Label: butalbital, aspirin, caffeine and codeine phosphate- Butalbital, Aspirin, Caffeine and Codeine Phosphate capsule
-
Contains inactivated NDC Code(s)
NDC Code(s): 50564-507-01, 50564-507-05 - Packager: Jerome Stevens Pharmaceuticals
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: CIII
Drug Label Information
Updated October 25, 2007
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- N/A - Section Title Not Found In Database
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Each capsule for oral administration contains:
- codeine phosphate, USP . . . . . . . . . . . . 30 mg (1/2 gr)
- butalbital, USP . . . . . . . . . . . . . . . . . . . . . . . . . . 50 mg
- caffeine, USP . . . . . . . . . . . . . . . . . . . . . . . . . . . 40 mg
- aspirin, USP . . . . . . . . . . . . . . . . . . . . . . . . . . . 325 mg
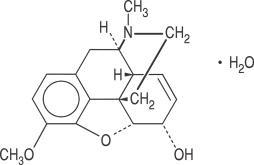
Codeine phosphate occurs as fine, white, needle-shaped crystals, or white, crystalline powder. It is affected by light. Its chemical name is 7,8-didehydro-4,5α-epoxy-3-methoxy-17-methylmorphinan-6α-ol phosphate (1:1) (salt) hemihydrate. Its molecular weight is 406.37 and its molecular formula is C18H21NO3•H3PO4•½H2O.
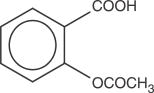
Butalbital, 5-allyl-5-isobutyl-barbituric acid, a white odorless crystalline powder, is a short- to intermediate-acting barbiturate. Its molecular weight is 224.26 and its molecular formula is C11H16N2O3.
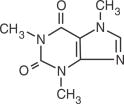
Caffeine, 1,3,7-trimethylxanthine, is a central nervous stimulant which occurs as a white powder or white glistening needles. Its molecular weight is (anhydrous) 194.19 and its molecular formula is C8H10N4O2.
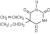
Aspirin is benzoic acid, 2-(acetyloxy)-, with a molecular formula of C9H8O4 and its molecular weight is 180.16.
Inactive Ingredients: D&C Yellow #10, D&C Yellow #10 Aluminum Lake, D&C Red #33, D&C Red #28, FD&C Blue #1, FD&C Blue #1 Aluminum Lake, FD&C Blue #2 Aluminum Lake, FD&C Blue #10 Aluminum Lake, FD&C Red #40 Aluminum Lake, gelatin, microcrystalline cellulose, pregelatinized starch, talc, titanium dioxide, stearic acid, colloidal silicon dioxide.
-
CLINICAL PHARMACOLOGY
Butalbital, Aspirin, Caffeine and Codeine Phosphate Capsules is a combination drug product intended as a treatment for tension headache.
Pharmacokinetics
Bioavailability: The bioavailability of the components of the fixed combination of butalbital, aspirin, caffeine and codeine is identical to their bioavailability when butalbital, aspirin, caffeine and codeine is administered separately in equivalent molar doses.
The behavior of the individual components is described below.
Aspirin
The systemic availability of aspirin after an oral dose is highly dependent on the dosage form, the presence of food, the gastric emptying time, gastric pH, antacids, buffering agents, and particle size. These factors affect not necessarily the extent of absorption of total salicylates but more the stability of aspirin prior to absorption.
During the absorption process and after absorption, aspirin is mainly hydrolyzed to salicylic acid and distributed to all body tissues and fluids, including fetal tissues, breast milk, and the central nervous system (CNS). Highest concentrations are found in plasma, liver, renal cortex, heart, and lung. In plasma, about 50%-80% of the salicylic acid and its metabolites are loosely bound to plasma proteins.
The clearance of total salicylates is subject to saturable kinetics; however, first-order elimination kinetics are still a good approximation for doses up to 650 mg. The plasma half-life for aspirin is about 12 minutes and for salicylic acid and/or total salicylates is about 3.0 hours.
The elimination of therapeutic doses is through the kidneys either as salicylic acid or other biotransformation products. The renal clearance is greatly augmented by an alkaline urine as is produced by concurrent administration of sodium bicarbonate or potassium citrate.
