Label: DOBUTAMINE HYDROCHLORIDE injection
- NDC Code(s): 51662-1566-1
- Packager: HF Acquisition Co LLC, DBA HealthFirst
- This is a repackaged label.
- Source NDC Code(s): 0338-1073
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated January 30, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Dobutamine Hydrochloride in 5% Dextrose Injection is a sterile, nonpyrogenic solution of Dobutamine Hydrochloride, USP and Dextrose, USP in Water for Injection, USP. Dobutamine hydrochloride is chemically designated as (±)-4-[2-[[3-(p-hydroxyphenyl)-1-methylpropyl]amino]ethyl]-pyrocatechol hydrochloride. It is a synthetic catecholamine. Dextrose Hydrous, USP is chemically designated as D-Glucopyranose monohydrate. Structural formulas are shown below:
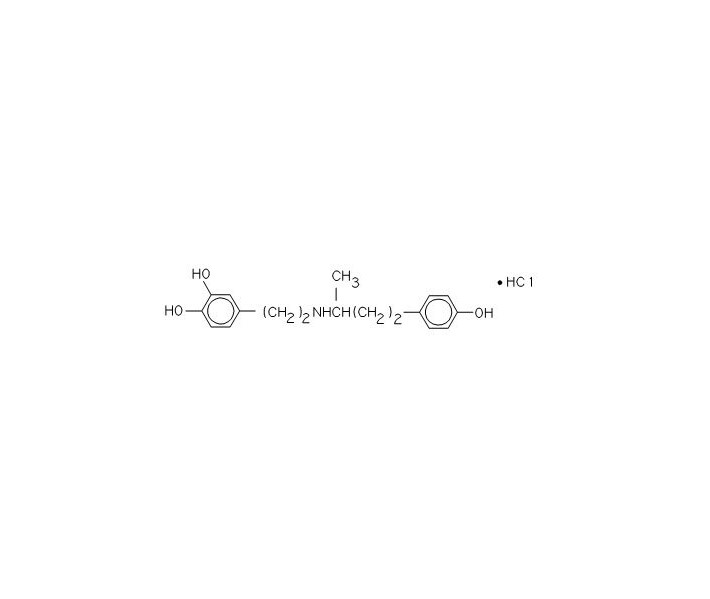 Dobutamine Hydrochloride, USP
Dobutamine Hydrochloride, USP
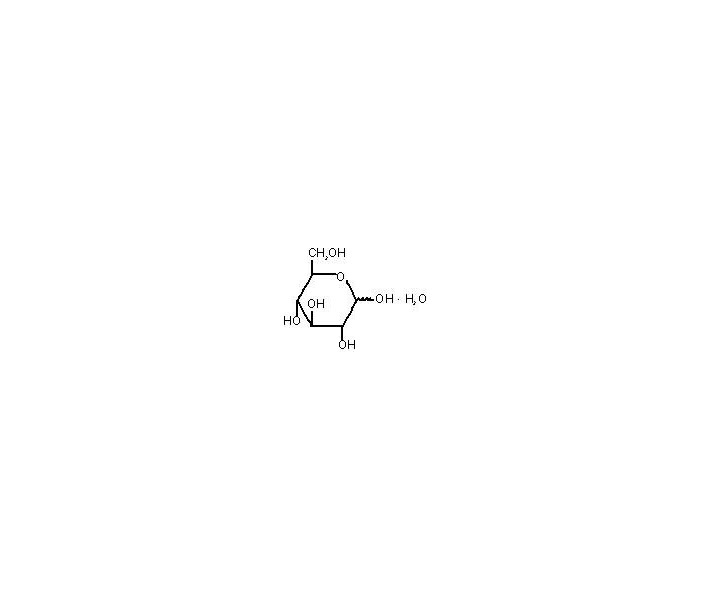
(D-Glucopyranose monohydrate) Dextrose Hydrous, USP
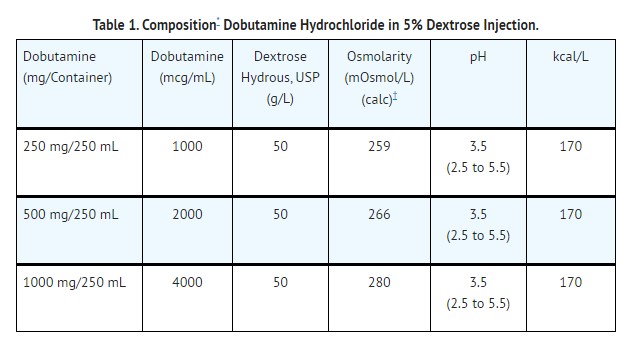
Dobutamine Hydrochloride in 5% Dextrose Injection is intended for intravenous use only. It contains no antimicrobial agents. The pH is adjusted with sodium hydroxide and/or hydrochloric acid. Sodium bisulfite is added as a stabilizer. The solution is intended for single use only. When smaller doses are required, the unused portion should be discarded. Composition, osmolarity, pH and caloric content are given in Table 1.
