3 or 4 reactions included neutropenia, anemia, thrombocytopenia, pneumonia, asthenia, fatigue, back pain, hypokalemia, rash, cataract, lymphopenia, dyspnea, DVT, hyperglycemia, and leukopenia. The highest frequency of infections occurred in Arm Rd Continuous (75%) compared to Arm MPT (56%). There were more grade 3 and 4 and serious adverse reactions of infection in Arm Rd Continuous than either Arm MPT or Rd18.
In the Rd Continuous arm, the most common adverse reactions leading to dose interruption of lenalidomide were infection events (28.8%); overall, the median time to the first dose interruption of lenalidomide was 7 weeks. The most common adverse reactions leading to dose reduction of lenalidomide in the Rd Continuous arm were hematologic events (10.7%); overall, the median time to the first dose reduction of lenalidomide was 16 weeks. In the Rd Continuous arm, the most common adverse reactions leading to discontinuation of lenalidomide were infection events (3.4%).
In both Rd arms, the frequencies of onset of adverse reactions were generally highest in the first 6 months of treatment and then the frequencies decreased over time or remained stable throughout treatment, except for cataracts. The frequency of onset of cataracts increased over time with 0.7% during the first 6 months and up to 9.6% by the 2nd year of treatment with Rd Continuous.
Table 4 summarizes the adverse reactions reported for the Rd Continuous, Rd18, and MPT treatment arms.
Table 4 All Adverse Reactions in ≥5% and Grade 3/4 Adverse Reactions in ≥1% of Patients with MM in the Rd Continuous or Rd18 Arms* Note: A subject with multiple occurrences of an adverse reaction is counted only once under the applicable Body System/Adverse Reaction.
a All treatment-emergent adverse events in at least 5% of subjects in the Rd Continuous or Rd18 Arms and at least a 2% higher frequency (%) in either the Rd Continuous or Rd18 Arms compared to the MPT Arm.
b All grade 3 or 4 treatment-emergent adverse events in at least 1% of subjects in the Rd Continuous or Rd18 Arms and at least a 1% higher frequency (%) in either the Rd Continuous or Rd18 Arms compared to the MPT Arm.
c Serious treatment-emergent adverse events in at least 1% of subjects in the Rd Continuous or Rd18 Arms and at least a 1% higher frequency (%) in either the Rd Continuous or Rd18 Arms compared to the MPT Arm.
d Preferred terms for the blood and lymphatic system disorders body system were included by medical judgment as known adverse reactions for Rd Continuous/Rd18, and have also been reported as serious.
e Footnote "a" not applicable.
f Footnote "b" not applicable.
@ -adverse reactions in which at least one resulted in a fatal outcome.
% -adverse reactions in which at least one was considered to be life threatening (if the outcome of the reaction was death, it is included with death cases).
*Adverse reactions included in combined adverse reaction terms:
Abdominal Pain: Abdominal pain, abdominal pain upper, abdominal pain lower, gastrointestinal pain
Pneumonias: Pneumonia, lobar pneumonia, pneumonia pneumococcal, bronchopneumonia, pneumocystis jiroveci pneumonia, pneumonia legionella, pneumonia staphylococcal, pneumonia klebsiella, atypical pneumonia, pneumonia bacterial, pneumonia escherichia, pneumonia streptococcal, pneumonia viral
Sepsis: Sepsis, septic shock, urosepsis, escherichia sepsis, neutropenic sepsis, pneumococcal sepsis, staphylococcal sepsis, bacterial sepsis, meningococcal sepsis, enterococcal sepsis, klebsiella sepsis, pseudomonal sepsis
Rash: Rash, rash pruritic, rash erythematous, rash maculo-papular, rash generalized, rash papular, exfoliative rash, rash follicular, rash macular, drug rash with eosinophilia and systemic symptoms, erythema multiforme, rash pustular
Deep Vein Thrombosis: Deep vein thrombosis, venous thrombosis limb, venous thrombosis
Body System
Adverse Reaction
All Adverse Reactionsa
Grade 3/4 Adverse Reactionsb
Rd Continuous
(N = 532)
Rd18
(N = 540)
MPT
(N = 541)
Rd Continuous
(N = 532)
Rd18
(N = 540)
MPT
(N = 541)
General disorders and administration site conditions
Fatigue%
173 (33)
177 (33)
154 (28)
39 (7)
46 (9)
31 (6)
Asthenia
150 (28)
123 (23)
124 (23)
41 (8)
33 (6)
32 (6)
Pyrexiac
114 (21)
102 (19)
76 (14)
13 (2)
7 (1)
7 (1)
Non-cardiac chest painf
29 (5)
31 (6)
18 (3)
<1%
<1%
<1%
Gastrointestinal disorders
Diarrhea
242 (45)
208 (39)
89 (16)
21 (4)
18 (3)
8 (1)
Abdominal pain%f
109 (20)
78 (14)
60 (11)
7 (1)
9 (2)
<1%
Dyspepsiaf
57 (11)
28 (5)
36 (7)
<1%
<1%
0 (0)
Musculoskeletal and connective tissue disorders
Back painc
170 (32)
145 (27)
116 (21)
37 (7)
34 (6)
28 (5)
Muscle spasmsf
109 (20)
102 (19)
61 (11)
<1%
<1%
<1%
Arthralgiaf
101 (19)
71 (13)
66 (12)
9 (2)
8 (1)
8 (1)
Bone painf
87 (16)
77 (14)
62 (11)
16 (3)
15 (3)
14 (3)
Pain in extremityf
79 (15)
66 (12)
61 (11)
8 (2)
8 (1)
7 (1)
Musculoskeletal painf
67 (13)
59 (11)
36 (7)
<1%
<1%
<1%
Musculoskeletal chest painf
60 (11)
51 (9)
39 (7)
6 (1)
<1%
<1%
Muscular weaknessf
43 (8)
35 (6)
29 (5)
<1%
8 (1)
<1%
Neck painf
40 (8)
19 (4)
10 (2)
<1%
<1%
<1%
Infections and infestations
Bronchitisc
90 (17)
59 (11)
43 (8)
9 (2)
6(1)
< 1%
Nasopharyngitisf
80 (15)
54 (10)
33 (6)
0 (0)
0(0)
0(0)
Urinary tract infectionf
76 (14)
63 (12)
41 (8)
8 (2)
8(1)
<1%
Upper respiratory tract infectionc% f
69 (13)
53 (10)
31 (6)
<1%
8 (1)
<1%
Pneumoniac@
93 (17)
87 (16)
56 (10)
60 (11)
57 (11)
41 (8)
Respiratory tract infection%
35 (7)
25 (5)
21 (4)
7 (1)
<1%
<1%
Influenzaf
33 (6)
23 (4)
15 (3)
<1%
<1%
0 (0)
Gastroenteritisf
32 (6)
17 (3)
13 (2)
0 (0)
<1%
<1%
Lower respiratory tract infection
29 (5)
14 (3)
16 (3)
10 (2)
<1%
<1%
Rhinitisf
29 (5)
24 (4)
14 (3)
0 (0)
0 (0)
0 (0)
Cellulitisc
<5%
<5%
<5%
8 (2)
<1%
<1%
Sepsisc@
33 (6)
26 (5)
18 (3)
26 (5)
20 (4)
13 (2)
Nervous system disorders
Headachef
75 (14)
52 (10)
56 (10)
<1%
<1%
<1%
Dysgeusiaf
39 (7)
45 (8)
22 (4)
<1%
0 (0)
<1%
Blood and lymphatic system disordersd
Anemia
233 (44)
193 (36)
229 (42)
97 (18)
85 (16)
102 (19)
Neutropenia
186 (35)
178 (33)
328 (61)
148 (28)
143 (26)
243 (45)
Thrombocytopenia
104 (20)
100 (19)
135 (25)
44 (8)
43 (8)
60 (11)
Febrile neutropenia
7 (1)
17 (3)
15 (3)
6 (1)
16 (3)
14 (3)
Pancytopenia
<1%
6 (1)
7 (1)
<1%
<1%
<1%
Respiratory, thoracic and mediastinal disorders
Cough f
121 (23)
94 (17)
68 (13)
<1%
<1%
<1%
Dyspneac,e
117 (22)
89 (16)
113 (21)
30 (6)
22 (4)
18 (3)
Epistaxisf
32 (6)
31 (6)
17 (3)
<1%
<1%
0 (0)
Oropharyngeal painf
30 (6)
22 (4)
14 (3)
0 (0)
0 (0)
0 (0)
Dyspnea exertional e
27 (5)
29 (5)
<5%
6 (1)
<1%
0 (0)
Metabolism and nutrition disorders
Decreased appetite
123 (23)
115 (21)
72 (13)
14 (3)
7 (1)
<1%
Hypokalemia%
91 (17)
62 (11)
38 (7)
35 (7)
20 (4)
11 (2)
Hyperglycemia
62 (12)
52 (10)
19 (4)
28 (5)
23 (4)
9 (2)
Hypocalcemia
57 (11)
56 (10)
31 (6)
23 (4)
19 (4)
8 (1)
Dehydration%
25 (5)
29 (5)
17 (3)
8 (2)
13 (2)
9 (2)
Goute
<5%
<5%
<5%
8 (2)
0 (0)
0 (0)
Diabetes mellitus%e
<5%
<5%
<5%
8 (2)
<1%
<1%
Hypophosphatemiae
<5%
<5%
<5%
7 (1)
<1%
<1%
Hyponatremia% e
<5%
<5%
<5%
7 (1)
13 (2)
6 (1)
Skin and subcutaneous tissue disorders
Rash
139 (26)
151 (28)
105 (19)
39 (7)
38 (7)
33 (6)
Pruritusf
47 (9)
49 (9)
24 (4)
<1%
<1%
<1%
Psychiatric disorders
Insomnia
147 (28)
127 (24)
53 (10)
<1%
6 (1)
0 (0)
Depression
58 (11)
46 (9)
30 (6)
10 (2)
<1%
<1%
Vascular disorders
Deep vein thrombosisc%
55 (10)
39 (7)
22 (4)
30 (6)
20 (4)
15 (3)
Hypotensionc%
51 (10)
35 (6)
36 (7)
11 (2)
8 (1)
6 (1)
Injury, Poisoning, and Procedural Complications
Fallf
43 (8)
25 (5)
25 (5)
<1%
6 (1)
6 (1)
Contusionf
33 (6)
24 (4)
15 (3)
<1%
<1%
0 (0)
Eye disorders
Cataract
73 (14)
31 (6)
<1%
31 (6)
14 (3)
<1%
Cataract subcapsulare
<5%
<5%
<5%
7 (1)
0 (0)
0 (0)
Investigations
Weight decreased
72 (14)
78 (14)
48 (9)
11 (2)
<1%
<1%
Cardiac disorders
Atrial fibrillationc
37 (7)
25 (5)
25 (5)
13 (2)
9 (2)
6 (1)
Myocardial infarction (including acute)c ,e
<5%
<5%
<5%
10 (2)
<1%
<1%
Renal and Urinary disorders
Renal failure (including acute)c@,f
49 (9)
54 (10)
37 (7)
28 (5)
33 (6)
29 (5)
Neoplasms benign, malignant and unspecified (Including cysts and polyps)
Squamous cell carcinomace
<5%
<5%
<5%
8 (2)
<1%
0 (0)
Basal cell carcinomace,f
<5%
<5%
<5%
<1%
<1%
0 (0)
After At Least One Prior Therapy for MM:
Data were evaluated from 703 patients in two studies who received at least one dose of lenalidomide/dexamethasone (353 patients) or placebo/dexamethasone (350 patients).
In the lenalidomide/dexamethasone treatment group, 269 patients (76%) had at least one dose interruption with or without a dose reduction of lenalidomide compared to 199 patients (57%) in the placebo/dexamethasone treatment group. Of these patients who had one dose interruption with or without a dose reduction, 50% in the lenalidomide/dexamethasone treatment group had at least one additional dose interruption with or without a dose reduction compared to 21% in the placebo/dexamethasone treatment group. Most adverse reactions and Grade 3/4 adverse reactions were more frequent in patients who received the combination of lenalidomide/dexamethasone compared to placebo/dexamethasone.