The biotransformation of aspirin occurs primarily in the hepatcytes. The major metabolites are salicyluric acid (75%), the phenolic and acyl glucuronides of salicylate (15%), and gentisic and gentisuric acid (1%). The bioavailability of the component of butalbital, aspirin, caffeine and codeine phosphate capsules is equivalent to that of a solution except for a slower rate of absorption. A peak concentration of 8.80 mcg/mL was obtained at 40 minutes after a 650 mg dose.
See OVERDOSAGE for toxicity information.
Codeine
Codeine is readily absorbed from the gastrointestinal tract. It is rapidly distributed from the intravascular spaces to the various body tissues, with preferential uptake by parenchymatous organs such as the liver, spleen, and kidney. Codeine crosses the blood-brain barrier, and is found in fetal tissue and breast milk. Codeine is not bound to plasma proteins and does not accumulate in body tissues.
The plasma half-life is about 2.9 hours. The elimination of codeine is primarily via the kidneys, and about 90% of an oral dose is excreted by the kidneys within 24 hours of dosing. The urinary secretion products consist of free and glucuronide-conjugated codeine (about 70%), free and conjugated norcodeine (about 10%), free and conjugated morphine (about 10%), normorphine (4%), and hydrocodone (1%). The remainder of the dose is excreted in the feces.
At therapeutic doses, the analgesic effect reaches a peak within 2 hours and persists between 4 and 6 hours.
The bioavailability of the codeine component is equivalent to that of a solution. Peak concentrations of 198 ng/mL were obtained at 1 hour after a 60 mg dose.
See OVERDOSAGE for toxicity information.
Butalbital
Butalbital is well absorbed from the gastrointestinal tract and is expected to distribute to most of the tissues in the body. Barbiturates, in general, may appear in breast milk and readily cross the placental barrier. They are bound to plasma and tissue proteins to a varying degree and binding increases directly as a function of lipid solubility.
Elimination of butalbital is primarily via the kidney (59%-88% of the dose) as unchanged drug or metabolites. The plasma half-life is about 35 hours. Urinary excretion products included parent drug (about 3.6% of the dose), 5-isobutyl-5-(2,3-dihydroxypropyl) barbituric acid (about 24% of the dose), 5-allyl-5(3-hydroxy-2-methyl-1-propyl) barbituric acid (about 4.8% of the dose), products with the barbituric acid ring hydrolyzed with excretion of urea (about 14% of the dose), as well as unidentified materials. Of the material excreted in the urine, 32% was conjugated.
The bioavailability of the component of butalbital, aspirin, caffeine and codeine phosphate capsules is equivalent to that of a solution except for a decrease in the rate of absorption. A peak concentration of 2020 ng/mL is obtained at about 1.5 hours after a 100 mg dose.
See OVERDOSAGE for toxicity information.
Caffeine
Like most xanthines, caffeine is rapidly absorbed and distributed in all body tissues and fluids, including the CNS, fetal tissues, and breast milk.
Caffeine is cleared rapidly through metabolism and excretion in the urine. The plasma half-life is about 3 hours. Hepatic biotransformation prior to excretion results in about equal amounts of 1-methyl-xanthine and 1-methyluric acid. Of the 70% of the dose that has been recovered in the urine, only 3% was unchanged drug.
The bioavailability of the component of butalbital, aspirin, caffeine and codeine phosphate capsules is equivalent to that of a solution except for a slightly longer time to peak. A peak concentration of 1660 ng/mL was obtained in less than an hour for an 80 mg dose.
See OVERDOSAGE for toxicity information.
-
INDICATIONS AND USAGE
Butalbital, Aspirin, Caffeine and Codeine Phosphate Capsules are indicated for the relief of the symptom complex of tension (or muscle contraction) headache.
Evidence supporting the efficacy of butalbital, aspirin, caffeine and codeine phosphate capsules is derived from 2 multi-clinic trials that compared patients with tension headache randomly assigned to 4 parallel treatments: 1) butalbital, aspirin, caffeine and codeine; 2) codeine; 3) butalbital, aspirin and caffeine; 4) placebo. Response was assessed over the course of the first 4 hours of each of 2 distinct headaches, separated by at least 24 hours. The combination product of butalbital, aspirin, caffeine and codeine proved statistically significantly superior to each of its components and to placebo on measures of pain relief.
Evidence supporting the efficacy and safety of butalbital, aspirin, caffeine and codeine in the treatment of multiple recurrent headaches is unavailable. Caution in this regard is required because codeine and butalbital are habit-forming and potentially abusable.