*
Approximately 5 mEq/L sodium bisulfite is added as a stabilizer.
†
Normal physiologic osmolarity range is approximately 280 to 310 mOsmol/L. Administration of substantially hypertonic solutions (≥ 600 mOsmol/L) may cause vein damage.This VIAFLEX PLUS plastic container is fabricated from a specially formulated polyvinyl chloride (PL 2207 Plastic). VIAFLEX containers, including VIAFLEX PLUS containers, are made of flexible plastic and are for parenteral use. VIAFLEX PLUS on the container indicates the presence of a drug additive in a drug vehicle. The amount of water that can permeate from inside the container into the overwrap is insufficient to affect the solution significantly. Solutions in contact with the plastic container can leach out certain of its chemical components in very small amounts within the expiration period, e.g., di-2-ethylhexyl phthalate (DEHP), up to 5 parts per million; however, the safety of the plastic has been confirmed in tests in animals according to USP biological tests for plastic containers as well as by tissue culture toxicity studies.
-
CLINICAL PHARMACOLOGY
Dobutamine hydrochloride is a direct-acting inotropic agent whose primary activity results from stimulation of the beta-receptors of the heart while producing comparatively mild chronotropic, hypertensive, arrhythmogenic, and vasodilative effects. It does not cause the release of endogenous norepinephrine, as does dopamine. In animal studies, dobutamine produces less increase in heart rate and less decrease in peripheral vascular resistance for a given inotropic effect than does isoproterenol.
In patients with depressed cardiac function, both dobutamine and isoproterenol increase the cardiac output to a similar degree. In the case of dobutamine, this increase is usually not accompanied by marked increases in heart rate (although tachycardia is occasionally observed), and the cardiac stroke volume is usually increased. In contrast, isoproterenol increases the cardiac index primarily by increasing the heart rate while stroke volume changes little or declines.
Facilitation of atrioventricular conduction has been observed in human electrophysiologic studies and in patients with atrial fibrillation.
Systemic vascular resistance is usually decreased with administration of dobutamine. Occasionally, minimum vasoconstriction has been observed.
Most clinical experience with dobutamine is short-term - not more than several hours in duration. In the limited number of patients who were studied for 24, 48, and 72 hours, a persistent increase in cardiac output occurred in some, whereas output returned toward baseline values in others.
The onset of action of dobutamine is within one to two minutes; however, as much as ten minutes may be required to obtain the peak effect of a particular infusion rate.
The plasma half-life of dobutamine in humans is two minutes. The principal routes of metabolism are methylation of the catechol and conjugation. In human urine, the major excretion products are the conjugates of dobutamine and 3-O-methyl dobutamine. The 3-O-methyl derivative of dobutamine is inactive.
Alteration of synaptic concentrations of catecholamines with either reserpine or tricyclic antidepressants does not alter the actions of dobutamine in animals, which indicates that the actions of dobutamine are not dependent on presynaptic mechanisms.