Tables 6, 7, and 8 summarize the adverse reactions reported for lenalidomide/dexamethasone and placebo/dexamethasone groups.
Table 6 Adverse Reactions Reported in ≥5% of Patients and with a ≥2% Difference in Proportion of Patients with MM between the Lenalidomide/dexamethasone and Placebo/dexamethasone Groups Body System
Adverse Reaction
Lenalidomide/Dex
(N=353)
n (%)
Placebo/Dex
(N=350)
n (%)
Blood and lymphatic system disorders
Neutropenia%
149 (42)
22 (6)
Anemia@
111 (31)
83 (24)
Thrombocytopenia@
76 (22)
37 (11)
Leukopenia
28 (8)
4 (1)
Lymphopenia
19 (5)
5 (1)
General disorders and administration site conditions
Fatigue
155 (44)
146 (42)
Pyrexia
97 (27)
82 (23)
Peripheral edema
93 (26)
74 (21)
Chest pain
29 (8)
20 (6)
Lethargy
24 (7)
8 (2)
Gastrointestinal disorders
Constipation
143 (41)
74 (21)
Diarrhea@
136 (39)
96 (27)
Nausea@
92 (26)
75 (21)
Vomiting@
43 (12)
33 (9)
Abdominal pain@
35 (10)
22 (6)
Dry mouth
25 (7)
13 (4)
Musculoskeletal and connective tissue disorders
Muscle cramp
118 (33)
74 (21)
Back pain
91 (26)
65 (19)
Bone pain
48 (14)
39 (11)
Pain in limb
42 (12)
32 (9)
Nervous system disorders
Dizziness
82 (23)
59 (17)
Tremor
75 (21)
26 (7)
Dysgeusia
54 (15)
34 (10)
Hypoesthesia
36 (10)
25 (7)
Neuropathya
23 (7)
13 (4)
Respiratory, thoracic and mediastinal disorders
Dyspnea
83 (24)
60 (17)
Nasopharyngitis
62 (18)
31 (9)
Pharyngitis
48 (14)
33 (9)
Bronchitis
40 (11)
30 (9)
Infectionsb and infestations
Upper respiratory tract infection
87 (25)
55 (16)
Pneumonia@
48 (14)
29 (8)
Urinary tract infection
30 (8)
19 (5)
Sinusitis
26 (7)
16 (5)
Skin and subcutaneous system disorders
Rashc
75 (21)
33 (9)
Sweating increased
35 (10)
25 (7)
Dry skin
33 (9)
14 (4)
Pruritus
27 (8)
18 (5)
Metabolism and nutrition disorders
Anorexia
55 (16)
34 (10)
Hypokalemia
48 (14)
21 (6)
Hypocalcemia
31 (9)
10 (3)
Appetite decreased
24 (7)
14 (4)
Dehydration
23 (7)
15 (4)
Hypomagnesemia
24 (7)
10 (3)
Investigations
Weight decreased
69 (20)
52 (15)
Eye disorders
Blurred vision
61 (17)
40 (11)
Vascular disorders
Deep vein thrombosis%
33 (9)
15 (4)
Hypertension
28 (8)
20 (6)
Hypotension
25 (7)
15 (4)
Table 7 Grade 3/4 Adverse Reactions Reported in ≥2% Patients and with a ≥1% Difference in Proportion of Patients with MM between the Lenalidomide/dexamethasone and Placebo/dexamethasone groups Body System
Adverse Reaction
Lenalidomide/Dex
(N=353)
n (%)
Placebo/Dex
(N=350)
n (%)
Blood and lymphatic system disorders
Neutropenia%
118 (33)
12 (3)
Thrombocytopenia@
43 (12)
22 (6)
Anemia@
35 (10)
20 (6)
Leukopenia
14 (4)
<1%
Lymphopenia
10 (3)
4 (1)
Febrile neutropenia%
8 (2)
0 (0)
General disorders and administration site conditions
Fatigue
23 (7)
17 (5)
Vascular disorders
Deep vein thrombosis%
29 (8)
12 (3)
Infections and infestations
Pneumonia@
30 (8)
19 (5)
Urinary tract infection
5 (1)
<1%
Metabolism and nutrition disorders
Hypokalemia
17 (5)
5 (1)
Hypocalcemia
13 (4)
6 (2)
Hypophosphatemia
9 (3)
0 (0)
Respiratory, thoracic and mediastinal disorders
Pulmonary embolism@
14 (4)
<1%
Respiratory distress@
4 (1)
0 (0)
Musculoskeletal and connective tissue disorders
Muscle weakness
20 (6)
10 (3)
Gastrointestinal disorders
Diarrhea@
11 (3)
4 (1)
Constipation
7 (2)
<1%
Nausea@
6 (2)
<1%
Cardiac disorders
Atrial fibrillation@
13 (4)
4 (1)
Tachycardia
6 (2)
<1%
Cardiac failure congestive@
5 (1)
<1%
Nervous system disorders
Syncope
10 (3)
<1%
Dizziness
7 (2)
<1%
Eye disorders
Cataract
6 (2)
<1%
Cataract unilateral
5 (1)
0 (0)
Psychiatric disorder
Depression
10 (3)
6 (2)
Table 8 Serious Adverse Reactions Reported in ≥1% Patients and with a ≥1% Difference in Proportion of Patients with MM between the Lenalidomide/dexamethasone and Placebo/dexamethasone Groups For Tables 6, 7 and 8 above:
@ adverse reactions in which at least one resulted in a fatal outcome.
% adverse reactions in which at least one was considered to be life threatening (if the outcome of the reaction was death, it is included with death cases).
Body System
Adverse Reaction
Lenalidomide/Dex
(N=353)
n (%)
Placebo/Dex
(N=350)
n (%)
Blood and lymphatic system disorders
Febrile neutropenia%
6 (2)
0 (0)
Vascular disorders
Deep vein thrombosis%
26 (7)
11 (3)
Infections and infestations
Pneumonia@
33 (9)
21 (6)
Respiratory, thoracic, and mediastinal disorders
Pulmonary embolism@
13 (4)
<1%
Cardiac disorders
Atrial fibrillation@
11 (3)
<1%
Cardiac failure congestive@
5 (1)
0 (0)
Nervous system disorders
Cerebrovascular accident@
7 (2)
<1%
Gastrointestinal disorders
Diarrhea@
6 (2)
<1%
Musculoskeletal and connective tissue disorders
Bone pain
4 (1)
0 (0)
Median duration of exposure among patients treated with lenalidomide/dexamethasone was 44 weeks while median duration of exposure among patients treated with placebo/dexamethasone was 23 weeks. This should be taken into consideration when comparing frequency of adverse reactions between two treatment groups lenalidomide/dexamethasone vs. placebo/dexamethasone.
Venous and Arterial Thromboembolism [see Boxed Warning, Warnings and Precautions (5.4)]
VTE and ATE are increased in patients treated with lenalidomide.
Deep vein thrombosis (DVT) was reported as a serious (7.4%) or severe (8.2%) adverse drug reaction at a higher rate in the lenalidomide/dexamethasone group compared to 3.1 % and 3.4% in the placebo/dexamethasone group, respectively in the 2 studies in patients with at least 1 prior therapy with discontinuations due to DVT adverse reactions reported at comparable rates between groups. In the NDMM study, DVT was reported as an adverse reaction (all grades: 10.3%, 7.2%, 4.1%), as a serious adverse reaction (3.6%, 2.0%, 1.7%), and as a Grade 3/4 adverse reaction (5.6%, 3.7%, 2.8%) in the Rd Continuous, Rd18, and MPT Arms, respectively. Discontinuations and dose reductions due to DVT adverse reactions were reported at comparable rates between the Rd Continuous and Rd18 Arms (both <1%). Interruption of lenalidomide treatment due to DVT adverse reactions was reported at comparable rates between the Rd Continuous (2.3%) and Rd18 (1.5%) arms. Pulmonary embolism (PE) was reported as a serious adverse drug reaction (3.7%) or Grade 3/4 (4.0%) at a higher rate in the lenalidomide/dexamethasone group compared to 0.9% (serious or grade 3/4) in the placebo/dexamethasone group in the 2 studies in patients with, at least 1 prior therapy, with discontinuations due to PE adverse reactions reported at comparable rates between groups. In the NDMM study, the frequency of adverse reactions of PE was similar between the Rd Continuous, Rd18, and MPT Arms for adverse reactions (all grades: 3.9%, 3.3%, and 4.3%, respectively), serious adverse reactions (3.8%, 2.8%, and 3.7%, respectively), and grade 3/4 adverse reactions (3.8%, 3.0%, and 3.7%, respectively).
Myocardial infarction was reported as a serious (1.7%) or severe (1.7%) adverse drug reaction at a higher rate in the lenalidomide/dexamethasone group compared to 0.6 % and 0.6% respectively in the placebo/dexamethasone group. Discontinuation due to MI (including acute) adverse reactions was 0.8% in lenalidomide/dexamethasone group and none in the placebo/dexamethasone group. In the NDMM study, myocardial infarction (including acute) was reported as an adverse reaction (all grades: 2.4%, 0.6%, and 1.1%), as a serious adverse reaction, (2.3%, 0.6%, and 1.1%), or as a severe adverse reaction (1.9%, 0.6%, and 0.9%) in the Rd Continuous, Rd18, and MPT Arms, respectively.
Stroke (CVA) was reported as a serious (2.3%) or severe (2.0%) adverse drug reaction in the lenalidomide/dexamethasone group compared to 0.9% and 0.9% respectively in the placebo/dexamethasone group. Discontinuation due to stroke (CVA) was 1.4% in lenalidomide/dexamethasone group and 0.3% in the placebo/dexamethasone group. In the NDMM study, CVA was reported as an adverse reaction (all grades: 0.8%, 0.6%, and 0.6%), as a serious adverse reaction (0.8%, 0.6 %, and 0.6%), or as a severe adverse reaction (0.6%, 0.6%, 0.2%) in the Rd Continuous, Rd18, and MPT arms respectively.
Other Adverse Reactions: After At Least One Prior Therapy for MM
In these 2 studies, the following adverse drug reactions (ADRs) not described above that occurred at ≥1% rate and of at least twice of the placebo percentage rate were reported:
Blood and lymphatic system disorders: pancytopenia, autoimmune hemolytic anemia
Cardiac disorders: bradycardia, myocardial infarction, angina pectoris
Endocrine disorders: hirsutism
Eye disorders: blindness, ocular hypertension
Gastrointestinal disorders: gastrointestinal hemorrhage, glossodynia
General disorders and administration site conditions: malaise
Investigations: liver function tests abnormal, alanine aminotransferase increased
Nervous system disorders: cerebral ischemia
Psychiatric disorders: mood swings, hallucination, loss of libido
Reproductive system and breast disorders: erectile dysfunction
Respiratory, thoracic and mediastinal disorders: cough, hoarseness
Skin and subcutaneous tissue disorders: exanthem, skin hyperpigmentation
Myelodysplastic Syndromes:
A total of 148 patients received at least 1 dose of 10 mg lenalidomide in the del 5q MDS clinical study. At least one adverse reaction was reported in all of the 148 patients who were treated with the 10 mg starting dose of lenalidomide. The most frequently reported
adverse reactions were related to blood and lymphatic system disorders, skin and subcutaneous tissue disorders, gastrointestinal disorders, and general disorders and administrative site conditions.
Thrombocytopenia (61.5%; 91/148) and neutropenia (58.8%; 87/148) were the most frequently reported adverse reactions. The next most common adverse reactions observed were diarrhea (48.6%; 72/148), pruritus (41.9%; 62/148), rash (35.8%; 53/148) and fatigue (31.1%; 46/148). Table 9 summarizes the adverse reactions that were reported in ≥ 5% of the lenalidomide treated patients in the del 5q MDS clinical study. Table 10 summarizes the most frequently observed Grade 3 and Grade 4 adverse reactions regardless of relationship to treatment with lenalidomide. In the single-arm studies conducted, it is often not possible to distinguish adverse reactions that are drug-related and those that reflect the patient's underlying disease.