-
CONTRAINDICATIONS
This combination product is contraindicated under the following conditions:
- Hypersensitivity or intolerance to aspirin, caffeine, butalbital or codeine.
- Patients with hemorrhagic diathesis (e.g., hemophilia, hypoprothrombinemia, von Willebrand's disease, the thrombocytopenias, thrombasthenia and other ill-defined hereditary platelet dysfunctions, severe vitamin K deficiency and severe liver damage.)
- Patients with the syndrome of nasal polyps, angioedema and bronchospastic reactivity to aspirin or other nonsteroidal anti-inflammatory drugs. Anaphylactoid reactions have occurred in such patients.
- Peptic ulcer or other serious gastrointestinal lesions.
- Patients with porphyria.
-
WARNINGS
Therapeutic doses of aspirin can cause anaphylactic shock and other severe allergic reactions. It should be ascertained if the patient is allergic to aspirin, although a specific history of allergy may be lacking.
Significant bleeding can result from aspirin therapy in patients with peptic ulcer or other gastrointestinal lesions, and in patients with bleeding disorders.
Aspirin administered pre-operatively may prolong the bleeding time.
In the presence of head injury or other intracranial lesions, the respiratory depressant effects of codeine and other narcotics may be markedly enhanced, as well as their capacity for elevating cerebrospinal fluid pressure. Narcotics also produce other CNS depressant effects, such as drowsiness, that may further obscure the clinical course of patients with head injuries.
Codeine or other narcotics may obscure the signs on which to judge the diagnosis or clinical course of patients with acute abdominal conditions.
Butalbital and codeine are both habit-forming and potentially abusable. Consequently, the extended use of this product is not recommended.
Results from epidemiologic studies indicate an association between aspirin and Reye Syndrome. Caution should be used in administering this product to children, including teenagers, with chicken pox or flu.
-
PRECAUTIONS
General
Butalbital, aspirin, caffeine and codeine should be prescribed with caution for certain special-risk patients such as the elderly or debilitated, and those with severe impairment of renal or hepatic function, coagulation disorders, or head injuries.
Aspirin should be used with caution in patients on anticoagulant therapy and in patients with underlying hemostatic defects.
Precautions should be taken when administering salicylates to persons with known allergies. Hypersensitivity to aspirin is particularly likely in patients with nasal polyps, and relatively common in those with asthma.
Ultra-rapid Metabolizers of Codeine
Some individuals may be ultra-rapid metabolizers due to a specific CYP2D6*2x2 genotype. These individuals convert codeine into its active metabolite, morphine, more rapidly and completely than other people. This rapid conversion results in higher than expected serum morphine levels. Even at labeled dosage regiments, individuals who are ultra-rapid metabolizers may experience overdose symptoms such as extreme sleepiness, confusion or shallow breathing.
The prevalence of this CYP2D6 phenotype varies widely and has been estimated at 0.5 to 1% in Chinese and Japanese, 0.5 to 1% in Hispanics, 1-10% in Caucasians, 3% in African Americans, and 16-28% in North Africans, Ethiopians and Arabs. Data in not available for other ethnic groups.
When physicians prescribe codeine-containing drugs, they should choose the lowest effective dose for the shortest period of time and should inform their patients about these risks and the signs of morphine overdose. (See PRECAUTIONS-Nursing Mothers)
Information for Patients
Patients should be informed that this combination product contains aspirin and should not be taken by patients with an aspirin allergy.
Butalbital, aspirin, caffeine and codeine may impair the mental and/or physical abilities required for performance of potentially hazardous tasks such as driving a car or operating machinery. Such tasks should be avoided while taking this product.
Alcohol and other CNS depressants may produce an additive CNS depression when taken with this product, and should be avoided.
Codeine and butalbital may be habit-forming. Patients should take the drug only for as long as it is prescribed, in the amounts prescribed, and no more frequently than prescribed.
Caution patients that some people have a variation in a liver enzyme and change codeine into morphine more rapidly and completely than other people. These people are ultra-rapid metabolizers and are more likely to have higher-than-normal levels of morphine in their blood after taking codeine which can result in overdose symptoms such as extreme sleepiness, confusion, or shallow breathing. In most cases, it is unknown if someone is an ultra-rapid codeine metabolizer.