The effective infusion rate of dobutamine varies widely from patient to patient, and titration is always necessary (see DOSAGE AND ADMINISTRATION). At least in pediatric patients, dobutamine-induced increases in cardiac output and systemic pressure are generally seen, in any given patient, at lower infusion rates than those that cause substantial tachycardia (see PRECAUTIONS, PEDIATRIC USE).
Dextrose provides a source of calories. Dextrose is readily metabolized, may decrease losses of body protein and nitrogen, promotes glycogen deposition and decreases or prevents ketosis if sufficient doses are provided.
-
INDICATIONS & USAGE
Dobutamine Hydrochloride in 5% Dextrose Injection is indicated when parenteral therapy is necessary for inotropic support in the short-term treatment of patients with cardiac decompensation due to depressed contractility resulting either from organic heart disease or from cardiac surgical procedures. Experience with intravenous dobutamine in controlled trials does not extend beyond 48 hours of repeated boluses and/or continuous infusions.
Whether given orally, continuously intravenously, or intermittently intravenously, neither dobutamine nor any other cyclic-AMP-dependent inotrope has been shown in controlled trials to be safe or effective in the long-term treatment of congestive heart failure. In controlled trials of chronic oral therapy with various such agents, symptoms were not consistently alleviated, and the cyclic-AMP-dependent inotropes were consistently associated with increased risks of hospitalization and death. Patients with NYHA Class IV symptoms appeared to be at particular risk.
-
CONTRAINDICATIONS
Dobutamine Hydrochloride in 5% Dextrose Injection is contraindicated in patients with idiopathic hypertrophic subaortic stenosis and in patients who have shown previous manifestations of hypersensitivity to dobutamine.
Solutions containing dextrose may be contraindicated in patients with known allergy to corn or corn products.
-
WARNINGS
Increase in Heart Rate or Blood Pressure
Dobutamine Hydrochloride in 5% Dextrose Injection may cause a marked increase in heart rate or blood pressure, especially systolic pressure. Approximately 10% of adult patients in clinical studies have had rate increases of 30 beats/minute or more, and about 7.5% have had a 50-mm Hg or greater increase in systolic pressure. Usually, reduction of dosage reverses these effects. Because dobutamine facilitates atrioventricular conduction, patients with atrial fibrillation are at risk of developing rapid ventricular response. Patients with preexisting hypertension appear to face an increased risk of developing an exaggerated pressor response. In patients who have atrial fibrillation with rapid ventricular response, a digitalis preparation should be used prior to institution of therapy with Dobutamine in D5W.
Ectopic Activity
Dobutamine Hydrochloride in 5% Dextrose Injection may precipitate or exacerbate ventricular ectopic activity, but it rarely has caused ventricular tachycardia.
Hypersensitivity
Reactions suggestive of hypersensitivity associated with administration of dobutamine including skin rash, fever, eosinophilia, and bronchospasm, have been reported occasionally.
Dobutamine Hydrochloride in 5% Dextrose Injection contains sodium bisulfite, a sulfite that may cause allergic-type reactions, including anaphylactic symptoms and life-threatening or less severe asthmatic episodes, in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
Solutions containing dextrose should not be administered through the same administration set as blood, as this may result in pseudoagglutination or hemolysis.
The intravenous administration of solutions may cause fluid overloading resulting in dilution of serum electrolyte concentrations, overhydration, congested states or pulmonary edema. The risk of dilutional states is inversely proportional to the electrolyte concentrations of the injections. The risk of solute overload causing congested states with peripheral and pulmonary edema is directly proportional to the electrolyte concentration of the injections.
Excess administration of potassium-free solutions may result in significant hypokalemia.
-
PRECAUTIONS
General
During the administration of Dobutamine Hydrochloride in 5% Dextrose Injection, as with any adrenergic agent, ECG and blood pressure should be continuously monitored. In addition, pulmonary wedge pressure and cardiac output should be monitored whenever possible to aid in the safe and effective infusion of dobutamine.
Hypovolemia should be corrected with suitable volume expanders before treatment with dobutamine is instituted.
Animal studies indicate that dobutamine may be ineffective if the patient has recently received a beta-blocking drug. In such a case, the peripheral vascular resistance may increase.
