Label: METFORMIN HYDROCHLORIDE ER- metformin hydrochloride tablet, extended release
-
Contains inactivated NDC Code(s)
NDC Code(s): 51655-550-25, 51655-550-82 - Packager: Northwind Pharmaceuticals
- This is a repackaged label.
- Source NDC Code(s): 60505-0260
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated September 30, 2014
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Patient Information
Information for Patients
Patients should be informed of the potential risks and benefits of metformin and of alternative modes of therapy. They should also be informed about the importance of adherence to dietary instructions, of a regular exercise program, and of regular testing of blood glucose, glycosylated hemoglobin, renal function, and hematologic parameters.
The risks of lactic acidosis, its symptoms, and conditions that predispose to its development, as noted in the WARNINGS and PRECAUTIONS sections, should be explained to patients. Patients should be advised to discontinue metformin immediately and to promptly notify their health practitioner if unexplained hyperventilation, myalgia, malaise, unusual somnolence, or other nonspecific symptoms occur. Once a patient is stabilized on any dose level of metformin, gastrointestinal symptoms, which are common during initiation of metformin therapy, are unlikely to be drug related. Later occurrence of gastrointestinal symptoms could be due to lactic acidosis or other serious disease.
Patients should be counselled against excessive alcohol intake, either acute or chronic, while receiving metformin.
Metformin alone does not usually cause hypoglycemia, although it may occur when metformin is used in conjunction with oral sulfonylureas and insulin. When initiating combination therapy, the risks of hypoglycemia, its symptoms and treatment, and conditions that predispose to its development should be explained to patients and responsible family members.
Patients should be informed that metformin hydrochloride extended-release tablets must be swallowed whole and not crushed or chewed, and that the inactive ingredients may occasionally be eliminated in the feces as a soft mass that may resemble the original tablet.
Laboratory Tests
Response to all diabetic therapies should be monitored by periodic measurements of fasting blood glucose and glycosylated hemoglobin levels, with a goal of decreasing these levels toward the normal range. During initial dose titration, fasting glucose can be used to determine the therapeutic response. Thereafter, both glucose and glycosylated hemoglobin should be monitored. Measurements of glycosylated hemoglobin may be especially useful for evaluating long-term control.
Initial and periodic monitoring of hematologic parameters (e.g., hemoglobin/hematocrit and red blood cell indices) and renal function (serum creatinine) should be performed, at least on an annual basis. While megaloblastic anemia has rarely been seen with metformin hydrochloride tablets therapy, if this is suspected, vitamin B12 deficiency should be excluded.
-
Drug Interactions
Drug Interactions (clinical evaluation of drug interactions done with metformin hydrochloride tablets)
Glyburide: In a single-dose interaction study in type 2 diabetes patients, co-administration of metformin and glyburide did not result in any changes in either metformin pharmacokinetics or pharmacodynamics. Decreases in glyburide AUC and Cmax were observed, but were highly variable. The single-dose nature of this study and the lack of correlation between glyburide blood levels and pharmacodynamic effects, makes the clinical significance of this interaction uncertain (see DOSAGE AND ADMINISTRATION: Concomitant Metformin and Oral Sulfonylurea Therapy in Adult Patients).
Furosemide: A single-dose, metformin-furosemide drug interaction study in healthy subjects demonstrated that pharmacokinetic parameters of both compounds were affected by co-administration. Furosemide increased the metformin plasma and blood Cmax by 22% and blood AUC by 15%, without any significant change in metformin renal clearance. When administered with metformin, the Cmax and AUC of furosemide were 31% and 12% smaller, respectively, than when administered alone, and the terminal half-life was decreased by 32%, without any significant change in furosemide renal clearance. No information is available about the interaction of metformin and furosemide when coadministered chronically.
Nifedipine: A single-dose, metformin-nifedipine drug interaction study in normal healthy volunteers demonstrated that co-administration of nifedipine increased plasma metformin Cmax and AUC by 20% and 9%, respectively, and increased the amount excreted in the urine. Tmax and half-life were unaffected. Nifedipine appears to enhance the absorption of metformin. Metformin had minimal effects on nifedipine.
