Label: OVUGEL- triptorelin acetate gel
- NDC Code(s): 51233-101-50
- Packager: United-AH II, LLC
- Category: OTC ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated June 16, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT:
- DOSAGE FORM & STRENGTH:
- INDICATIONS FOR USE:
- DESCRIPTION:
-
WARNINGS:
WITHDRAWAL PERIOD:
No withdrawal period is required when used according to labeling.
USER SAFETY WARNINGS:
Not for Use in Humans. Keep Out of Reach of Children. The Material Safety Data Sheet (MSDS) contains more detailed occupational safety information.
ANIMAL SAFETY WARNINGS:
OvuGel should not be used in sows with obvious reproductive tract abnormalities.
- RECOMMENDATIONS FOR SAFE AND EFFECTIVE USE:
-
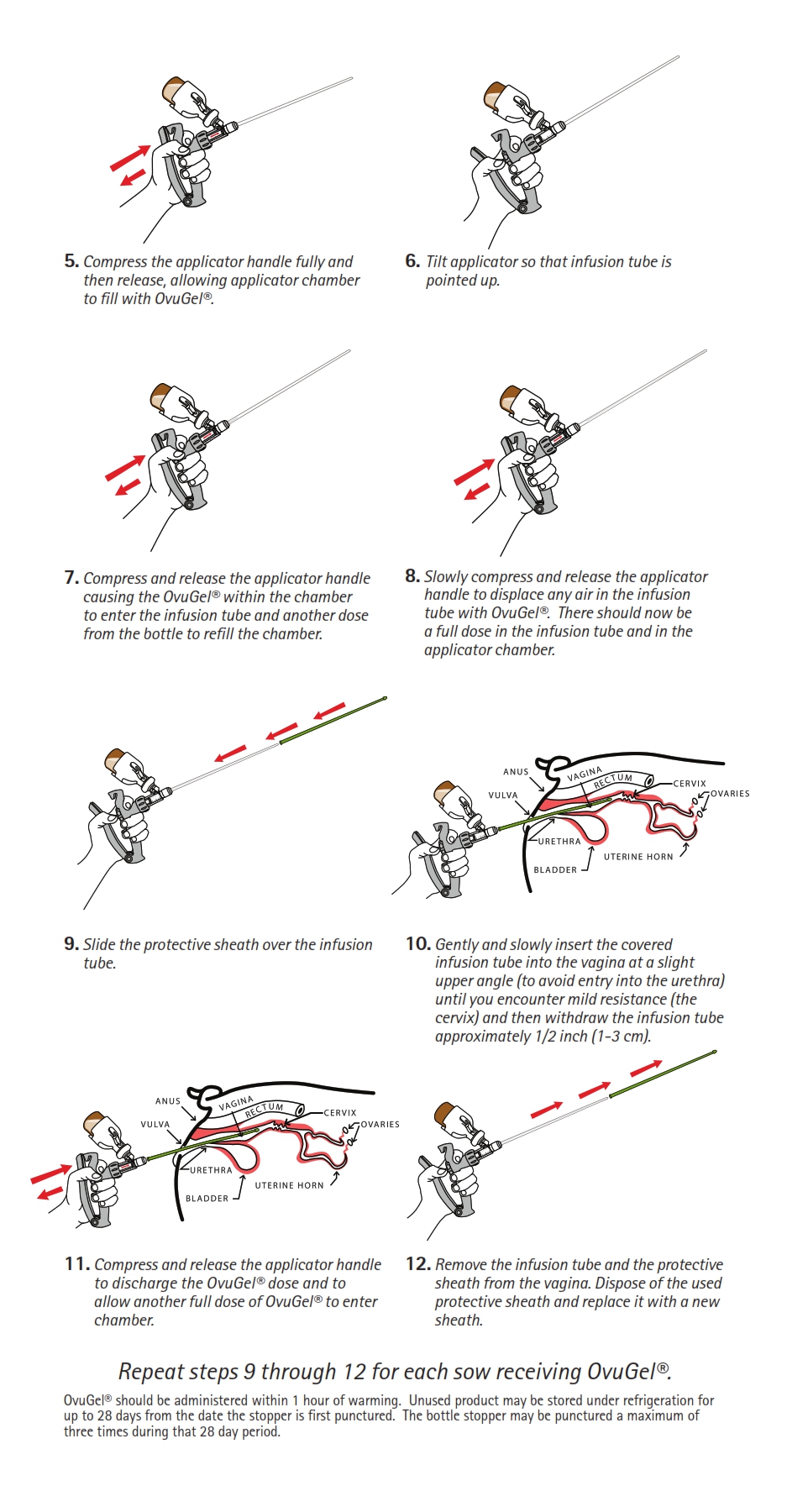
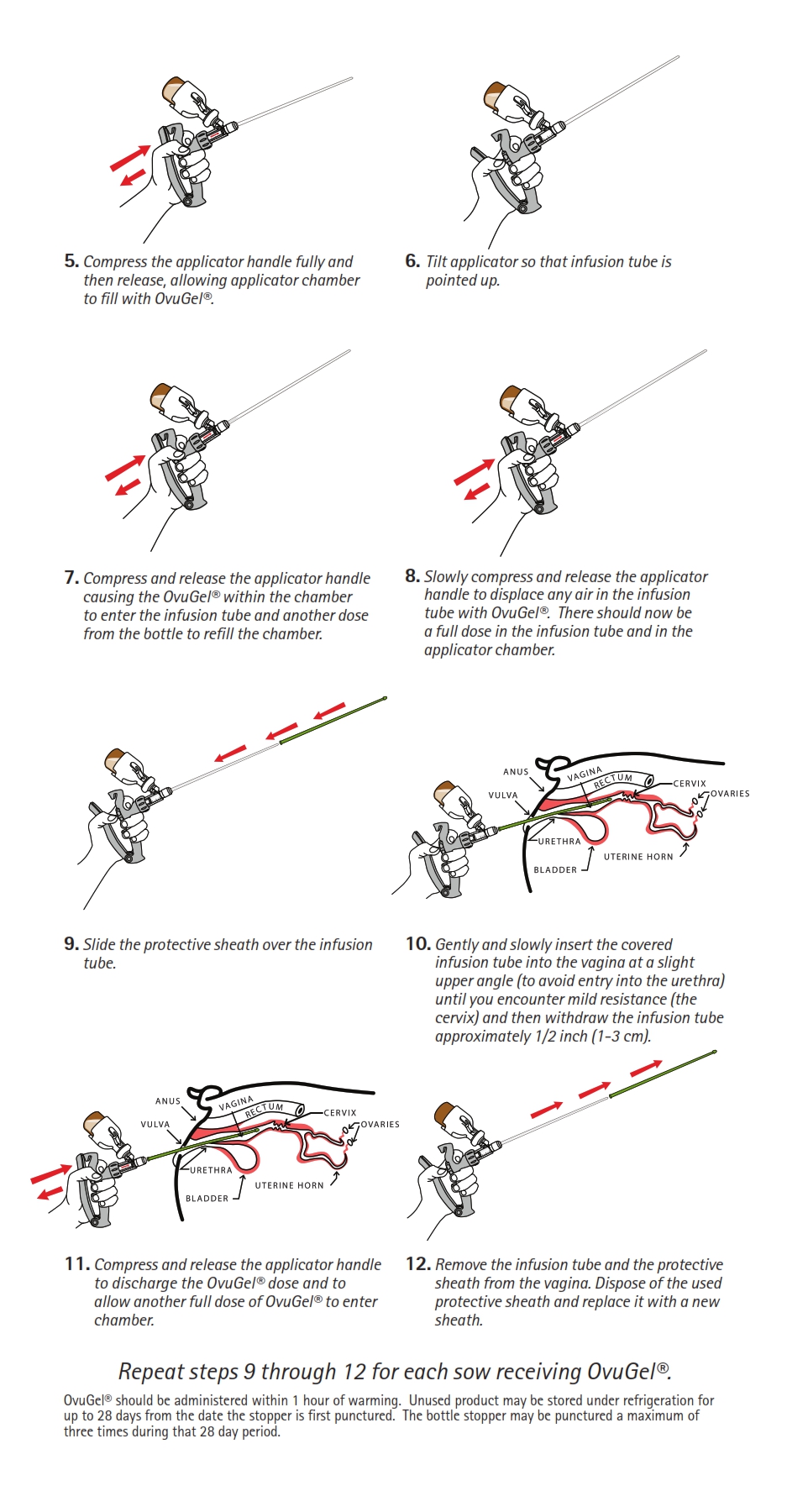
DIRECTIONS FOR USE:
OvuGel should be administered intravaginally at approximately 96 hours after weaning. The product should be warmed at room temperature for a minimum of 10 minutes prior to use. Insert the delivery tube into the vagina so that the tip rests 1/2 inch (1-3 cm) posterior to the cervix. Use a separate sheath over the delivery tube for each sow treated. Each sow should receive a single 2 mL dose of OvuGel. Sows should be inseminated 22 +/- 2 hours following administration of OvuGel using standard artificial insemination techniques. Sows should be exposed to a boar during time of insemination. (See end of package insert for more detailed directions for OvuGel administration).
-
STORAGE, HANDLING, AND DISPOSAL:
Store at refrigerator temperature 36-46°F (2-8°C). Excursions permitted to 77°F (25°C) for no more than 5 days during shipping.
OvuGel should be administered within 1 hour of warming; unused portions may be refrigerated immediately after use. The bottle stopper may be punctured a maximum of three times. Discard 28 days after the stopper is first punctured. - HOW SUPPLIED:
- QUESTIONS/COMMENTS:
- BOTTLE LABEL:
- PACKAGE INSERT (FRONT):
- PACKAGE INSERT (BACK):
- CARTON:
- SHIPPING LABEL:
-
INGREDIENTS AND APPEARANCE
OVUGEL
triptorelin acetate gelProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:51233-101 Route of Administration VAGINAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Triptorelin Acetate (UNII: 43OFW291R9) (Triptorelin - UNII:9081Y98W2V) Triptorelin 100 ug in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51233-101-50 52.5 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141339 02/08/2013 Labeler - United-AH II, LLC (783511723)