Table 9
Summary of Adverse Reactions Reported in ≥5% of the
Lenalidomide Treated Patients in del 5q MDS Clinical Study
Body System
Adverse Reactiona
10 mg Overall
(N=148)
Patients with at least one adverse reaction
148
(100)
Blood and Lymphatic System Disorders
Thrombocytopenia
91
(61)
Neutropenia
87
(59)
Anemia
17
(11)
Leukopenia
12
(8)
Febrile Neutropenia
8
(5)
Skin and Subcutaneous Tissue Disorders
Pruritus
62
(42)
Rash
53
(36)
Dry Skin
21
(14)
Contusion
12
(8)
Night Sweats
12
(8)
Sweating Increased
10
(7)
Ecchymosis
8
(5)
Erythema
8
(5)
Gastrointestinal Disorders
Diarrhea
72
(49)
Constipation
35
(24)
Nausea
35
(24)
Abdominal Pain
18
(12)
Vomiting
15
(10)
Abdominal Pain Upper
12
(8)
Dry Mouth
10
(7)
Loose Stools
9
(6)
Respiratory, Thoracic and Mediastinal Disorders
Nasopharyngitis
34
(23)
Cough
29
(20)
Dyspnea
25
(17)
Pharyngitis
23
(16)
Epistaxis
22
(15)
Dyspnea Exertional
10
(7)
Rhinitis
10
(7)
Bronchitis
9
(6)
General Disorders and Administration Site Conditions
Fatigue
46
(31)
Pyrexia
31
(21)
Edema Peripheral
30
(20)
Asthenia
22
(15)
Edema
15
(10)
Pain
10
(7)
Rigors
9
(6)
Chest Pain
8
(5)
Musculoskeletal and Connective Tissue Disorders
Arthralgia
32
(22)
Back Pain
31
(21)
Muscle Cramp
27
(18)
Pain in Limb
16
(11)
Myalgia
13
(9)
Peripheral Swelling
12
(8)
Nervous System Disorders
Dizziness
29
(20)
Headache
29
(20)
Hypoesthesia
10
(7)
Dysgeusia
9
(6)
Peripheral Neuropathy
8
(5)
Infections and Infestations
Upper Respiratory Tract Infection
22
(15)
Pneumonia
17
(11)
Urinary Tract Infection
16
(11)
Sinusitis
12
(8)
Cellulitis
8
(5)
Metabolism and Nutrition Disorders
Hypokalemia
16
(11)
Anorexia
15
(10)
Hypomagnesemia
9
(6)
Investigations
Alanine Aminotransferase Increased
12
(8)
Psychiatric Disorders
Insomnia
15
(10)
Depression
8
(5)
Renal and Urinary Disorders
Dysuria
10
(7)
Vascular Disorders
Hypertension
9
(6)
Endocrine Disorders
Acquired Hypothyroidism
10
(7)
Cardiac Disorders
Palpitations
8
(5)
In other clinical studies of lenalidomide in MDS patients, the following serious adverse reactions (regardless of relationship to study drug treatment) not described in Table 9 or 10 were reported:
Blood and lymphatic system disorders: warm type hemolytic anemia, splenic infarction, bone marrow depression, coagulopathy, hemolysis, hemolytic anemia, refractory anemia
Cardiac disorders: cardiac failure congestive, atrial fibrillation, angina pectoris, cardiac arrest, cardiac failure, cardio-respiratory arrest, cardiomyopathy, myocardial infarction, myocardial ischemia, atrial fibrillation aggravated, bradycardia, cardiogenic shock, pulmonary edema, supraventricular arrhythmia, tachyarrhythmia, ventricular dysfunction
Ear and labyrinth disorders: vertigo
Endocrine disorders: Basedow's disease
Gastrointestinal disorders: gastrointestinal hemorrhage, colitis ischemic, intestinal perforation, rectal hemorrhage, colonic polyp, diverticulitis, dysphagia, gastritis, gastroenteritis, gastroesophageal reflux disease, obstructive inguinal hernia, irritable bowel syndrome, melena, pancreatitis due to biliary obstruction, pancreatitis, perirectal abscess, small intestinal obstruction, upper gastrointestinal haemorrhage
General disorders and administration site conditions: disease progression, fall, gait abnormal, intermittent pyrexia, nodule, rigors, sudden death
Hepatobiliary disorders: hyperbilirubinemia, cholecystitis, acute cholecystitis, hepatic failure
Immune system disorders: hypersensitivity
Infections and infestations: infection bacteremia, central line infection, clostridial infection, ear infection, Enterobacter sepsis, fungal infection, herpes viral infection NOS, influenza, kidney infection, Klebsiella sepsis, lobar pneumonia, localized infection, oral infection, Pseudomonas infection, septic shock, sinusitis acute, sinusitis, Staphylococcal infection, urosepsis
Injury, poisoning and procedural complications: femur fracture, transfusion reaction, cervical vertebral fracture, femoral neck fracture, fractured pelvis, hip fracture, overdose, post procedural hemorrhage, rib fracture, road traffic accident, spinal compression fracture
Investigations: blood creatinine increased, hemoglobin decreased, liver function tests abnormal, troponin I increased
Metabolism and nutrition disorders: dehydration, gout, hypernatremia, hypoglycemia
Musculoskeletal and connective tissue disorders: arthritis, arthritis aggravated, gouty arthritis, neck pain, chondrocalcinosis pyrophosphate
Neoplasms benign, malignant and unspecified: acute leukemia, acute myeloid leukemia, bronchoalveolar carcinoma, lung cancer metastatic, lymphoma, prostate cancer metastatic
Nervous system disorders: cerebrovascular accident, aphasia, cerebellar infarction, cerebral infarction, depressed level of consciousness, dysarthria, migraine, spinal cord compression, subarachnoid hemorrhage, transient ischemic attack
Psychiatric disorders: confusional state
Renal and urinary disorders: renal failure, hematuria, renal failure acute, azotemia, calculus ureteric, renal mass
Reproductive system and breast disorders: pelvic pain
Respiratory, thoracic and mediastinal disorders: bronchitis, chronic obstructive airways disease exacerbated, respiratory failure, dyspnea exacerbated, interstitial lung disease, lung infiltration, wheezing
Skin and subcutaneous tissue disorders: acute febrile neutrophilic dermatosis
Vascular system disorders: deep vein thrombosis, hypotension, aortic disorder, ischemia, thrombophlebitis superficial, thrombosis
6.2 Postmarketing Experience
The following adverse drug reactions have been identified from the worldwide post-marketing experience with lenalidomide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure [see Warnings and Precautions Section (5.8 to 5.11 and 5.13)]
Endocrine disorders: Hypothyroidism, hyperthyroidism
Hepatobiliary disorders: Hepatic failure (including fatality), toxic hepatitis, cytolytic hepatitis, cholestatic hepatitis, mixed cytolytic/cholestatic hepatitis, transient abnormal liver laboratory tests
Immune system disorders: Angioedema, anaphylaxis, acute graft-versus-host disease (following allogeneic hematopoietic transplant), solid organ transplant rejection
Infections and infestations: Viral reactivation (such as hepatitis B virus and herpes zoster), progressive multifocal leukoencephalopathy (PML)
Neoplasms benign, malignant and unspecified (including cysts and polyps): Tumor lysis syndrome, tumor flare reaction
Respiratory, thoracic and mediastinal disorders: Pneumonitis
Skin and subcutaneous tissue disorders: Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS)
-
7 DRUG INTERACTIONS
7.1 Digoxin
When digoxin was co-administered with multiple doses of lenalidomide (10 mg/day) the digoxin Cmax and AUCinf were increased by 14%. Periodically monitor digoxin plasma levels, in accordance with clinical judgment and based on standard clinical practice in patients receiving this medication, during administration of lenalidomide.
7.2 Concomitant Therapies That May Increase the Risk of Thrombosis
Erythropoietic agents, or other agents that may increase the risk of thrombosis, such as estrogen containing therapies, should be used with caution after making a benefit-risk assessment in patients receiving lenalidomide [see Warnings and Precautions (5.4)].
7.3 Warfarin
Co-administration of multiple doses of lenalidomide (10 mg/day) with a single dose of warfarin (25 mg) had no effect on the pharmacokinetics of lenalidomide or R- and S-warfarin. Expected changes in laboratory assessments of PT and INR were observed after warfarin administration, but these changes were not affected by concomitant lenalidomide administration. It is not known whether there is an interaction between dexamethasone and warfarin. Close monitoring of PT and INR is recommended in patients with MM taking concomitant warfarin.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
There is a pregnancy exposure registry that monitors pregnancy outcomes in females exposed to lenalidomide during pregnancy as well as female partners of male patients who are exposed to lenalidomide. This registry is also used to understand the root cause for the pregnancy. Report any suspected fetal exposure to lenalidomide to the FDA via the MedWatch program at 1-800-FDA-1088 and also to the REMS Call Center at 1‐888‐423‐5436.
Risk Summary
Based on the mechanism of action [see Clinical Pharmacology (12.1)] and findings from animal studies [see Data], lenalidomide can cause embryo-fetal harm when administered to a pregnant female and is contraindicated during pregnancy [see Boxed Warning, Contraindications (4.1), and Use in Specific Populations (5.1)].
Lenalidomide is a thalidomide analogue. Thalidomide is a human teratogen, inducing a high frequency of severe and life-threatening birth defects such as amelia (absence of limbs), phocomelia (short limbs), hypoplasticity of the bones, absence of bones, external ear abnormalities (including anotia, micropinna, small or absent external auditory canals), facial palsy, eye abnormalities (anophthalmos, microphthalmos), and congenital heart defects. Alimentary tract, urinary tract, and genital malformations have also been documented and mortality at or shortly after birth has been reported in about 40% of infants.
Lenalidomide caused thalidomide-type limb defects in monkey offspring. Lenalidomide crossed the placenta after administration to pregnant rabbits and pregnant rats [see Data]. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential risk to a fetus.
If pregnancy does occur during treatment, immediately discontinue the drug. Under these conditions, refer patient to an obstetrician/gynecologist experienced in reproductive toxicity for further evaluation and counseling. Report any suspected fetal exposure to lenalidomide to the FDA via the MedWatch program at 1-800-FDA-1088 and also to REMS Call Center at 1‐888‐423‐5436.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. The estimated background risk in the U.S. general population of major birth defects is 2% to 4% and of miscarriage is 15% to 20% of clinically recognized pregnancies.
Data
Animal data
In an embryo-fetal developmental toxicity study in monkeys, teratogenicity, including thalidomide-like limb defects, occurred in offspring when pregnant monkeys received oral lenalidomide during organogenesis. Exposure (AUC) in monkeys at the lowest dose was 0.17 times the human exposure at the maximum recommended human dose (MRHD) of 25 mg. Similar studies in pregnant rabbits and rats at 20 times and 200 times the MRHD respectively, produced embryo lethality in rabbits and no adverse reproductive effects in rats.
In a pre- and post-natal development study in rats, animals received lenalidomide from organogenesis through lactation. The study revealed a few adverse effects on the offspring of female rats treated with lenalidomide at doses up to 500 mg/kg (approximately 200 times the human dose of 25 mg based on body surface area). The male offspring exhibited slightly delayed sexual maturation and the female offspring had slightly lower body weight gains during gestation when bred to male offspring. As with thalidomide, the rat model may not adequately address the full spectrum of potential human embryo-fetal developmental effects for lenalidomide.
Following daily oral administration of lenalidomide from Gestation Day 7 through Gestation Day 20 in pregnant rabbits, fetal plasma lenalidomide concentrations were approximately 20-40% of the maternal Cmax. Following a single oral dose to pregnant rats, lenalidomide was detected in fetal plasma and tissues; concentrations of radioactivity in fetal tissues were generally lower than those in maternal tissues. These data indicated that lenalidomide crossed the placenta.
8.2 Lactation
There is no information regarding the presence of lenalidomide in human milk, the effects of lenalidomide on the breastfed child, or the effects of lenalidomide on milk production. Because many drugs are excreted in human milk and because of the potential for adverse reactions in breastfed children from lenalidomide, advise women not to breastfeed during treatment with lenalidomide.
