Label: INDIGESTION COMPLEX- alumina, anacardium orientale, atropinum sulphuricum, berberis vulgaris, capsicum annuum, carbo vegetabilis, ignatia amara, lycopodium clavatum, nux vomica, robinia pseud., sulphuricum acidum, valeriana officinalis liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 52731-7018-1, 52731-7018-2 - Packager: Nova Homeopathic Therapeutics, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated June 1, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Purpose:
- Usage and Dosage:
- DOSAGE & ADMINISTRATION
- Warnings:
- WARNINGS
- Active Ingredients:
- Inactive Ingredients:
- QUESTIONS
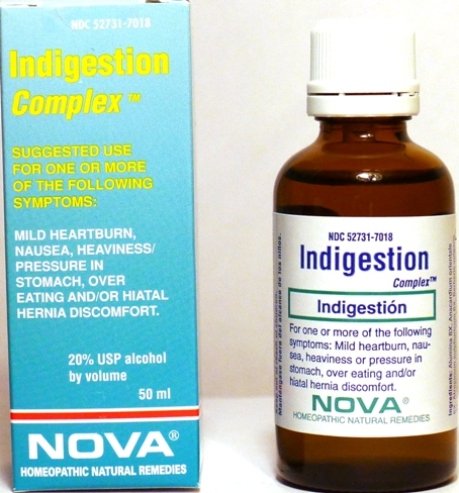
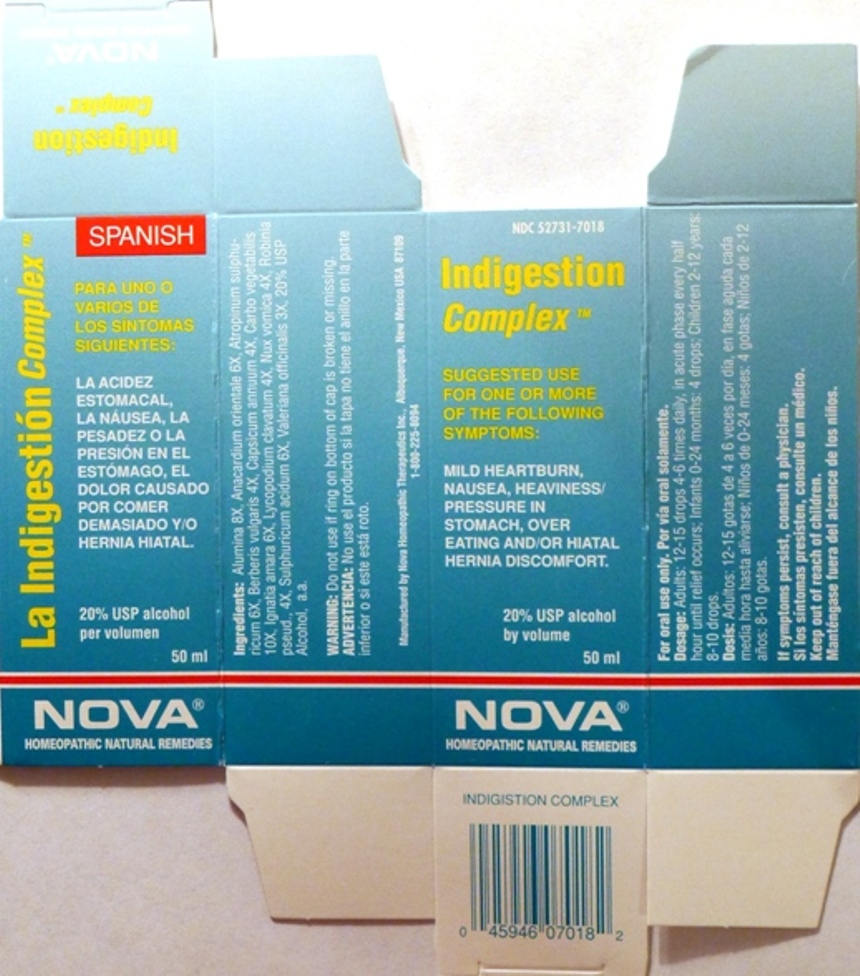
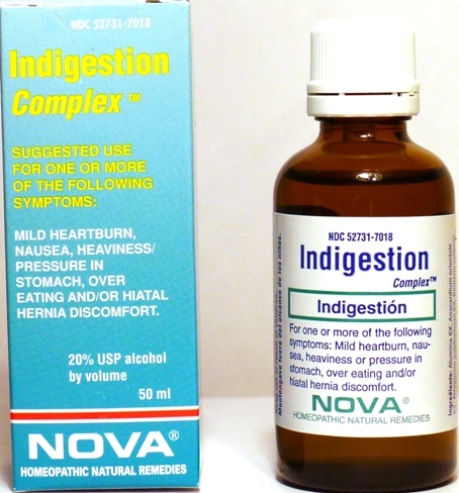
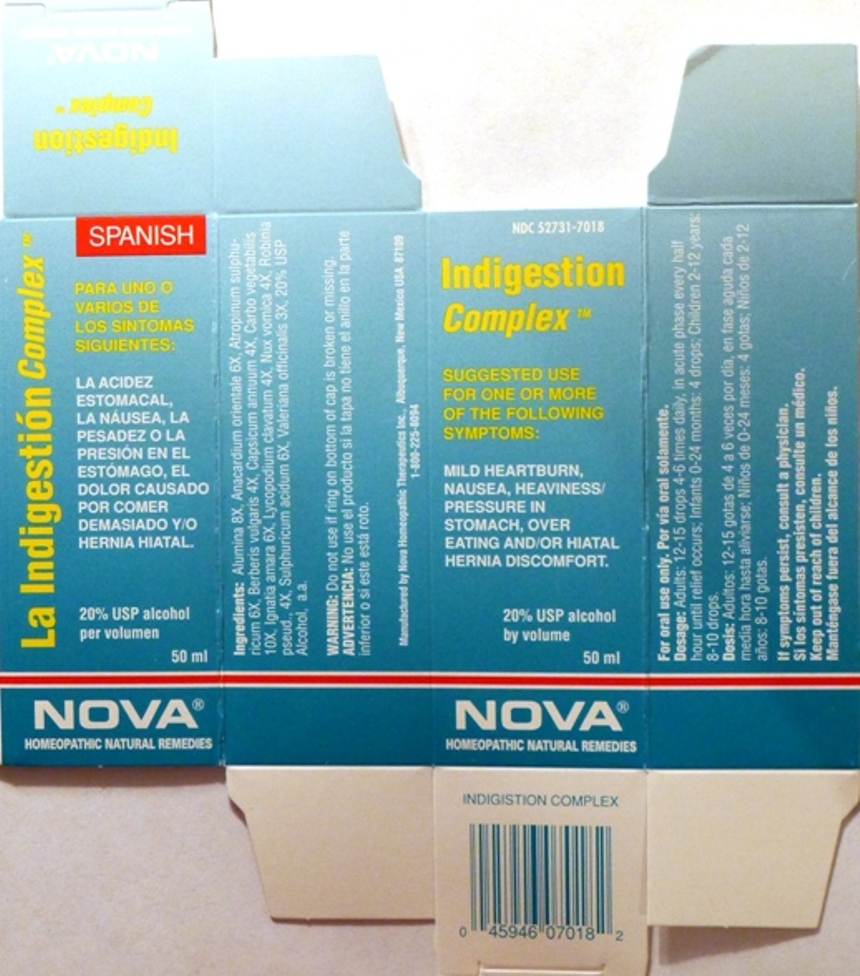
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
INDIGESTION COMPLEX
alumina, anacardium orientale, atropinum sulphuricum, berberis vulgaris, capsicum annuum, carbo vegetabilis, ignatia amara, lycopodium clavatum, nux vomica, robinia pseud., sulphuricum acidum, valeriana officinalis liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:52731-7018 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM OXIDE (UNII: LMI26O6933) (ALUMINUM OXIDE - UNII:LMI26O6933) ALUMINUM OXIDE 8 [hp_X] in 1 mL SEMECARPUS ANACARDIUM JUICE (UNII: Y0F0BU8RDU) (SEMECARPUS ANACARDIUM JUICE - UNII:Y0F0BU8RDU) SEMECARPUS ANACARDIUM JUICE 6 [hp_X] in 1 mL ATROPINE SULFATE (UNII: 03J5ZE7KA5) (SULFATE ION - UNII:7IS9N8KPMG) ATROPINE SULFATE 6 [hp_X] in 1 mL BERBERIS VULGARIS ROOT BARK (UNII: 1TH8Q20J0U) (BERBERIS VULGARIS ROOT BARK - UNII:1TH8Q20J0U) BERBERIS VULGARIS ROOT BARK 4 [hp_X] in 1 mL CAPSICUM (UNII: 00UK7646FG) (CAPSICUM - UNII:00UK7646FG) CAPSICUM 4 [hp_X] in 1 mL ACTIVATED CHARCOAL (UNII: 2P3VWU3H10) (ACTIVATED CHARCOAL - UNII:2P3VWU3H10) ACTIVATED CHARCOAL 10 [hp_X] in 1 mL STRYCHNOS IGNATII SEED (UNII: 1NM3M2487K) (STRYCHNOS IGNATII SEED - UNII:1NM3M2487K) STRYCHNOS IGNATII SEED 6 [hp_X] in 1 mL LYCOPODIUM CLAVATUM SPORE (UNII: C88X29Y479) (LYCOPODIUM CLAVATUM SPORE - UNII:C88X29Y479) LYCOPODIUM CLAVATUM SPORE 4 [hp_X] in 1 mL STRYCHNOS NUX-VOMICA SEED (UNII: 269XH13919) (STRYCHNOS NUX-VOMICA SEED - UNII:269XH13919) STRYCHNOS NUX-VOMICA SEED 4 [hp_X] in 1 mL ROBINIA PSEUDOACACIA BARK (UNII: 7TPC058OWY) (ROBINIA PSEUDOACACIA BARK - UNII:7TPC058OWY) ROBINIA PSEUDOACACIA BARK 4 [hp_X] in 1 mL SULFURIC ACID (UNII: O40UQP6WCF) (SULFURIC ACID - UNII:O40UQP6WCF) SULFURIC ACID 6 [hp_X] in 1 mL VALERIAN (UNII: JWF5YAW3QW) (VALERIAN - UNII:JWF5YAW3QW) VALERIAN 3 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:52731-7018-2 1 in 1 BOX 1 NDC:52731-7018-1 50 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 06/01/2011 Labeler - Nova Homeopathic Therapeutics, Inc. (194394540) Registrant - Nova Homeopathic Therapeutics, Inc. (194394540) Establishment Name Address ID/FEI Business Operations Nova Homeopathic Therapeutics, Inc. 194394540 manufacture, label, pack