Nursing mothers taking codeine can also have higher morphine levels in their breast milk if they are ultra-rapid metabolizers. These higher levels of morphine in breast milk may lead to life-threatening or fatal side effects in nursing babies. Instruct nursing mothers to watch for signs of morphine toxicity in their infants including increased sleepiness (more than usual), difficulty breastfeeding, breathing difficulties, or limpness. Instruct nursing mothers to talk to the baby's doctor immediately if they notice these signs and, if they cannot reach the doctor right away, to take the baby to an emergency room or call 911 (or local emergency services).
Laboratory Tests
In patients with severe hepatic or renal disease, effects of therapy should be monitored with serial liver and/or renal function tests.
Drug Interactions
The CNS effects of butalbital may be enhanced by monoamine oxidase (MAO) inhibitors.
In patients receiving concomitant corticosteroids and chronic use of aspirin, withdrawal of corticosteroids may result in salicylism because corticosteroids enhance renal clearance of salicylates and their withdrawal is followed by return to normal rates of renal clearance.
Butalbital, aspirin, caffeine and codeine may enhance the effects of:
- Oral anticoagulants, causing bleeding by inhibiting prothrombin formation in the liver and displacing anticoagulants from plasma protein binding sites.
- Oral antidiabetic agents and insulin, causing hypoglycemia by contributing to an additive effect, if dosage of this product exceeds maximum recommended daily dosage.
- 6-mercaptopurine and methotrexate, causing bone marrow toxicity and blood dyscrasias by displacing these drugs from secondary binding sites, and, in the case of methotrexate, also reducing its excretion.
- Non-steroidal anti-inflammatory agents, increasing the risk of peptic ulceration and bleeding by contributing additive effects.
- Other narcotic analgesics, alcohol, general anesthetics, tranquilizers such as chlordiazepoxide, sedative-hypnotics, or other CNS depressants, causing increased CNS depression.
Butalbital, aspirin, caffeine and codeine may diminish the effects of:
Uricosuric agents such as probenecid and sulfinpyrazone, reducing their effectiveness in the treatment of gout. Aspirin competes with these agents for protein binding sites.
Drug/Laboratory Test Interactions
Aspirin: Aspirin may interfere with the following laboratory determinations in blood: serum amylase, fasting blood glucose, cholesterol, protein, serum glutamic-oxalacetic transaminase (SGOT), uric acid, prothrombin time and bleeding time. Aspirin may interfere with the following laboratory determinations in urine: glucose, 5-hydroxy-indoleacetic acid, Gerhardt ketone, vanillylmandelic acid (VMA), uric acid, diacetic acid, and spectrophotometric detection of barbiturates.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Adequate long-term studies have been conducted in mice and rats with aspirin, alone or in combination with other drugs, in which no evidence of carcinogenesis was seen. No adequate studies have been conducted in animals to determine whether aspirin has a potential for mutagenesis or impairment of fertility. No adequate studies have been conducted in animals to determine whether butalbital has a potential for carcinogenesis, mutagenesis, or impairment of fertility.
Pregnancy
Teratogenic Effects:
Pregnancy Category C. Animal reproduction studies have not been conducted with butalbital, aspirin, caffeine and codeine. It is also not known whether this combination product can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity and should be given to a pregnant woman only when clearly needed.
Nonteratogenic Effects:
Although Butalbital, Aspirin, Caffeine with Codeine was not implicated in the birth defect, a female infant was born with lissencephaly, pachygyria and heterotopic gray matter. The infant was born 8 weeks prematurely to a woman who had taken an average of 90 Butalbital, Aspirin, Caffeine with Codeine capsules each month from the first few days of pregnancy. The child's development was mildly delayed and from one year of age she had partial simple motor seizures.
Withdrawal seizures were reported in a two-day-old male infant whose mother had taken a butalbital-containing drug during the last 2 months of pregnancy. Butalbital was found in the infant's serum. The infant was given phenobarbital 5 mg/kg, which was tapered without further seizure or other withdrawal symptoms.
Studies of aspirin use in pregnant women have not shown that aspirin increases the risk of abnormalities when administered during the first trimester of pregnancy. In controlled studies involving 41,337 pregnant women and their offspring, there was no evidence that aspirin taken during pregnancy caused stillbirth, neonatal death, or reduced birth weight. In controlled studies of 50,282 pregnant women and their offspring, aspirin administration in moderate and heavy doses during the first four lunar months of pregnancy showed no teratogenic effect.
Reproduction studies have been performed in rabbits and rats at doses up to 150 times the human dose and have revealed no evidence of impaired fertility or harm to the fetus due to codeine.