No improvement may be observed in the presence of marked mechanical obstruction, such as severe valvular aortic stenosis.
Solutions containing dextrose should be used with caution in patients with known subclinical or overt diabetes mellitus.
Do not administer unless solution is clear and seal is intact.
If administration is controlled by a pumping device, care must be taken to discontinue pumping action before the container runs dry or air embolism may result.
Usage Following Acute Myocardial Infarction
Clinical experience with dobutamine following myocardial infarction has been insufficient to establish the safety of the drug for this use. There is concern that any agent that increases contractile force and heart rate may increase the size of an infarction by intensifying ischemia, but it is not known whether dobutamine does so.
Drug Interactions
There was no evidence of drug interactions in clinical studies in which dobutamine was administered concurrently with other drugs, including digitalis preparations, furosemide, spironolactone, lidocaine, glyceryl trinitrate, isosorbide dinitrate, morphine, atropine, heparin, protamine, potassium chloride, folic acid, and acetaminophen. Preliminary studies indicate that the concomitant use of dobutamine and nitroprusside results in a higher cardiac output and, usually, a lower pulmonary wedge pressure than when either drug is used alone.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies to evaluate the carcinogenic or mutagenic potential of dobutamine or the potential of the drug to affect fertility adversely have not been performed.
Pregnancy
Reproduction studies performed in rats and rabbits have revealed no evidence of harm to the fetus due to dobutamine. The drug, however, has not been administered to pregnant women and should be used only when the expected benefits clearly outweigh the potential risks to the fetus.
Pediatric Use
Dobutamine has been shown to increase cardiac output and systemic pressure in pediatric patients of every age group. In premature neonates, however, dobutamine is less effective than dopamine in raising systemic blood pressure without causing undue tachycardia, and dobutamine has not been shown to provide any added benefit when given to such infants already receiving optimal infusions of dopamine.
Geriatric Use
Clinical studies of dobutamine injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or drug therapy.
-
ADVERSE REACTIONS
Increased Heart Rate, Blood Pressure, and Ventricular Ectopic Activity
A 10 to 20-mm Hg increase in systolic blood pressure and an increase in heart rate of 5 to 15 beats/minute have been noted in most patients (see WARNINGS regarding exaggerated chronotropic and pressor effects). Approximately 5% of adult patients have had increased premature ventricular beats during infusions. These effects are dose related.
Hypotension
Precipitous decreases in blood pressure have occasionally been described in association with dobutamine therapy. Decreasing the dose or discontinuing the infusion typically results in rapid return of blood pressure to baseline values. In rare cases, however, intervention may be required and reversibility may not be immediate.
Stress Cardiomyopathy
Stress cardiomyopathy has been reported with dobutamine in association with cardiac stress testing.
Reactions at Sites of Intravenous Infusion
Phlebitis has occasionally been reported. Local inflammatory changes have been described following inadvertent infiltration.
Miscellaneous Uncommon Effects
The following adverse effects have been reported in 1% to 3% of adult patients: nausea, headache, anginal pain, nonspecific chest pain, palpitations, and shortness of breath.
Administration of dobutamine, like other catecholamines, has been associated with decreases in serum potassium concentrations, rarely to hypokalemic values.
-
OVERDOSAGE
Overdoses of dobutamine have been reported rarely. The following is provided to serve as a guide if such an overdose is encountered.
Signs and Symptoms
Toxicity from dobutamine is usually due to excessive cardiac beta-receptor stimulation. The duration of action of dobutamine is generally short (T1/2 = two minutes) because it is rapidly metabolized by catechol-O-methyltransferase. The symptoms of toxicity may include anorexia, nausea, vomiting, tremor, anxiety, palpitations, headache, shortness of breath, and anginal and nonspecific chest pain. The positive inotropic and chronotropic effects of dobutamine on the myocardium may cause hypertension, tachyarrhythmias, myocardial ischemia, and ventricular fibrillation. Hypotension may result from vasodilation.
If the product is ingested, unpredictable absorption may occur from the mouth and the gastrointestinal tract.