8.3 Females and Males of Reproductive Potential
Lenalidomide can cause fetal harm when administered during pregnancy [see Use in Specific Populations (8.1)]. Verify the pregnancy status of females of reproductive potential prior to initiating lenalidomide therapy and during therapy. Advise females of reproductive potential that they must avoid pregnancy 4 weeks before therapy, while taking lenalidomide, during dose interruptions and for at least 4 weeks after completing therapy.
Females of reproductive potential must have 2 negative pregnancy tests before initiating lenalidomide. The first test should be performed within 10-14 days, and the second test within 24 hours prior to prescribing lenalidomide. Once treatment has started and during dose interruptions, pregnancy testing for females of reproductive potential should occur weekly during the first 4 weeks of use, then pregnancy testing should be repeated every 4 weeks in females with regular menstrual cycles. If menstrual cycles are irregular, the pregnancy testing should occur every 2 weeks. Pregnancy testing and counseling should be performed if a patient misses her period or if there is any abnormality in her menstrual bleeding. Lenalidomide treatment must be discontinued during this evaluation.
Contraception
Females
Females of reproductive potential must commit either to abstain continuously from heterosexual sexual intercourse or to use 2 methods of reliable birth control simultaneously: one highly effective form of contraception – tubal ligation, IUD, hormonal (birth control pills, injections, hormonal patches, vaginal rings, or implants), or partner's vasectomy, and 1 additional effective contraceptive method – male latex or synthetic condom, diaphragm, or cervical cap. Contraception must begin 4 weeks prior to initiating treatment with lenalidomide, during therapy, during dose interruptions, and continuing for 4 weeks following discontinuation of lenalidomide therapy. Reliable contraception is indicated even where there has been a history of infertility, unless due to hysterectomy. Females of reproductive potential should be referred to a qualified provider of contraceptive methods, if needed.
Males
Lenalidomide is present in the semen of males who take lenalidomide. Therefore, males must always use a latex or synthetic condom during any sexual contact with females of reproductive potential while taking lenalidomide and for up to 4 weeks after discontinuing lenalidomide, even if they have undergone a successful vasectomy. Male patients taking lenalidomide must not donate sperm and for up to 4 weeks after discontinuing lenalidomide.
8.5 Geriatric Use
MM In Combination: Overall, of the 1613 patients in the NDMM study who received study treatment, 94% (1521 /1613) were 65 years of age or older, while 35% (561/1613) were over 75 years of age. The percentage of patients over age 75 was similar between study arms (Rd Continuous: 33%; Rd18: 34%; MPT: 33%). Overall, across all treatment arms, the frequency in most of the adverse reaction categories (e.g., all adverse reactions, grade 3/4 adverse reactions, serious adverse reactions) was higher in older (> 75 years of age) than in younger (≤ 75 years of age) subjects. Grade 3 or 4 adverse reactions in the General Disorders and Administration Site Conditions body system were consistently reported at a higher frequency (with a difference of at least 5%) in older subjects than in younger subjects across all treatment arms. Grade 3 or 4 adverse reactions in the Infections and Infestations, Cardiac Disorders (including cardiac failure and congestive cardiac failure), Skin and Subcutaneous Tissue Disorders, and Renal and Urinary Disorders (including renal failure) body systems were also reported slightly, but consistently, more frequently (<5% difference), in older subjects than in younger subjects across all treatment arms. For other body systems (e.g., Blood and Lymphatic System Disorders, Infections and Infestations, Cardiac Disorders, Vascular Disorders), there was a less consistent trend for increased frequency of grade 3/4 adverse reactions in older vs younger subjects across all treatment arms Serious adverse reactions were generally reported at a higher frequency in the older subjects than in the younger subjects across all treatment arms.
MM After At Least One Prior Therapy
Of the 703 MM patients who received study treatment in Studies 1 and 2, 45% were age 65 or over while 12% of patients were age 75 and over. The percentage of patients age 65 or over was not significantly different between the lenalidomide/dexamethasone and placebo/dexamethasone groups. Of the 353 patients who received lenalidomide/dexamethasone, 46% were age 65 and over. In both studies, patients > 65 years of age were more likely than patients ≤ 65 years of age to experience DVT, pulmonary embolism, atrial fibrillation, and renal failure following use of lenalidomide. No differences in efficacy were observed between patients over 65 years of age and younger patients.
Of the 148 patients with del 5q MDS enrolled in the major study, 38% were age 65 and over, while 33% were age 75 and over. Although the overall frequency of adverse reactions (100%) was the same in patients over 65 years of age as in younger patients, the frequency of serious adverse reactions was higher in patients over 65 years of age than in younger patients (54% vs. 33%). A greater proportion of patients over 65 years of age discontinued from the clinical studies because of adverse reactions than the proportion of younger patients (27% vs.16%). No differences in efficacy were observed between patients over 65 years of age and younger patients.
Since elderly patients are more likely to have decreased renal function, care should be taken in dose selection. Monitor renal function.
8.6 Renal Impairment
Adjust the starting dose of lenalidomide based on the creatinine clearance value and for patients on dialysis [see Dosage and Administration (2.6)].
-
10 OVERDOSAGE
There is no specific experience in the management of lenalidomide overdose in patients with MM or MDS. In dose-ranging studies in healthy subjects, some were exposed to up to 200 mg (administered 100 mg BID) and in single-dose studies, some subjects were exposed to up to 400 mg. Pruritus, urticaria, rash, and elevated liver transaminases were the primary reported AEs. In clinical trials, the dose-limiting toxicity was neutropenia and thrombocytopenia.
-
11 DESCRIPTION
Lenalidomide, a thalidomide analogue, is an immunomodulatory agent with antiangiogenic and antineoplastic properties. The chemical name is 3-(4-amino-1-oxo 1,3-dihydro-2H-isoindol-2-yl) piperidine-2,6-dione and it has the following chemical structure:
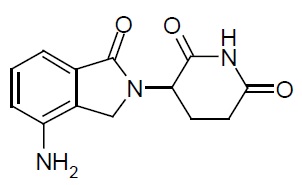
3-(4-amino-1-oxo 1,3-dihydro-2H-isoindol-2-yl) piperidine-2,6-dione
The molecular formula for lenalidomide is C13H13N3O3, and the gram molecular weight is 259.3.
Lenalidomide is white to off-white to pale-yellow solid powder. Lenalidomide has an asymmetric carbon atom and can exist as the optically active forms S(-) and R(+), and is produced as a racemic mixture with a net optical rotation of zero.
Lenalidomide capsule is available in 2.5 mg, 5 mg, 10 mg, 15 mg, 20 mg and 25 mg for oral administration. Each capsule contains lenalidomide as the active ingredient and the following inactive ingredients: croscarmellose sodium, gelatin, lactose anhydrous, lactose monohydrate, magnesium stearate, microcrystalline cellulose and titanium dioxide.
Each 2.5 mg capsule shell contains: FD& C Blue # 1, FD& C Red#40 and FD&C Yellow # 6.
Each 10 mg capsule shell contains: D&C Red # 33, D&C Yellow# 10 and FD & C Blue # 1.
Each 15 mg capsule shell contains: D&C Red # 28 and FD & C Blue # 1.
Each 20 mg capsule shell contains: D&C Red # 28, D&C Red # 33, D&C Yellow # 10 and FD & C Blue # 1.
Each 25 mg capsule shell contains: D&C Red # 28 and FD & C Blue # 1.
Each capsule is imprinted with black pharmaceutical ink which contains following inactive ingredients: black iron oxide, potassium hydroxide and shellac
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Lenalidomide is an analogue of thalidomide with immunomodulatory, antiangiogenic, and antineoplastic properties. Cellular activities of lenalidomide are mediated through its target cereblon, a component of a cullin ring E3 ubiquitin ligase enzyme complex. In vitro, in the presence of drug, substrate proteins (including Aiolos, Ikaros, and CK1α) are targeted for ubiquitination and subsequent degradation leading to direct cytotoxic and immunomodulatory effects. Lenalidomide inhibits proliferation and induces apoptosis of certain hematopoietic tumor cells including MM and del (5q) myelodysplastic syndromes in vitro. Lenalidomide causes a delay in tumor growth in some in vivo nonclinical hematopoietic tumor models including MM. Immunomodulatory properties of lenalidomide include increased number and activation of T cells and natural killer (NK) cells leading to direct and enhanced antibody-dependent cell-mediated cytotoxicity (ADCC) via increased secretion of interleukin-2 and interferon-gamma, increased numbers of NKT cells, and inhibition of pro-inflammatory cytokines (e.g., TNF-α and IL-6) by monocytes. In MM cells, the combination of lenalidomide and dexamethasone synergizes the inhibition of cell proliferation and the induction of apoptosis.
12.2 Pharmacodynamics
The effect of lenalidomide on the QTc interval was evaluated in 60 healthy male subjects in a thorough QT study. At a dose two times the maximum recommended dose, lenalidomide did not prolong the QTc interval. The largest upper bound of the two-sided 90% CI for the mean differences between lenalidomide and placebo was below 10 ms.
12.3 Pharmacokinetics
Following single and multiple doses of lenalidomide in patients with MM or MDS the maximum plasma concentrations occurred between 0.5 and 6 hours post-dose. The single and multiple dose pharmacokinetic disposition of lenalidomide is linear with AUC and Cmax values increasing proportionally with dose. Multiple doses of lenalidomide at the recommended dosage does not result in drug accumulation.
Administration of a single 25 mg dose of lenalidomide with a high-fat meal in healthy subjects reduces the extent of absorption, with an approximate 20% decrease in AUC and 50% decrease in Cmax. In the trials where the efficacy and safety were established for lenalidomide, the drug was administered without regard to food intake. Lenalidomide can be administered with or without food.
Distribution
In vitro [14C]-lenalidomide binding to plasma proteins is approximately 30%.
Lenalidomide is present in semen at 2 hours (1379 ng/ejaculate) and 24 hours (35 ng/ejaculate) after the administration of lenalidomide 25 mg daily.
Elimination
The mean half-life of lenalidomide is 3 hours in healthy subjects and 3 to 5 hours in patients with MM or MDS.
Metabolism
Lenalidomide undergoes limited metabolism. Unchanged lenalidomide is the predominant circulating component in humans. Two identified metabolites are 5hydroxy-lenalidomide and N-acetyl-lenalidomide; each constitutes less than 5% of parent levels in circulation.
Excretion
Elimination is primarily renal. Following a single oral administration of [14C]-lenalidomide 25 mg to healthy subjects, approximately 90% and 4% of the radioactive dose was eliminated within ten days in urine and feces, respectively. Approximately 82% of the radioactive dose was excreted as lenalidomide in the urine within 24 hours. Hydroxy-lenalidomide and N-acetyl-lenalidomide represented 4.6% and 1.8% of the excreted dose, respectively. The renal clearance of lenalidomide exceeds the glomerular filtration rate.
Specific Populations
Renal Impairment: Eight subjects with mild renal impairment (creatinine clearance (CLcr) 50 to 79 mL/min calculated using Cockcroft-Gault), 9 subjects with moderate renal impairment (CLcr 30 to 49 mL/min), 4 subjects with severe renal impairment (CLcr < 30 mL/min), and 6 patients with end stage renal disease (ESRD) requiring dialysis were administered a single 25 mg dose of lenalidomide. Three healthy subjects of similar age with normal renal function (CLcr > 80 mL/min) were also administered a single 25 mg dose of lenalidomide. As CLcr decreased, half-life increased and drug clearance decreased linearly. Patients with moderate and severe impairment had a 3-fold increase in half-life and a 66% to 75% decrease in drug clearance compared to healthy subjects. Patients on hemodialysis (n=6) had an approximate 4.5-fold increase in half-life and an 80% decrease in drug clearance compared to healthy subjects. Approximately 30% of the drug in body was removed during a 4-hour hemodialysis session.
Adjust the starting dose of lenalidomide in patients with renal impairment based on the CLcr value [see Dosage and Administration (2.6)].
Hepatic Impairment: Mild hepatic impairment (defined as total bilirubin > 1 to 1.5 times upper limit normal (ULN) or any aspartate transaminase greater than ULN) did not influence the disposition of lenalidomide. No pharmacokinetic data is available for patients with moderate to severe hepatic impairment.