Cationic drugs: Cationic drugs (e.g., amiloride, digoxin, morphine, procainamide, quinidine, quinine, ranitidine, triamterene, trimethoprim, or vancomycin) that are eliminated by renal tubular secretion theoretically have the potential for interaction with metformin by competing for common renal tubular transport systems. Such interaction between metformin and oral cimetidine has been observed in normal healthy volunteers in both single- and multiple-dose, metformin-cimetidine drug interaction studies, with a 60% increase in peak metformin plasma and whole blood concentrations and a 40% increase in plasma and whole blood metformin AUC. There was no change in elimination half-life in the single-dose study. Metformin had no effect on cimetidine pharmacokinetics. Although such interactions remain theoretical (except for cimetidine), careful patient monitoring and dose adjustment of metformin and/or the interfering drug is recommended in patients who are taking cationic medications that are excreted via the proximal renal tubular secretory system.
Other: Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control. These drugs include the thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blocking drugs, and isoniazid. When such drugs are administered to a patient receiving metformin, the patient should be closely observed for loss of blood glucose control. When such drugs are withdrawn from a patient receiving metformin, the patient should be observed closely for hypoglycemia.
In healthy volunteers, the pharmacokinetics of metformin and propranolol, and metformin and ibuprofen were not affected when coadministered in single-dose interaction studies.
Metformin is negligibly bound to plasma proteins and is, therefore, less likely to interact with highly protein-bound drugs such as salicylates, sulfonamides, chloramphenicol, and prombenecid, as compared to the sulfonylureas, which are extensively bound to serum proteins.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity studies have been performed in rats (dosing duration of 104 weeks) and mice (dosing duration of 91 weeks) at doses up to and including 900 mg/kg/day and 1500 mg/kg/day, respectively. These doses are both approximately four times the maximum recommended human daily dose of 2000 mg based on body surface area comparisons. No evidence of carcinogenicity with metformin was found in either male or female mice. Similarly, there was no tumorigenic potential observed with metformin in male rats. There was, however, an increased incidence of benign stromal uterine polyps in female rats treated with 900 mg/kg/day.
There was no evidence of a mutagenic potential of metformin in the following in vitro tests: Ames test (S. typhimurium), gene mutation test (mouse lymphoma cells), or chromosomal aberrations test (human lymphocytes). Results in the in vivo mouse micronucleus test were also negative.
Fertility of male or female rats was unaffected by metformin when administered at doses as high as 600 mg/kg/day, which is approximately three times the maximum recommended human daily dose based on body surface area comparisons.
Pregnancy
Teratogenic Effects: Pregnancy Category B
Recent information strongly suggests that abnormal blood glucose levels during pregnancy are associated with a higher incidence of congenital abnormalities. Most experts recommend that insulin be used during pregnancy to maintain blood glucose levels as close to normal as possible. Because animal reproduction studies are not always predictive of human response, metformin should not be used during pregnancy unless clearly needed.
There are no adequate and well-controlled studies in pregnant women with metformin. Metformin was not teratogenic in rats and rabbits at doses up to 600 mg/kg/day. This represents an exposure of about two and six times the maximum recommended human daily dose of 2000 mg based on body surface area comparisons for rats and rabbits, respectively. Determination of fetal concentrations demonstrated a partial placental barrier to metformin.
Nursing Mothers
Studies in lactating rats show that metformin is excreted into milk and reaches levels comparable to those in plasma. Similar studies have not been conducted in nursing mothers. Because the potential for hypoglycemia in nursing infants may exist, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. If metformin is discontinued, and if diet alone is inadequate for controlling blood glucose, insulin therapy should be considered.
Pediatric Use
The safety and effectiveness of metformin hydrochloride tablets for the treatment of type 2 diabetes have been established in pediatric patients ages 10 to 16 years (studies have not been conducted in pediatric patients below the age of 10 years). Use of metformin hydrochloride tablets in this age group is supported by evidence from adequate and well-controlled studies of metformin hydrochloride tablets in adults with additional data from a controlled clinical study in pediatric patients ages 10-16 years with type 2 diabetes, which demonstrated a similar response in glycemic control to that seen in adults. In this study, adverse effects were similar to those described in adults. A maximum daily dose of 2000 mg is recommended.
