Label: CLARINS SUPER RESTORATIVE DAY ILLUMINATING LIFTING REPLENISHING BROAD SPECTRUM - SPF 20 - SUNSCREEN- avobenzone, homosalate, octocrylene cream
- NDC Code(s): 58668-0961-1
- Packager: Laboratoires Clarins S.A.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 8, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- CLARINS Super Restorative Day Illuminating lifting replenishing cream BROAD SPECTRUM - SPF 20 - Sunscreen
- Active ingredients
- Purpose
- Uses:
- Warnings:
-
Directions:
- apply liberally 15 minutes before sun exposure
- apply to all skin exposed to the sun
- children under 6 month: Ask a doctor
- Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease the risk, regularly use a sunscreen with broad spectrum SPF of 15 or higher and other sun protection measures including: Sun Protection Measures.
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- reapply at least every 2 hours
- use a water resistant sunscreen if swimming or sweating
-
Inactive ingredients:
AQUA/WATER/EAU - GLYCERIN - BUTYLOCTYL SALICYLATE - C12-15 ALKYL BENZOATE - ALCOHOL - DIMETHICONE - CETEARYL ALCOHOL - OLUS OIL/VEGETABLE OIL/HUILE VEGETALE - PENTYLENE GLYCOL - BUTYROSPERMUM PARKII (SHEA) BUTTER - POTASSIUM CETYL PHOSPHATE - TROMETHAMINE - CETEARYL GLUCOSIDE - PARFUM/FRAGRANCE - ACRYLATES/C10-30 ALKYL ACRYLATE CROSSPOLYMER - ASCORBYL GLUCOSIDE - SUCROSE PALMITATE - TOCOPHERYL ACETATE - HYDROGENATED VEGETABLE OIL - ETHYLHEXYLGLYCERIN - XANTHAN GUM - BUTYLENE GLYCOL - GLYCERYL LINOLEATE - SODIUM HYALURONATE - DISODIUM EDTA - CANDELILLA CERA/EUPHORBIA CERIFERA (CANDELILLA) WAX/CIRE DE CANDELILLA - TOCOPHEROL - MICA - CI 77120/BARIUM SULFATE - CI 77891/TITANIUM DIOXIDE - HARUNGANA MADAGASCARIENSIS EXTRACT - CITRIC ACID - POTASSIUM SORBATE - PROPANEDIOL - OLEA EUROPAEA (OLIVE) FRUIT EXTRACT - PECTIN - THERMUS THERMOPHILUS FERMENT - HELIANTHUS ANNUUS (SUNFLOWER) SEED OIL - GUAR HYDROXYPROPYLTRIMONIUM CHLORIDE - PHENOXYETHANOL - LAPSANA COMMUNIS FLOWER/LEAF/STEM EXTRACT - CAMELLIA SINENSIS LEAF EXTRACT - RHODIOLA ROSEA ROOT EXTRACT - CI 14700/RED 4 - SODIUM LAURYL SULFATE - ACETYL TETRAPEPTIDE-2 - DEXTRAN. [S2628A]
- Other information:
- Questions or comments?