Other Intrinsic Factors: Age (39 to 85 years), body weight (33 to 135 kg), sex, race, and type of hematological malignancies (MM or MDS) did not have a clinically relevant effect on lenalidomide clearance in adult patients.
Drug Interactions
Co-administration of a single dose or multiple doses of dexamethasone (40 mg) had no clinically relevant effect on the multiple dose pharmacokinetics of lenalidomide (25 mg).
Co-administration of lenalidomide (25 mg) after multiple doses of a P-gp inhibitor such as quinidine (600 mg twice daily) did not significantly increase the Cmax or AUC of lenalidomide.
Co-administration of the P-gp inhibitor and substrate temsirolimus (25 mg),with lenalidomide (25 mg) did not significantly alter the pharmacokinetics of lenalidomide, temsirolimus, or sirolimus (metabolite of temsirolimus).
In vitro studies demonstrated that lenalidomide is a substrate of P-glycoprotein (P-gp). Lenalidomide is not a substrate of human breast cancer resistance protein (BCRP), multidrug resistance protein (MRP) transporters MRP1, MRP2, or MRP3, organic anion transporters (OAT) OAT1 and OAT3, organic anion transporting polypeptide 1B1 (OATP1B1), organic cation transporters (OCT) OCT1 and OCT2, multidrug and toxin extrusion protein (MATE) MATE1, and organic cation transporters novel (OCTN) OCTN1 and OCTN2. Lenalidomide is not an inhibitor of P-gp, bile salt export pump (BSEP), BCRP, MRP2, OAT1, OAT3, OATP1B1, OATP1B3, or OCT2. Lenalidomide does not inhibit or induce CYP450 isoenzymes. Also, lenalidomide does not inhibit bilirubin glucuronidation formation in human liver microsomes with UGT1A1 genotyped as UGT1A1*1/*1, UGT1A1*1/*28, and UGT1A1*28/*28.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with lenalidomide have not been conducted.
Lenalidomide was not mutagenic in the bacterial reverse mutation assay (Ames test) and did not induce chromosome aberrations in cultured human peripheral blood lymphocytes, or mutations at the thymidine kinase (tk) locus of mouse lymphoma L5178Y cells. Lenalidomide did not increase morphological transformation in Syrian Hamster Embryo assay or induce micronuclei in the polychromatic erythrocytes of the bone marrow of male rats.
A fertility and early embryonic development study in rats, with administration of lenalidomide up to 500 mg/kg (approximately 200 times the human dose of 25 mg, based on body surface area) produced no parental toxicity and no adverse effects on fertility.
-
14 CLINICAL STUDIES
14.1 Multiple Myeloma
Randomized, Open-Label Clinical Trial in Patients with Newly Diagnosed MM
A randomized multicenter, open-label, 3-arm trial of 1,623 patients, was conducted to compare the efficacy and safety of lenalidomide and low-dose dexamethasone (Rd) given for 2 different durations of time to that of melphalan, prednisone and thalidomide (MPT) in newly diagnosed MM patients who were not a candidate for stem cell transplant. In the first arm of the study, Rd was given continuously until progressive disease [Arm Rd Continuous]. In the second arm, Rd was given for up to eighteen 28-day cycles [72 weeks, Arm Rd18]). In the third arm, melphalan, prednisone and thalidomide (MPT) was given for a maximum of twelve 42-day cycles (72 weeks). For the purposes of this study, a patient who was < 65 years of age was not a candidate for SCT if the patient refused to undergo SCT therapy or the patient did not have access to SCT due to cost or other reasons. Patients were stratified at randomization by age (≤75 versus >75 years), stage (ISS Stages I and II versus Stage III), and country.
Patients in the Rd Continuous and Rd18 arms received lenalidomide 25 mg once daily on Days 1 to 21 of 28-day cycles. Dexamethasone was dosed 40 mg once daily on Days 1, 8, 15, and 22 of each 28-day cycle. For patients over > 75 years old, the starting dose of dexamethasone was 20 mg orally once daily on days 1,8,15, and 22 of repeated 28-day cycles. Initial dose and regimens for Rd Continuous and Rd18 were adjusted according to age and renal function. All patients received prophylactic anticoagulation with the most commonly used being aspirin.
The demographics and disease-related baseline characteristics of the patients were balanced among the 3 arms. In general, study subjects had advanced-stage disease. Of the total study population, the median age was 73 in the 3 arms with 35% of total patients > 75 years of age; 59% had ISS Stage I/II; 41% had ISS stage III; 9% had severe renal impairment (creatinine clearance [CLcr] < 30 mL/min); 23% had moderate renal impairment (CLcr > 30 to 50 mL/min; 44% had mild renal impairment (CLcr > 50 to 80 mL/min). For ECOG Performance Status, 29% were Grade 0, 49% Grade 1, 21% Grade 2, 0.4% ≥ Grade 3.
The primary efficacy endpoint, progression-free survival (PFS), was defined as the time from randomization to the first documentation of disease progression as determined by Independent Response Adjudication Committee (IRAC), based on International Myeloma Working Group [IMWG] criteria or death due to any cause, whichever occurred first during the study until the end of the PFS follow-up phase. For the efficacy analysis of all endpoints, the primary comparison was between Rd Continuous and MPT arms. The efficacy results are summarized in the table below. PFS was significantly longer with Rd Continuous than MPT: HR 0.72 (95% CI: 0.61-0.85 p <0.0001). A lower percentage of subjects in the Rd Continuous arm compared with the MPT arm had PFS events (52% versus 61%, respectively). The improvement in median PFS time in the Rd Continuous arm compared with the MPT arm was 4.3 months. The myeloma response rate was higher with Rd Continuous compared with MPT (75.1% versus 62.3%); with a complete response in 15.1% of Rd Continuous arm patients versus 9.3% in the MPT arm. The median time to first response was 1.8 months in the Rd Continuous arm versus 2.8 months in the MPT arm.
For the interim OS analysis with 03 March 2014 data cutoff, the median follow-up time for all surviving patients is 45.5 months, with 697 death events, representing 78% of prespecified events required for the planned final OS analysis (697/896 of the final OS events). The observed OS HR was 0.75 for Rd Continuous versus MPT (95% CI = 0.62, 0.90).
Table 13 Overview of Efficacy Results – Study MM-020 (Intent-to-treat Population) CR = complete response; d = low-dose dexamethasone; HR = hazard ratio; IRAC = Independent Response Adjudication Committee;
M = melphalan; NE = not estimable; OS = overall survival; P = prednisone; PFS = progression-free survival; PR = partial response;
R = lenalidomide; Rd Continuous = Rd given until documentation of progressive disease; Rd18 = Rd given for ≤ 18 cycles; T = thalidomide; VGPR = very good partial response; vs = versus.
a The median is based on the Kaplan-Meier estimate.
b The 95% Confidence Interval (CI) about the median.
c Based on Cox proportional hazards model comparing the hazard functions associated with the indicated treatment arms.
d The p-value is based on the unstratified log-rank test of Kaplan-Meier curve differences between the indicated treatment arms.
e Best assessment of response during the treatment phase of the study.
f Including patients with no response assessment data or whose only assessment was "response not evaluable."
g Data cutoff date = 24 May 2013.
h Data cutoff date = 3 March 2014.
Rd Continuous
(N = 535)
Rd18 (N = 541)
MPT
(N = 547)
PFS − IRAC (months)g
Number of PFS events
278 (52)
348 (64.3)
334 (61.1)
Mediana PFS time, months (95% CI)b
25.5 (20.7, 29.4)
20.7 (19.4, 22)
21.2 (19.3, 23.2)
HR [95% CI]c; p-valued
Rd Continuous vs. MPT
0.72 (0.61, 0.85);
<0.0001
Rd Continuous vs. Rd18
0.70 (0.60, 0.82)
Rd18 vs. MPT
1.03 (0.89, 1.20)
Overall Survival (months)h
Number of Death events
208 (38.9)
228 (42.1)
261 (47.7)
Mediana OS time, months (95% CI)b
58.9 (56, NE)f
56.7 (50.1, NE)
48.5 (44.2, 52)
HR [95% CI]c
Rd Continuous vs. MPT
0.75 (0.62, 0.90)
Rd Continuous vs. Rd18
0.91 (0.75, 1.09)
Rd18 vs. MPT
0.83 (0.69, 0.99)
Response Ratee − IRAC, n (%)g
CR
81 (15.1)
77 (14.2)
51 (9.3)
VGPR
152 (28.4)
154 (28.5)
103 (18.8)
PR
169 (31.6)
166 (30.7)
187 (34.2)
Overall response: CR, VGPR, or PR
402 (75.1)
397 (73.4)
341 (62.3)
Kaplan-Meier Curves of Progression-free Survival Based on IRAC Assessment (ITT MM Population) Between Arms Rd Continuous, Rd18 and MPT
Cutoff date: 24 May 2013
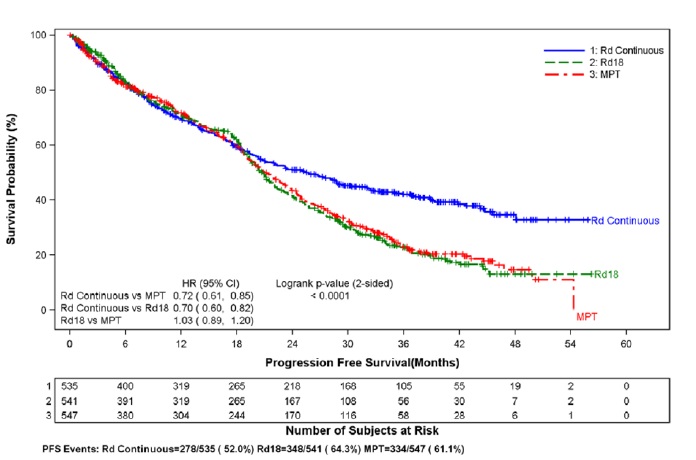
CI = confidence interval; d = low-dose dexamethasone; HR = hazard ratio; IRAC = Independent Response Adjudication Committee; M = melphalan; P = prednisone; R = lenalidomide; Rd Continuous = Rd given until documentation of progressive disease; Rd18 = Rd given for ≤18 cycles; T = thalidomide.
Kaplan-Meier Curves of Overall Survival (ITT MM Population) Between Arms Rd Continuous, Rd18 and MPT
Cutoff date: 03 Mar 2014
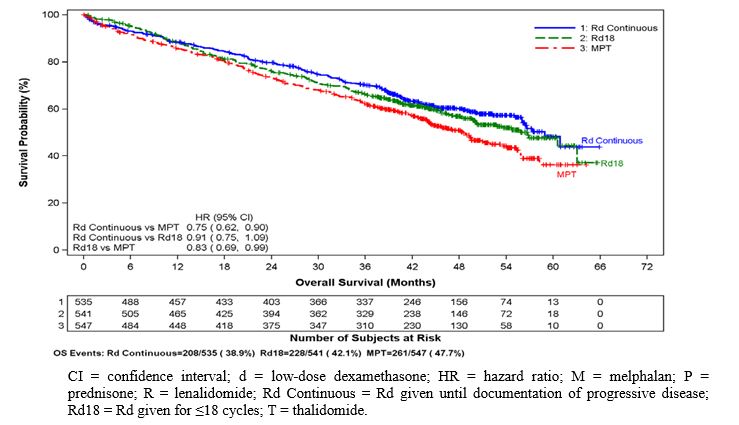
Randomized, Open-Label Clinical Studies in Patients with MM After At Least One Prior Therapy
Two randomized studies (Studies 1 and 2) were conducted to evaluate the efficacy and safety of lenalidomide. These multicenter, multinational, double-blind, placebo-controlled studies compared lenalidomide plus oral pulse high-dose dexamethasone therapy to dexamethasone therapy alone in patients with MM who had received at least one prior treatment. These studies enrolled patients with absolute neutrophil counts (ANC) ≥1000/mm3, platelet counts ≥75,000/mm3, serum creatinine ≤2.5 mg/dL, serum SGOT/AST or SGPT/ALT ≤3 x upper limit of normal (ULN), and serum direct bilirubin ≤2 mg/dL.