Treatment
To obtain up-to-date information about the treatment of overdose, a good resource is your certified Regional Poison Control Center. Telephone numbers of certified poison control centers are listed in the Physicians’ Desk Reference (PDR). In managing overdosage, consider the possibility of multiple drug overdoses, interaction among drugs, and unusual drug kinetics in your patient.
The initial actions to be taken in a dobutamine overdose are discontinuing administration, establishing an airway, and ensuring oxygenation and ventilation. Resuscitative measures should be initiated promptly. Severe ventricular tachyarrhythmias may be successfully treated with propranolol or lidocaine. Hypertension usually responds to a reduction in dose or discontinuation of therapy.
Protect the patient’s airway and support ventilation and perfusion. If needed, meticulously monitor and maintain, within acceptable limits, the patient’s vital signs, blood gases, serum electrolytes, etc. Absorption of drugs from the gastrointestinal tract may be decreased by giving activated charcoal, which, in many cases, is more effective than emesis or lavage; consider charcoal instead of or in addition to gastric emptying. Repeated doses of charcoal over time may hasten elimination of some drugs that have been absorbed. Safeguard the patient’s airway when employing gastric emptying or charcoal.
Forced diuresis, peritoneal dialysis, hemodialysis, or charcoal hemoperfusion have not been established as beneficial for an overdose of dobutamine.
-
DOSAGE & ADMINISTRATION
Recommended Dosage
Dobutamine Hydrochloride in 5% Dextrose Injection is administered intravenously through a suitable intravenous catheter or needle. A calibrated electronic infusion device is recommended for controlling the rate of flow in mL/hour or drops/minute.
Infusion of dobutamine should be started at a low rate (0.5-1.0 mcg/kg/min) and titrated at intervals of a few minutes, guided by the patient’s response, including systemic blood pressure, urine flow, frequency of ectopic activity, heart rate, and (whenever possible) measurements of cardiac output, central venous pressure, and/or pulmonary capillary wedge pressure. In reported trials, the optimal infusion rates have varied from patient to patient, usually 2-20 mcg/kg/min but sometimes slightly outside of this range. On rare occasions, infusion rates up to 40 mcg/kg/min have been required to obtain the desired effect.
Rates of infusion in mL/hour for dobutamine hydrochloride concentrations of 1,000, 2,000 and 4,000 mcg/mL are in Table 2.
This container system may be inappropriate for the dosage requirements of pediatric patients under 30 kg. Other dosage forms may be more appropriate.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Dobutamine Hydrochloride in 5% Dextrose Injection solutions may exhibit a pink color that, if present, will increase with time. This color change is due to slight oxidation of the drug, but there is no significant loss of potency.
The rate of administration and the duration of therapy should be adjusted according to the patient’s response, as determined by heart rate, presence of ectopic activity, blood pressure, urine flow, and, whenever possible, measurement of central venous or pulmonary wedge pressure and cardiac output.
Do not add supplementary medications to Dobutamine Hydrochloride in 5% Dextrose Injection. Do not administer Dobutamine Hydrochloride in 5% Dextrose Injection simultaneously with solutions containing sodium bicarbonate or strong alkaline solutions.
-
HOW SUPPLIED
Dobutamine Hydrochloride in 5% Dextrose Injection in VIAFLEX PLUS plastic containers is available as follows:

Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. Protect from freezing. It is recommended the product be stored at room temperature (25°C); brief exposure up to 40°C does not adversely affect the product.
Directions for use of VIAFLEX PLUS Plastic Container
Do not remove unit from overwrap until ready for use.
To open
Tear overwrap down side at notch and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. Check for minute leaks by squeezing inner bag firmly. If leaks are found, discard solution as sterility may be impaired.
Preparation for Administration
Visually inspect the container. If the administration port protector is damaged, detached, or not present, discard container as solution path sterility may be impaired.
Caution: Do not use plastic containers in series connections. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed.
1.
Suspend container from eyelet support.
2.
Remove plastic protector from administration port at bottom of container.
3.