Safety and effectiveness of metformin hydrochloride extended-release tablets in pediatric patients have not been established.
Geriatric Use
Controlled clinical studies of metformin did not include sufficient numbers of elderly patients to determine whether they respond differently from younger patients, although other reported clinical experience has not identified differences in responses between the elderly and younger patients. Metformin is known to be substantially excreted by the kidney and because the risk of serious adverse reactions to the drug is greater in patients with impaired renal function, metformin should only be used in patients with normal renal function. Because aging is associated with reduced renal function, metformin should be used with caution as age increases. Care should be taken in dose selection and should be based on careful and regular monitoring of renal function. Generally, elderly patients should not be titrated to the maximum dose of metformin.
- Information for Patients
-
Package Label Principal Display
NDC: 51655-550-82
MFG: 60505-0260-2
Metformin HCL ER 500MG
120 Tablets
Rx Only
Lot#:
Exp Date:
Each tablet contains Metformin hydrochloride...500mg
Dosage: See package insert
Store between 68-77 degrees F.
Protect from moisture. Sotre in a tight, light-resistant container (See USP). Keep out of the reach of children.
Mfg by: Apotex Inc. Toronto Canada M9L 1T9
Mfg for: Apotex Corp, Weston, FL 33326
Repackaged by: Northwind Pharmaceuticals, Indianapolis, IN 46256

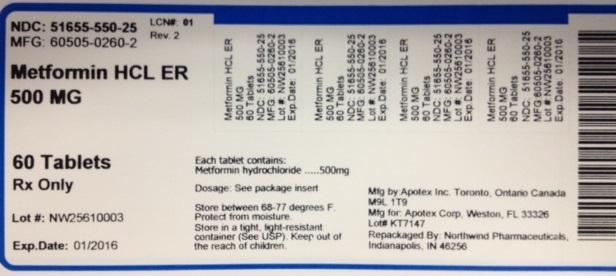
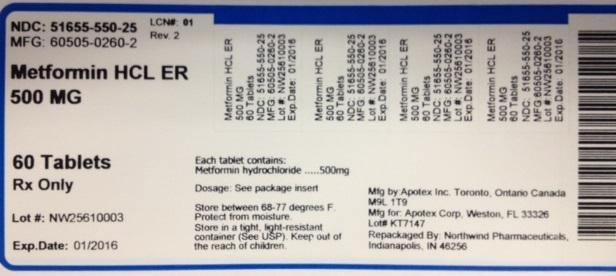
NDC: 51655-550-25
MFG: 60505-0260-2
Metformin HCL ER 500MG
60 Tablets
Rx Only
Lot#:
Exp Date:
Each tablet contains Metformin hydrochloride...500mg
Dosage: See package insert
Store between 68-77 degrees F.
Protect from moisture. Sotre in a tight, light-resistant container (See USP). Keep out of the reach of children.
Mfg by: Apotex Inc. Toronto Canada M9L 1T9
Mfg for: Apotex Corp, Weston, FL 33326
Repackaged by: Northwind Pharmaceuticals, Indianapolis, IN 46256
-
INGREDIENTS AND APPEARANCE
METFORMIN HYDROCHLORIDE ER
metformin hydrochloride tablet, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:51655-550(NDC:60505-0260) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METFORMIN HYDROCHLORIDE (UNII: 786Z46389E) (METFORMIN - UNII:9100L32L2N) METFORMIN HYDROCHLORIDE 500 mg Product Characteristics Color white Score no score Shape OVAL Size 19mm Flavor Imprint Code APO;XR500 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51655-550-82 120 in 1 BOTTLE, DISPENSING 2 NDC:51655-550-25 60 in 1 BOTTLE, DISPENSING Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076706 04/23/2014 Labeler - Northwind Pharmaceuticals (036986393) Registrant - Northwind Pharmaceuticals (036986393) Establishment Name Address ID/FEI Business Operations Northwind Pharmaceuticals 036986393 relabel(51655-550)