In both studies, patients in the lenalidomide/dexamethasone group took 25 mg of lenalidomide orally once daily on Days 1 to 21 and a matching placebo capsule once daily on Days 22 to 28 of each 28-day cycle. Patients in the placebo/dexamethasone group took 1 placebo capsule on Days 1 to 28 of each 28-day cycle. Patients in both treatment groups took 40 mg of dexamethasone orally once daily on Days 1 to 4, 9 to 12, and 17 to 20 of each 28-day cycle for the first 4 cycles of therapy.
The dose of dexamethasone was reduced to 40 mg orally once daily on Days 1 to 4 of each 28-day cycle after the first 4 cycles of therapy. In both studies, treatment was to continue until disease progression.
In both studies, dose adjustments were allowed based on clinical and laboratory findings. Sequential dose reductions to 15 mg daily, 10 mg daily and 5 mg daily were allowed for toxicity [see Dosage and Administration (2.1)].
Table 16 summarizes the baseline patient and disease characteristics in the two studies. In both studies, baseline demographic and disease-related characteristics were comparable between the lenalidomide/dexamethasone and placebo/dexamethasone groups.
Table 16 Baseline Demographic and Disease-Related Characteristics – MM Studies 1 and 2 Study 1
Study 2
Lenalidomide/Dex
Placebo/Dex
Lenalidomide/Dex
Placebo/Dex
N=177
N=176
N=176
N=175
Patient Characteristics
Age (years)
Median
64
62
63
64
Min, Max
36, 86
37, 85
33, 84
40, 82
Sex
Male
106 (60%)
104 (59%)
104 (59%)
103 (59%)
Female
71 (40%)
72 (41%)
72 (41%)
72 (41%)
Race/Ethnicity
White
141(80%)
148 (84%)
172 (98%)
175(100%)
Other
36 (20%)
28 (16%)
4 (2%)
0 (0%)
ECOG Performance
Status 0 to 1
157 (89%)
168 (95%)
150 (85%)
144 (82%)
Disease Characteristics
Multiple Myeloma Stage (Durie-Salmon)
I
3%
3%
6%
5%
II
32%
31%
28%
33%
III
64%
66%
65%
63%
B2-microglobulin (mg/L)
≤2.5 mg/L
52 (29%)
51 (29%)
51 (29%)
48 (27%)
>2.5 mg/L
125 (71%)
125 (71%)
125 (71%)
127 (73%)
Number of Prior Therapies
1
38%
38%
32%
33%
≥2
62%
62%
68%
67%
Types of Prior Therapies
Stem Cell Transplantation
62%
61%
55%
54%
Thalidomide
42%
46%
30%
38%
Dexamethasone
81%
71%
66%
69%
Bortezomib
11%
11%
5%
4%
Melphalan
33%
31%
56%
52%
Doxorubicin
55%
51%
56%
57%
The primary efficacy endpoint in both studies was time to progression (TTP). TTP was defined as the time from randomization to the first occurrence of progressive disease.
Preplanned interim analyses of both studies showed that the combination of lenalidomide/dexamethasone was significantly superior to dexamethasone alone for TTP. The studies were unblinded to allow patients in the placebo/dexamethasone group to receive treatment with the lenalidomide/dexamethasone combination. For both studies, the extended follow-up survival data with crossovers were analyzed. In study 1, the median survival time was 39.4 months (95%CI: 32.9, 47.4) in lenalidomide/dexamethasone group and 31.6 months (95%CI: 24.1, 40.9) in placebo/dexamethasone group, with a hazard ratio of 0.79 (95% CI: 0.61 to 1.03). In study 2, the median survival time was 37.5 months (95%CI: 29.9, 46.6) in lenalidomide/dexamethasone group and 30.8 months (95%CI: 23.5, 40.3) in placebo/dexamethasone group, with a hazard ratio of 0.86 (95% CI: 0.65 to 1.14).
Table 17 TTP Results in MM Study 1 and Study 2 Study 1
Study 2
Lenalidomide/Dex
N=177
Placebo/Dex
N=176
Lenalidomide/Dex
N=176
Placebo/Dex
N=175
TTP
Events n (%)
73 (41)
120 (68)
68 (39)
130 (74)
Median TTP in months
13.9
4.7
12.1
4.7
[95% CI]
[9.5, 18.5]
[3.7, 4.9]
[9.5, NE]
[3.8, 4.8]
Hazard Ratio
0.285
0.324
[95% CI]
[0.210, 0.386]
[0.240, 0.438]
Log-rank Test p-value 3
<0.001
<0.001
Response
Complete Response (CR) n (%)
23 (13)
1 (1)
27 (15)
7 (4)
Partial Response (RR/PR) n (%)
84 (48)
33 (19)
77 (44)
34 (19)
Overall Response n (%)
107 (61)
34 (19)
104 (59)
41 (23)
p-value
<0.001
<0.001
Odds Ratio
6.38
4.72
[95% CI]
[3.95, 10.32]
[2.98, 7.49]
Kaplan-Meier Estimate of Time to Progression — MM Study 1
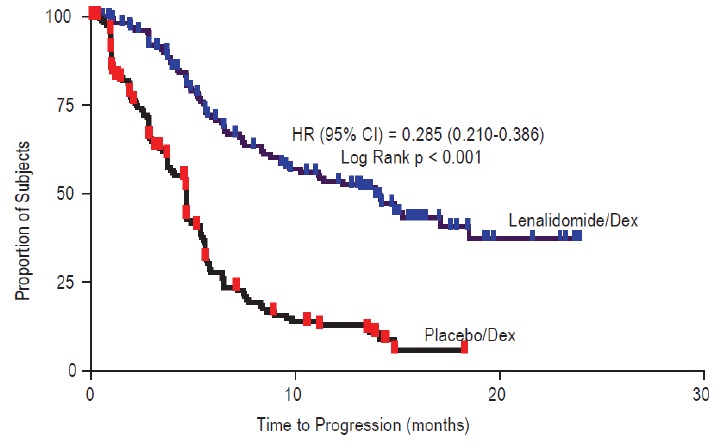
Kaplan-Meier Estimate of Time to Progression — MM Study 2
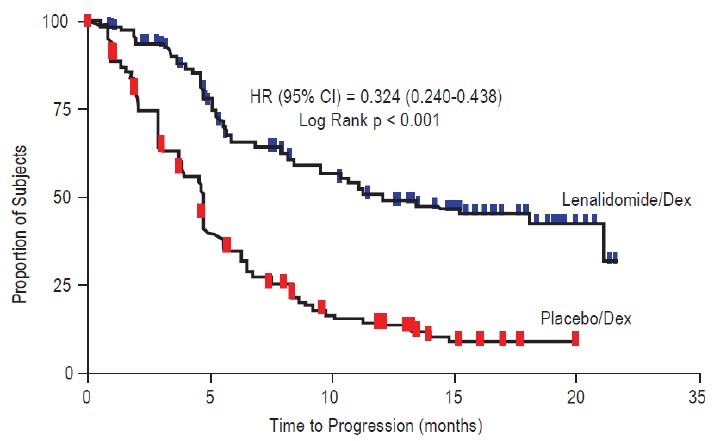
14.2 Myelodysplastic Syndromes (MDS) with a Deletion 5q Cytogenetic Abnormality
The efficacy and safety of lenalidomide were evaluated in patients with transfusion-dependent anemia in low- or intermediate-1- risk MDS with a 5q (q31-33) cytogenetic abnormality in isolation or with additional cytogenetic abnormalities, at a dose of 10 mg once daily or 10 mg once daily for 21 days every 28 days in an open-label, single-arm, multi-center study. The major study was not designed nor powered to prospectively compare the efficacy of the 2 dosing regimens. Sequential dose reductions to 5 mg daily and 5 mg every other day, as well as dose delays, were allowed for toxicity [Dosage and Administration (2.2)].
This major study enrolled 148 patients who had RBC transfusion dependent anemia. RBC transfusion dependence was defined as having received ≥ 2 units of RBCs within 8 weeks prior to study treatment. The study enrolled patients with absolute neutrophil counts (ANC) ≥ 500/mm3, platelet counts ≥ 50,000/mm3, serum creatinine ≤ 2.5 mg/dL, serum SGOT/AST or SGPT/ALT ≤ 3 x upper limit of normal (ULN), and serum direct bilirubin ≤ 2 mg/dL. Granulocyte colony-stimulating factor was permitted for patients who developed neutropenia or fever in association with neutropenia. Baseline patient and disease-related characteristics are summarized in Table 18.
Table 18
Baseline Demographic and Disease-Related Characteristics in the MDS Study
Overall
(N=148)
Age (years)
Median
Min, Max
71
37, 95
G ender
n (%)
Male
51 (34.5)
Female
97 (65.5)
Race
n (%)
White
143 (96.6)
Other
5 (3.4)
Duration of MDS (years)
n (%)
Median
2.5
Min, Max
0.1, 20.7
Del 5 (q31-33) Cytogenetic Abnormality
n (%)
Yes
148 (100)
Other cytogenetic abnormalities
37 (25.2)
IPSS Score a
n (%)
Low (0)
55 (37.2)
Intermediate-1 (0.5-1.0)
65 (43.9)
Intermediate-2 (1.5-2.0)
6 (4.1)
High (≥2.5)
2 (1.4)
Missing
20 (13.5)
FAB Classification b from central review
n (%)
RA
77 (52)
RARS
16 (10.8)
RAEB
30 (20.3)
CMML
3 (2)
The frequency of RBC transfusion independence was assessed using criteria modified from the International Working Group (IWG) response criteria for MDS. RBC transfusion independence was defined as the absence of any RBC transfusion during any consecutive "rolling" 56 days (8 weeks) during the treatment period.
Transfusion independence was seen in 99/148 (67%) patients (95% CI [59, 74]). The median duration from the date when RBC transfusion independence was first declared (i.e., the last day of the 56-day RBC transfusion-free period) to the date when an additional transfusion was received after the 56-day transfusion-free period among the 99 responders was 44 weeks (range of 0 to >67 weeks). Ninety percent of patients who achieved a transfusion benefit did so by completion of three months in the study.
RBC transfusion independence rates were unaffected by age or gender.
The dose of lenalidomide was reduced or interrupted at least once due to an adverse event in 118 (79.7%) of the 148 patients; the median time to the first dose reduction or interruption was 21 days (mean, 35.1 days; range, 2-253 days), and the median duration of the first dose interruption was 22 days (mean, 28.5 days; range, 2-265 days). A second dose reduction or interruption due to adverse events was required in 50 (33.8%) of the 148 patients. The median interval between the first and second dose reduction or interruption was 51 days (mean, 59.7 days; range, 15-205 days) and the median duration of the second dose interruption was 21 days (mean, 26 days; range, 2-148 days).
-
15 REFERENCES
- OSHA Hazardous Drugs. OSHA [Accessed on 29 January 2013, from http://www.osha.gov/SLTC/hazardousdrugs/index.html]
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
Lenalidomide Capsules, 2.5 mg are white to off-white powder filled in size '5' hard gelatin capsule with white opaque colored cap imprinted with "1030" in black ink and blue opaque color body and are supplied as follows:
NDC 70710-1030-7 in bottle of 28 capsules with child-resistant closure.
NDC 70710-1030-1 in bottle of 100 capsules with child-resistant closure.
Lenalidomide Capsules, 5 mg are white to off-white powder filled in size '5' hard gelatin capsule with white opaque colored cap imprinted with "1031" in black ink and white opaque color body and are supplied as follows:
NDC 70710-1031-7 in bottle of 28 capsules with child-resistant closure.
NDC 70710-1031-1 in bottle of 100 capsules with child-resistant closure.
Lenalidomide Capsules, 10 mg are white to off-white powder filled in size '3' hard gelatin capsule with white opaque colored cap imprinted with "1032" in black ink and turquoise blue opaque color body and are supplied as follows:
NDC 70710-1032-7 in bottle of 28 capsules with child-resistant closure.
NDC 70710-1032-1 in bottle of 100 capsules with child-resistant closure.