-
"CLARINS: effective, serious treatments, ensuring maximum results with the highest quality ingredients." Jacques Courtin Clarins
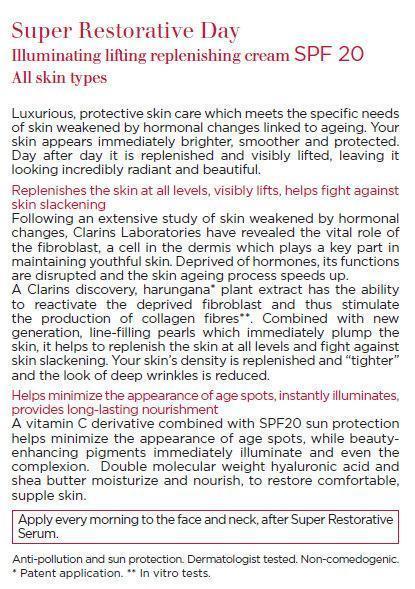
Super Restorative Day SPF 20 All skin types Protective skin care which meets the specific needs of skin weakened by hormonal changes linked to ageing. For skin that looks incredibly radiant and beautiful. • Replenishes the skin at all levels, visibly lifts, helps fight against skin slackening A powerful plant extract of harungana* helps to replenish the skin and fight against skin slackening**. The look of wrinkles is reduced and the skin appears toned and lifted. • Helps minimize the appearance of age spots, instantly illuminates, provides long-lasting nourishment A vitamin C derivative and sun filters help encourage a more even complexion and beauty-enhancing pigments revive complexion radiance. Hyaluronic acid and shea butter provide moisturization and comfort. Anti-pollution and sun protection. Dermatologist tested. Noncomedogenic. *Patent application (FR). **In vitro tests.
CLARINS PARIS - 92200 Neuilly - 95300 Pontoise - France Made in France - Brevets (FR, WO) et marques déposés Patent applications (FR, WO) www.clarins.com CLARINS-PARIS
18 M ref. 10961 9
- PRINCIPAL DISPLAY PANEL
- CLARINS Super Restorative Day Illuminating lifting replenishing cream BROAD SPECTRUM - SPF 20 - Sunscreen 50ml (58668-0961-1)
-
INGREDIENTS AND APPEARANCE
CLARINS SUPER RESTORATIVE DAY ILLUMINATING LIFTING REPLENISHING BROAD SPECTRUM - SPF 20 - SUNSCREEN
avobenzone, homosalate, octocrylene creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58668-0961 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 100 mg in 1 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 27 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) ALCOHOL (UNII: 3K9958V90M) DIMETHICONE (UNII: 92RU3N3Y1O) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) PENTYLENE GLYCOL (UNII: 50C1307PZG) SHEA BUTTER (UNII: K49155WL9Y) POTASSIUM CETYL PHOSPHATE (UNII: 03KCY6P7UT) TROMETHAMINE (UNII: 023C2WHX2V) CETEARYL GLUCOSIDE (UNII: 09FUA47KNA) ASCORBYL GLUCOSIDE (UNII: 2V52R0NHXW) SUCROSE PALMITATE (UNII: 3OSQ643ZK5) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) XANTHAN GUM (UNII: TTV12P4NEE) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) GLYCERYL MONOLINOLEATE (UNII: 4763AXI84L) HYALURONATE SODIUM (UNII: YSE9PPT4TH) EDETATE DISODIUM (UNII: 7FLD91C86K) CANDELILLA WAX (UNII: WL0328HX19) TOCOPHEROL (UNII: R0ZB2556P8) MICA (UNII: V8A1AW0880) BARIUM SULFATE (UNII: 25BB7EKE2E) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) PROPANEDIOL (UNII: 5965N8W85T) BLACK OLIVE (UNII: 2M6QWV94OC) PECTIN (UNII: 89NA02M4RX) THERMUS THERMOPHILUS LYSATE (UNII: 775R692494) SUNFLOWER OIL (UNII: 3W1JG795YI) PHENOXYETHANOL (UNII: HIE492ZZ3T) LAPSANA COMMUNIS FLOWERING TOP (UNII: T9B77O7PZA) GREEN TEA LEAF (UNII: W2ZU1RY8B0) SEDUM ROSEUM ROOT (UNII: 3S5ITS5ULN) FD&C RED NO. 4 (UNII: X3W0AM1JLX) SODIUM LAURYL SULFATE (UNII: 368GB5141J) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58668-0961-1 1 in 1 CARTON 07/29/2013 1 50 mL in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 07/29/2013 Labeler - Laboratoires Clarins S.A. (266317555) Establishment Name Address ID/FEI Business Operations Laboratoires Clarins S.A. 266317555 manufacture(58668-0961)