Therapeutic doses of aspirin in pregnant women close to term may cause bleeding in mother, fetus, or neonate. During the last 6 months of pregnancy, regular use of aspirin in high doses may prolong pregnancy and delivery.
Labor and Delivery
Ingestion of aspirin prior to delivery may prolong delivery or lead to bleeding in the mother or neonate. Use of codeine during labor may lead to respiratory depression in the neonate.
Nursing Mothers
Aspirin, caffeine, barbiturates and codeine are excreted in breast milk in small amounts, but the significance of their effects on nursing infants is not known. Because of potential for serious adverse reactions in nursing infants from this product, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Codeine is secreted into human milk. In women with normal codeine metabolism (normal CYP2D6 activity), the amount of codeine secreted into human milk is low and dose-dependent. Despite the common use of codeine products to manage postpartum pain, reports of adverse events in infants are rare. However, some women are ultra-rapid metabolizers of codeine. These women achieve higher-than-expected serum levels of codeine's active metabolite, morphine, leading to higher-than-expected levels of morphine in breast milk and potentially dangerously high serum morphine levels in their breastfed infants. Therefore, maternal use of codeine can potentially lead to serious adverse reactions, including death, in nursing infants.
The prevalence of this CYP2D6 phenotype varies widely and has been estimated at 0.5 to 1% in Chinese and Japanese, 0.5 to 1% in Hispanics, 1-10% in Caucasians, 3% in African Americans, and 16-28% in North Africans, Ethiopians and Arabs. Data is not available for other ethnic groups.
The risk of infant exposure to codeine and morphine through breast milk should be weighed against the benefits of breastfeeding for both the mother and baby. Caution should be exercised when codeine is administered to a nursing woman. If a codeine containing product is selected, the lowest dose should be prescribed for the shortest period of time to achieve the desired clinical effect. Mothers using codeine should be informed about when to seek immediate medical care and how to identify the signs and symptoms of neonatal toxicity, such as drowsiness or sedation, difficulty breastfeeding, breathing difficulties, and decreased tone, in their baby. Nursing mothers who are ultra-rapid metabolizers may also experience overdose symptoms such as extreme sleepiness, confusion or shallow breathing. Presenters should closely monitor mother-infant pairs and notify treating pediatricians about the use of codeine during breastfeeding. (See PRECAUTIONS-General-Ultra-rapid Metabolizers of Codeine)
-
ADVERSE REACTIONS
Commonly Observed
The most commonly reported adverse events associated with the use of butalbital, aspirin, caffeine and codeine and not reported at an equivalent incidence by placebo-treated patients were nausea and/or abdominal pain, drowsiness, and dizziness.
Associated with Treatment Discontinuation
Of the 382 patients treated with Butalbital, Aspirin, Caffeine and Codeine in controlled clinical trials, three (0.8%) discontinued treatment because of adverse events. One patient each discontinued treatment for the following reasons: gastrointestinal upset; lightheadedness and heavy eyelids; and drowsiness and generalized tingling.
Incidence in Controlled Clinical Trials.
The following table summarizes the incidence rates of the adverse events reported by at least 1% of the Butalbital, Aspirin, Caffeine and Codeine treated patients in controlled clinical trials comparing the combination product to placebo, and provides a comparison to the incidence rates reported by the placebo-treated patients.
The prescriber should be aware that these figures cannot be used to predict the incidence of side effects in the course of usual medical practice where patient characteristics and other factors differ from those that prevailed in the clinical trials. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses, and investigators.
Adverse Events Reported by at Least 1% of Butalbital, Aspirin, Caffeine and Codeine Treated Patients During Placebo Controlled Clinical Trials Incidence Rate of Adverse Events Butalbital, Aspirin, Body System/ Caffeine and Codeine Placebo Adverse Event (N=382) N=(377) Central Nervous Drowsiness 2.4% 0.5% Dizziness/lightheadedness 2.6% 0.5% Intoxicated Feeling 1.0% 0% Gastrointestinal Nausea/Abdominal Pain 3.7% 0.8% Other Adverse Events Reported During Controlled Clinical Trials: The listing that follows represents the proportion of the 382 patients exposed to butalbital, aspirin, caffeine and codeine while participating in controlled clinical trials who reported, on at least one occasion, an adverse event of the type cited. All reported adverse events, except those already presented in the previous table, are included. It is important to emphasize that, although the adverse events reported did occur while the patient was receiving the combination product, the adverse events were not necessarily caused by butalbital, aspirin, caffeine and codeine.