Lenalidomide Capsules, 15 mg are white to off-white powder filled in size '2' hard gelatin capsule with white opaque colored cap imprinted with "1033" in black ink and light blue opaque color body and are supplied as follows:
NDC 70710-1033-8 in bottle of 21 capsules with child-resistant closure.
NDC 70710-1033-1 in bottle of 100 capsules with child-resistant closure.
Lenalidomide Capsules, 20 mg are white to off-white powder filled in size '1' hard gelatin capsule with light blue opaque colored cap imprinted with "1034" in black ink and turquoise blue opaque color body and are supplied as follows:
NDC 70710-1034-8 in bottle of 21 capsules with child-resistant closure.
NDC 70710-1034-1 in bottle of 100 capsules with child-resistant closure.
Lenalidomide Capsules, 25 mg are white to off-white powder filled in size '0' hard gelatin capsule with white opaque colored cap imprinted with "1035" in black ink and light blue opaque color body and are supplied as follows:
NDC 70710-1035-8 in bottle of 21 capsules with child-resistant closure.
NDC 70710-1035-1 in bottle of 100 capsules with child-resistant closure.
16.2 Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature].
16.3 Handling and Disposal
Care should be exercised in the handling of lenalidomide. Lenalidomide capsules should not be opened or broken. If powder from lenalidomide contacts the skin, wash the skin immediately and thoroughly with soap and water. If lenalidomide contacts the mucous membranes, flush thoroughly with water.
Procedures for the proper handling and disposal of anticancer drugs should be considered. Several guidelines on the subject have been published.1
Dispense no more than a 28-day supply.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved Patient labeling (Medication Guide)
Embryo-Fetal Toxicity
Advise patients that lenalidomide is contraindicated in pregnancy [see Boxed Warning and Contraindications (4.1)]. Lenalidomide is a thalidomide analogue and can cause serious birth defects or death to a developing baby [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
- Advise females of reproductive potential that they must avoid pregnancy while taking lenalidomide and for at least 4 weeks after completing therapy.
- Initiate lenalidomide treatment in females of reproductive potential only following a negative pregnancy test.
- Advise females of reproductive potential of the importance of monthly pregnancy tests and the need to use 2 different forms of contraception including at least 1 highly effective form, simultaneously during lenalidomide therapy, during dose interruption and for 4 weeks after she has completely finished taking lenalidomide. Highly effective forms of contraception other than tubal ligation include IUD and hormonal (birth control pills, injections, patch or implants) and a partner's vasectomy. Additional effective contraceptive methods include latex or synthetic condom, diaphragm and cervical cap.
- Instruct patient to immediately stop taking lenalidomide and contact her healthcare provider if she becomes pregnant while taking this drug, if she misses her menstrual period, or experiences unusual menstrual bleeding, if she stops taking birth control, or if she thinks FOR ANY REASON that she may be pregnant.
- Advise patient that if her healthcare provider is not available, she should call the REMS Call Center at 1‐888‐423‐5436 [see Warnings and Precautions (5.1) and Use in Specific Populations (8.3)] .
- Advise males to always use a latex or synthetic condom during any sexual contact with females of reproductive potential while taking lenalidomide and for up to 4 weeks after discontinuing lenalidomide, even if they have undergone a successful vasectomy.
- Advise male patients taking lenalidomide that they must not donate sperm and for up to 4 weeks after discontinuation of lenalidomide [see Warnings and Precautions (5.1) and Use in Specific Populations (8.3)].
- All patients must be instructed to not donate blood while taking lenalidomide, during dose interruptions and for 4 weeks following discontinuation of lenalidomide [see Warnings and Precautions (5.1)].
Because of the risk of embryo-fetal toxicity, lenalidomide is only available through a restricted program called the Lenalidomide REMS program [see Warnings and Precautions (5.2)].
- Patients must sign a Patient-Physician agreement form and comply with the requirements to receive lenalidomide. In particular, females of reproductive potential must comply with the pregnancy testing, contraception requirements and participate in monthly telephone surveys. Males must comply with the contraception requirements [see Use in Specific Populations (8.3)].
- Lenalidomide is available only from pharmacies that are certified in Lenalidomide REMS program. Provide patients with the telephone number and website for information on how to obtain the product.
Inform females there is a Pregnancy Exposure Registry that monitors pregnancy outcomes in females exposed to lenalidomide during pregnancy and that they can contact the Pregnancy Exposure Registry by calling 1-888-423-5436 [see Use in Specific Populations (8.1)].
Hematologic Toxicity
Inform patients that lenalidomide is associated with significant neutropenia and thrombocytopenia [see Boxed Warning and Warnings and Precautions (5.3)].
Venous and Arterial Thromboembolism
Inform patients of the risk of thrombosis including DVT, PE, MI, and stroke and to report immediately any signs and symptoms suggestive of these events for evaluation [see Boxed Warning and Warnings and Precautions (5.4)].
Increased Mortality in Patients with CLL
Inform patients that lenalidomide had increased mortality in patients with CLL and serious adverse cardiovascular reactions, including atrial fibrillation, myocardial infarction, and cardiac failure [see Warnings and Precautions (5.5)].
Second Primary Malignancies
Inform patients of the potential risk of developing second primary malignancies during treatment with lenalidomide [see Warnings and Precautions (5.6)].
Hepatotoxicity
Inform patients of the risk of hepatotoxicity, including hepatic failure and death, and to report any signs and symptoms associated with this event to their healthcare provider for evaluation [see Warnings and Precautions (5.8)].
Severe Cutaneous Reactions
Inform patients of the potential risk for severe skin reactions such as SJS, TEN, and DRESS and report any signs and symptoms associated with these reactions to their healthcare provider for evaluation. Patients with a prior history of Grade 4 rash associated with thalidomide treatment should not receive lenalidomide [see Warnings and Precautions (5.9)].
Tumor Lysis Syndrome
Inform patients of the potential risk of tumor lysis syndrome and to report any signs and symptoms associated with this event to their healthcare provider for evaluation [see Warnings and Precautions (5.10)].
Tumor Flare Reaction
Inform patients of the potential risk of tumor flare reaction and to report any signs and symptoms associated with this event to their healthcare provider for evaluation [see Warnings and Precautions (5.11)].
Hypersensitivity
Inform patients of the potential for severe hypersensitivity reactions such as angioedema and anaphylaxis to lenalidomide. Instruct patients to contact their healthcare provider right away for signs and symptoms of these reactions. Advise patients to seek emergency medical attention for signs or symptoms of severe hypersensitivity reactions [see Warnings and Precautions (5.15)].
Dosing Instructions
Inform patients how to take lenalidomide [see Dosage and Administration (2)]
- Lenalidomide should be taken once daily at about the same time each day,
- Lenalidomide may be taken either with or without food.
- The capsules should not be opened, broken, or chewed. Lenalidomide should be swallowed whole with water.
- Instruct patients that if they miss a dose of lenalidomide, they may still take it up to 12 hours after the time they would normally take it. If more than 12 hours have elapsed, they should be instructed to skip the dose for that day. The next day, they should take lenalidomide at the usual time. Warn patients to not take 2 doses to make up for the one that they missed.
Medication Guide available at www.zydususa.com/medguides or call 1-877-993-8779.
- SPL UNCLASSIFIED SECTION
-
SPL MEDGUIDE
MEDICATION GUIDE
Lenalidomide (len" a lid' oh mide) Capsules
What is the most important information I should know about lenalidomide capsules?
Before you begin taking lenalidomide, you must read and agree to all of the instructions in the Lenalidomide REMS program. Before prescribing lenalidomide, your healthcare provider will explain the Lenalidomide REMS program to you and have you sign the Patient-Physician Agreement Form.
Lenalidomide may cause serious side effects including:
- Possible birth defects (deformed babies) or death of an unborn baby.
Lenalidomide is similar to the medicine thalidomide. We know thalidomide can cause severe life-threatening birth defects. Lenalidomide has not been tested in pregnant females. Lenalidomide has harmed unborn animals in animal testing.
Females must not get pregnant:
o For at least 4 weeks before starting lenalidomide
o While taking lenalidomide
o During any breaks (interruptions) in your treatment with lenalidomide
o For at least 4 weeks after stopping lenalidomide
Females who can become pregnant:
- Will have pregnancy tests weekly for 4 weeks, then every 4 weeks if your menstrual cycle is regular, or every 2 weeks if your menstrual cycle is irregular.
- If you miss your period or have unusual bleeding, you will need to have a pregnancy test and receive counseling.
- Must agree to use two acceptable forms of birth control at the same time, for at least 4 weeks before, while taking, during any breaks (interruptions) in your treatment, and for at least 4 weeks after stopping lenalidomide.
- Talk with your healthcare provider to find out about options for acceptable forms of birth control that you may use to prevent pregnancy before, during, and after treatment with lenalidomide.
- If you had unprotected sex or if you think your birth control has failed, stop taking lenalidomide immediately and call your healthcare provider right away.
Healthcare providers and patients should report all cases of pregnancy to:
- FDA MedWatch at 1-800-FDA-1088
- The Lenalidomide REMS program at 1-888-423-5436
Lenalidomide can pass into human semen:
- Males, including those who have had a vasectomy, must always use a latex or synthetic condom during any sexual contact with a pregnant female or a female that can become pregnant while taking lenalidomide, during any breaks (interruptions) in your treatment with lenalidomide, and for up to 4 weeks after stopping lenalidomide.
- Do not have unprotected sexual contact with a female who is or could become pregnant. Tell your healthcare provider if you do have unprotected sexual contact with a female who is or could become pregnant.
- Do not donate sperm while taking lenalidomide, during any breaks (interruptions) in your treatment, and for up to 4 weeks after stopping lenalidomide. If a female becomes pregnant with your sperm, the baby may be exposed to lenalidomide and may be born with birth defects.
- Low white blood cells (neutropenia) and low platelets (thrombocytopenia). Lenalidomide causes low white blood cells and low platelets in most people. You may need a blood transfusion or certain medicines if your blood counts drop too low. Your healthcare provider should check your blood counts often especially during the first several months of treatment with lenalidomide, and then at least monthly. Tell your healthcare provider if you develop any bleeding or bruising, during treatment with lenalidomide.
- Blood clots. Blood clots in the arteries, veins, and lungs happen more often in people who take lenalidomide. This risk is even higher for people with multiple myeloma who take the medicine dexamethasone with lenalidomide. Heart attacks and strokes also happen more often in people who take lenalidomide with dexamethasone. To reduce this increased risk, most people who take lenalidomide will also take a blood thinner medicine.
o If you have had a blood clot in the past
o If you have high blood pressure, smoke, or if you have been told you have a high level of fat in your blood (hyperlipidemia)
o About all the medicines you take. Certain other medicines can also increase your risk for blood clots
Call your healthcare provider or get medical help right away if you get any of the following during treatment with lenalidomide:
- Signs or symptoms of a blood clot in the lung, arm, or leg may include: shortness of breath, chest pain, or arm or leg swelling
- Signs or symptoms of a heart attack may include: chest pain that may spread to the arms, neck, jaw, back, or stomach area (abdomen), feeling sweaty, shortness of breath, feeling sick or vomiting
- Signs or symptoms of stroke may include: sudden numbness or weakness, especially on one side of the body, severe headache or confusion, or problems with vision, speech, or balance.
What are lenalidomide capsules?
Lenalidomide capsules are prescription medicine, used to treat adults with:
- multiple myeloma (MM)
- a condition called myelodysplastic syndromes (MDS). Lenalidomide is for the type of MDS with a chromosome problem where part of chromosome 5 is missing. This type of MDS is known as deletion 5q MDS. People with this type of MDS may have low red blood cell counts that require treatment with blood transfusions.
It is not known if lenalidomide is safe and effective in children.
Who should not take lenalidomide capsules?
Do not take lenalidomide capsules if you :
- are pregnant, plan to become pregnant, or become pregnant during treatment with lenalidomide capsules. See "What is the most important information I should know about lenalidomide capsules?"
- are allergic to lenalidomide or any of the ingredients in lenalidomide capsules. See the end of this Medication Guide for a complete list of ingredients in lenalidomide capsules.