Adverse events are classified by body system and frequency. ``Frequent′′ is defined as an adverse event which occurred in at least 1/100 (1%) of the patients; all adverse events listed in the previous table are frequent. ``Infrequent′′ is defined as an adverse event that occurred in less than 1/100 patients but at least 1/1000 patients. All adverse events tabulated below are classified as infrequent.
Central Nervous: headache, shaky feeling, tingling, agitation, fainting, fatigue, heavy eyelids, high energy, hot spells, numbness, and sluggishness.
Autonomic Nervous: dry mouth and hyperhidrosis.
Gastrointestinal: vomiting, difficulty swallowing, and heartburn.
Cardiovascular: tachycardia.
Musculoskeletal: leg pain and muscle fatigue.
Genitourinary: diuresis.
Miscellaneous: pruritus, fever, earache, nasal congestion, and tinnitus.
Voluntary reports of adverse drug events, temporally associated with Butalbital, Aspirin, Caffeine and Codeine, that have been received since market introduction and that were not reported in clinical trials by the patients treated with the combination product, are listed below. Many or most of these events may have no causal relationship with the drug and are listed according to body system.
Central Nervous: Abuse, addiction, anxiety, depression, disorientation, hallucination, hyperactivity, insomnia, libido decrease, nervousness, neuropathy, psychosis, sedation,sexual activity increase, slurred speech, twitching, unconsciousness, vertigo.
Autonomic Nervous: epitaxis, flushing, miosis, salivation.
Gastrointestinal: anorexia, appetite increased, constipation, diarrhea, esophagitis, gastroenteritis, gastrointestinal spasm, hiccup, mouth burning, pyloric ulcer.
Cardiovascular: chest pain, hypotensive reaction, palpitations,syncope.
Skin: erythema, erythema multiforme, exfoliative dermatitis, hives, rash, toxic epidermal necrolysis.
Urinary: kidney impairment, urinary difficulty.
Miscellaneous: allergic reaction, anaphylactic shock, cholangiocarcinoma, drug interaction with erythromycin (stomach upset), edema.
The following adverse drug events may be borne in mind as potential effects of the components of this product. Potential effects of high dosage are listed in the OVERDOSAGE section of this insert.
Aspirin: occult blood loss, hemolytic anemia, iron deficiency anemia, gastric distress, heartburn, nausea, peptic ulcer, prolonged bleeding time, acute airway obstruction, renal toxicity when taken in high doses for prolonged periods, impaired urate excretion, hepatitis.
Caffeine: cardiac stimulation, irritability, tremor, dependence, nephrotoxicity, hyperglycemia.
Codeine: nausea, vomiting, drowsiness, lightheadedness, constipation, pruritus.
-
DRUG ABUSE AND DEPENDENCE
Butalbital, Aspirin, Caffeine and Codeine Phosphate Capsules are controlled by the Drug Enforcement Administration and is classified under Schedule III.
Codeine
Codeine can produce drug dependence of the morphine type and, therefore, has the potential for being abused. Psychological dependence, physical dependence, and tolerance may develop upon repeated administration and it should be prescribed and administered with the same degree of caution appropriate to the use of other oral narcotic medications.
Butalbital
Barbiturates may be habit-forming: Tolerance, psychological dependence, and physical dependence may occur especially following prolonged use of high doses of barbiturates. The average daily dose for the barbiturate addict is usually about 1,500 mg. As tolerance to barbiturates develops, the amount needed to maintain the same level of intoxication increases; tolerance to a fatal dosage, however, does not increase more than two-fold. As this occurs, the margin between an intoxication dosage and fatal dosage becomes smaller. The lethal dose of a barbiturate is far less if alcohol is also ingested. Major withdrawal symptoms (convulsions and delirium) may occur within 16 hours and last up to 5 days after abrupt cessation of these drugs. Intensity of withdrawal symptoms gradually declines over a period of approximately 15 days. Treatment of barbiturate dependence consists of cautious and gradual withdrawal of the drug. Barbiturate-dependent patients can be withdrawn by using a number of different withdrawal regimens. One method involves initiating treatment at the patient's regular dosage level and gradually decreasing the daily dosage as tolerated by the patient.
-
OVERDOSAGE
The toxic effects of acute overdosage of Butalbital, Aspirin, Caffeine and Codeine Phosphate Capsules are attributable mainly to the barbiturate and codeine components, and, to a lesser extent, aspirin. Because toxic effects of caffeine occur in very high dosages only, the possibility of significant caffeine toxicity from this combination product is unlikely.
Signs and Symptoms.
Symptoms attributable to acute barbiturate poisoning include drowsiness, confusion, and coma; respiratory depression; hypotension; shock. Symptoms attributable to acute aspirin poisoning include hyperpnea; acid-base disturbances with development of metabolic acidosis; vomiting and abdominal pain; tinnitus, hyperthermia; hypoprothrombinemia; restlessness; delirium; convulsions. Acute caffeine poisoning may cause insomnia, restlessness, tremor, and delirium; tachycardia and extrasystoles. Symptoms of acute codeine poisoning include the triad of: pinpoint pupils, marked depression of respiration, and loss of consciousness. Convulsions may occur.
Treatment
The following paragraphs describe one approach to the treatment of overdose with this combination product. However, because strategies for the management of an overdose continually evolve, consultation with a regional Poison Control Center is strongly encouraged.
Treatment consists primarily of management of barbiturate intoxication, reversal of the effects of codeine, and the correction of the acid-base imbalance due to salicylism. Vomiting should be induced mechanically or with emetics in the conscious patient. Gastric lavage may be used if the pharyngeal and laryngeal reflexes are present and if less than 4 hours have elapsed since ingestion. A cuffed endotracheal tube should be inserted before gastric lavage of the unconscious patient and when necessary to provide assisted respiration. Diuresis, alkalinization of the urine, and correction of electrolyte disturbances should be accomplished through administration of intravenous fluids such as 1% sodium bicarbonate and 5% dextrose in water.
Meticulous attention should be given to maintaining adequate pulmonary ventilation. Correction of hypotension may require the administration of levarterenol bitartrate or phenylephrine hydrochloride by intravenous infusion. In severe cases of intoxication, peritoneal dialysis, hemodialysis, or exchange transfusion may be lifesaving. Hypoprothrombinemia should be treated with vitamin K, intravenously.
Methemoglobinemia over 30% should be treated with methylene blue by slow intravenous administration.
Naloxone, a narcotic antagonist, can reverse respiratory depression and coma associated with opioid overdose. Typically, a dose of 0.4 mg to 2 mg is given parenterally and may be repeated if an adequate response is not achieved. Since the duration of action of codeine may exceed that of the antagonist, the patient should be kept under continued surveillance and repeated doses of the antagonist should be administered as needed to maintain adequate respiration. A narcotic antagonist should not be administered in the absence of clinically significant respiratory or cardiovascular depression.
- DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
-
INGREDIENTS AND APPEARANCE
BUTALBITAL, ASPIRIN, CAFFEINE AND CODEINE PHOSPHATE
butalbital, aspirin, caffeine and codeine phosphate capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:50564-507 Route of Administration ORAL DEA Schedule CIII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Codeine Phosphate (UNII: GSL05Y1MN6) (Codeine - UNII:Q830PW7520) 30 mg Butalbital (UNII: KHS0AZ4JVK) (Butalbital - UNII:KHS0AZ4JVK) 50 mg Caffeine (UNII: 3G6A5W338E) (Caffeine - UNII:3G6A5W338E) 40 mg Aspirin (UNII: R16CO5Y76E) (Aspirin - UNII:R16CO5Y76E) 325 mg Inactive Ingredients Ingredient Name Strength D&C Yellow #10 () D&C Yellow #10 Aluminum Lake () D&C Red #33 () D&C Red #28 () FD&C Blue #2 Aluminum Lake () FD&C Blue #10 Aluminum Lake () FD&C Red #40 Aluminum Lake () Gelatin (UNII: 2G86QN327L) Microcrystalline Cellulose () Pregelatinized Starch () Talc (UNII: 7SEV7J4R1U) Titanium Dioxide (UNII: 15FIX9V2JP) Stearic Acid (UNII: 4ELV7Z65AP) Colloidal Silicon Dioxide () Product Characteristics Color YELLOW (YELLOW) , BLUE (BLUE) Score no score Shape CAPSULE (CAPSULE) Size 22mm Flavor Imprint Code JSP507 Contains Coating false Symbol false Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50564-507-01 100 in 1 BOTTLE, PLASTIC 2 NDC:50564-507-05 500 in 1 BOTTLE, PLASTIC Labeler - Jerome Stevens Pharmaceuticals