What should I tell my healthcare provider before taking lenalidomide capsules?
Before you take Lenalidomide, tell your healthcare provider about all of your medical conditions, including if you:
- have liver problems
- have kidney problems or receive kidney dialysis treatment
- have thyroid problems
- have had a serious skin rash with thalidomide treatment. You should not take lenalidomide.
- are lactose intolerant. Lenalidomide capsules contain lactose.
- are breastfeeding. Do not breastfeed during treatment with lenalidomide. It is not known if lenalidomide passes into your breast milk and can harm your baby.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist.
How should I take lenalidomide capsules?
- Take lenalidomide capsules exactly as prescribed and follow all the instructions of the Lenalidomide REMS program
- Swallow lenalidomide capsules whole with water 1 time a day. Do not open, break, or chew your capsules.
- Lenalidomide capsules may be taken with or without food.
- Take lenalidomide capsules at about the same time each day.
- Do not open or break lenalidomide capsules or handle them any more than needed.
o If powder from the lenalidomide capsule comes in contact with the inside of your eyes, nose, or mouth, flush well with water.
- If you miss a dose of lenalidomide and it has been less than 12 hours since your regular time, take it as soon as you remember. If it has been more than 12 hours, just skip your missed dose. Do not take 2 doses at the same time.
- If you take too much lenalidomide, call your healthcare provider right away.
What should I avoid while taking lenalidomide capsules?
- See "What is the most important information I should know about lenalidomide capsules?"
- Females: Do not get pregnant and do not breastfeed while taking lenalidomide capsules.
- Males: Do not donate sperm while taking lenalidomide capsules, during any breaks (interruptions) in your treatment, and for up to 4 weeks after stopping lenalidomide capsules.
- Do not share lenalidomide capsules with other people. It may cause birth defects and other serious problems.
- Do not donate blood while you take lenalidomide capsules, during any breaks (interruptions) in your treatment, and for 4 weeks after stopping lenalidomide capsules. If someone who is pregnant gets your donated blood, her baby may be exposed to lenalidomide capsules and may be born with birth defects.
What are the possible side effects of lenalidomide capsules?
Lenalidomide capsules can cause serious side effects, including:
- See "What is the most important information I should know about lenalidomide capsules?"
- Increased risk of death in people who have chronic lymphocytic leukemia (CLL). People with CLL who take lenalidomide capsules have an increased risk of death compared with people who take the medicine chlorambucil. Lenalidomide capsules may cause you to have serious heart problems that can lead to death, including atrial fibrillation, heart attack, or heart failure. You should not take lenalidomide capsules if you have CLL unless you are participating in a controlled clinical trial.
- Risk of new cancers (malignancies). An increase in new (second) cancers has happened in patients who received lenalidomide capsules and melphalan, or a blood stem cell transplant, including certain blood cancers, such as acute myelogenous leukemia (AML), and myelodysplastic syndrome (MDS) and certain other types of cancers of the skin and other organs. Talk with your healthcare provider about your risk of developing new cancers if you take lenalidomide capsules. Your healthcare provider will check you for new cancers during your treatment with lenalidomide capsules.
- Severe liver problems, including liver failure and death. Your healthcare provider should do blood tests to check your liver function during your treatment with lenalidomide capsules. Tell your healthcare provider right away if you develop any of the following symptoms of liver problems:
- yellowing of your skin or the white part of your eyes (jaundice)
- pain on the upper right side of your stomach area (abdomen)
- dark or brown (tea colored) urine
- bleeding or bruising more easily than normal
- feeling very tired
- Severe skin reactions and severe allergic reactions can happen with lenalidomide capsules and may cause death.
- a red, itchy, skin rash
- peeling of your skin or blisters
- severe itching
- fever
Get emergency medical help right away if you develop any of the following signs or symptoms during treatment with lenalidomide capsules:
- swelling of your lips, mouth, tongue, or throat
- raised red areas on your skin (hives)
- trouble breathing or swallowing
- a very fast heartbeat
- you feel dizzy or faint
- Tumor lysis syndrome (TLS). TLS is caused by the fast breakdown of cancer cells. TLS can cause kidney failure and the need for dialysis treatment, abnormal heart rhythm, seizure and sometimes death. Your healthcare provider may do blood tests to check you for TLS.
- Worsening of your tumor (tumor flare reaction) can happen with lenalidomide capsules and may cause death. Tell your healthcare provider if you get any of these symptoms of tumor flare reaction during treatment with lenalidomide capsules: tender swollen lymph nodes, low grade fever, pain, or rash. Your healthcare provider may tell you to decrease your dose, temporarily stop or permanently stop taking lenalidomide capsules if you develop certain serious side effects during treatment with lenalidomide capsules.
- Thyroid problems. Your healthcare provider may check your thyroid function before you start taking lenalidomide capsules and during treatment with lenalidomide capsules.
- diarrhea
- swelling of your arms, hands, legs, feet and skin
- upper respiratory tract infection or bronchitis
- rash
- sleep problems (insomnia)
- inflammation of the stomach and intestine ("stomach flu")
- nausea
- headache
- nose bleed
- constipation
- muscle cramps or spasms
- shaking or trembling (tremor)
- tiredness or weakness
- shortness of breath
- joint aches
- fever
- cough, sore throat, and other symptoms of a cold
- pain in your back or stomach-area (abdomen)
- itching
These are not all the possible side effects of lenalidomide capsules.
Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
How should I store lenalidomide capsules?
- Store lenalidomide capsules at room temperature between 68°F to 77°F (20°C to 25°C).
- Return any unused lenalidomide capsules to your healthcare provider.
- Drug lenalidomide capsules come in child-resistant closure.
General information about the safe and effective use of lenalidomide capsules
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not take lenalidomide capsules for conditions for which it was not prescribed. Do not give lenalidomide capsules to other people, even if they have the same symptoms you have. It may harm them and may cause birth defects. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about lenalidomide capsules that is written for health professionals.
Please address medical inquiries to, MedicalAffairs@zydususa.com or Tel: 1-877-993-8779.
What are the ingredients in lenalidomide capsules?
Active ingredient: lenalidomide
Inactive ingredients: croscarmellose sodium, gelatin, lactose anhydrous, lactose monohydrate, magnesium stearate, microcrystalline cellulose and titanium dioxide.
Each 2.5 mg capsule shell contains: FD& C Blue # 1, FD& C Red#40, FD&C Yellow # 6 and black ink.
Each 10 mg capsule shell contains: D&C Red # 33, D&C Yellow# 10, FD & C Blue # 1 and black ink.
Each 15 mg capsule shell contains: D&C Red # 28, FD & C Blue # 1 and black ink.
Each 20 mg capsule shell contains: D&C Red # 28, D&C Red # 33, D&C Yellow # 10, FD & C Blue # 1 and black ink.
Each 25 mg capsule shell contains: D&C Red # 28, FD & C Blue # 1 and black ink.
Medication Guide available at www.zydususa.com/medguides or call 1-877-993-8779.
Manufactured by:
Zydus Lifesciences Ltd.
Ahmedabad, India
Distributed by:
Zydus Pharmaceuticals (USA) Inc.
Pennington, NJ 08534
Rev.:04/23
This Medication Guide has been approved by the U.S. Food and Drug Administration.
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Lenalidomide Capsules, 2.5 mg
28 Capsules
Rx only
Zydus

sl-2.5mg-28c
Lenalidomide Capsules, 5 mg
28 Capsules
Rx only
Zydus

sl-5mg-28c
Lenalidomide Capsules, 10mg
28 Capsules
Rx only
Zydus

sl-10mg-28c
Lenalidomide Capsules, 15mg
21 Capsules
Rx only
Zydus

sl-15mg-21c
Lenalidomide Capsules, 20mg
21 Capsules
Rx only
Zydus

SL-20mg-21c
Lenalidomide Capsules, 25mg
21 Capsules
Rx only
Zydus

SL-25mg-21c
-
INGREDIENTS AND APPEARANCE
LENALIDOMIDE
lenalidomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70710-1031 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LENALIDOMIDE (UNII: F0P408N6V4) (LENALIDOMIDE - UNII:F0P408N6V4) LENALIDOMIDE 5 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color WHITE (white opaque cap with white opaque body) Score no score Shape CAPSULE (capsule) Size 11mm Flavor Imprint Code 1031 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70710-1031-7 28 in 1 BOTTLE; Type 0: Not a Combination Product 09/12/2022 2 NDC:70710-1031-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 09/12/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210154 09/12/2022 LENALIDOMIDE
lenalidomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70710-1032 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LENALIDOMIDE (UNII: F0P408N6V4) (LENALIDOMIDE - UNII:F0P408N6V4) LENALIDOMIDE 10 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C RED NO. 33 (UNII: 9DBA0SBB0L) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color WHITE (white opaque cap) , BLUE (Turquoise blue opaque body) Score no score Shape CAPSULE (capsule) Size 16mm Flavor Imprint Code 1032 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70710-1032-7 28 in 1 BOTTLE; Type 0: Not a Combination Product 09/12/2022 2 NDC:70710-1032-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 09/12/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210154 09/12/2022 LENALIDOMIDE
lenalidomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70710-1033 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LENALIDOMIDE (UNII: F0P408N6V4) (LENALIDOMIDE - UNII:F0P408N6V4) LENALIDOMIDE 15 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C RED NO. 28 (UNII: 767IP0Y5NH) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color WHITE (white opaque cap) , BLUE (Light blue opaque body) Score no score Shape CAPSULE (capsule) Size 18mm Flavor Imprint Code 1033 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70710-1033-8 21 in 1 BOTTLE; Type 0: Not a Combination Product 09/12/2022 2 NDC:70710-1033-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 09/12/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210154 09/12/2022 LENALIDOMIDE
lenalidomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70710-1035 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LENALIDOMIDE (UNII: F0P408N6V4) (LENALIDOMIDE - UNII:F0P408N6V4) LENALIDOMIDE 25 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C RED NO. 28 (UNII: 767IP0Y5NH) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color WHITE (white opaque cap) , BLUE (Light Blue opaque body) Score no score Shape CAPSULE (capsule) Size 22mm Flavor Imprint Code 1035 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70710-1035-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 09/12/2022 2 NDC:70710-1035-8 21 in 1 BOTTLE; Type 0: Not a Combination Product 09/12/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210154 09/12/2022 LENALIDOMIDE
lenalidomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70710-1030 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LENALIDOMIDE (UNII: F0P408N6V4) (LENALIDOMIDE - UNII:F0P408N6V4) LENALIDOMIDE 2.5 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color WHITE (opaque white cap) , BLUE (blue opaque body) Score no score Shape CAPSULE (capsule) Size 11mm Flavor Imprint Code 1030 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70710-1030-7 28 in 1 BOTTLE; Type 0: Not a Combination Product 03/07/2023 2 NDC:70710-1030-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/07/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210154 03/07/2023 LENALIDOMIDE
lenalidomide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70710-1034 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LENALIDOMIDE (UNII: F0P408N6V4) (LENALIDOMIDE - UNII:F0P408N6V4) LENALIDOMIDE 20 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C RED NO. 28 (UNII: 767IP0Y5NH) D&C RED NO. 33 (UNII: 9DBA0SBB0L) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FERROSOFERRIC OXIDE (UNII: XM0M87F357) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color BLUE (light blue opaque cap turquoise blue opaque body) Score no score Shape CAPSULE (capsule) Size 19mm Flavor Imprint Code 1034 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70710-1034-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/07/2023 2 NDC:70710-1034-8 21 in 1 BOTTLE; Type 0: Not a Combination Product 03/07/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210154 03/07/2023 Labeler - Zydus Pharmaceuticals USA Inc. (156861945) Registrant - Zydus Pharmaceuticals USA Inc. (156861945) Establishment Name Address ID/FEI Business Operations Zydus Lifesciences Limited 863362789 ANALYSIS(70710-1030, 70710-1031, 70710-1032, 70710-1033, 70710-1034, 70710-1035) , MANUFACTURE(70710-1030, 70710-1031, 70710-1032, 70710-1033, 70710-1034, 70710-1035)