Attach administration set. Refer to complete directions accompanying set.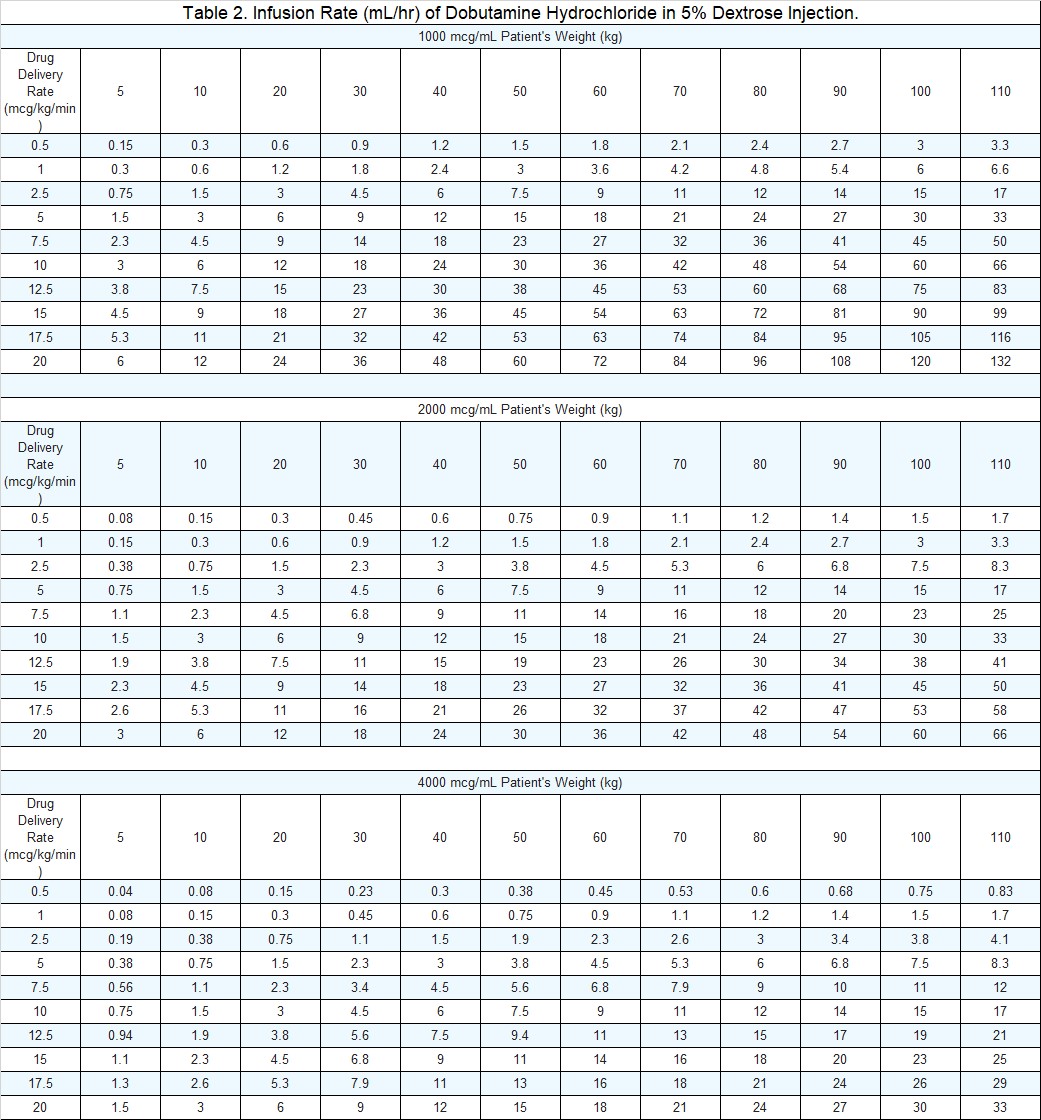
Baxter, Viaflex and Viaflex Plus are trademarks of Baxter International Inc.
Baxter Healthcare Corporation
Deerfield, IL 60015 USA
Printed in USA07-19-00-0594
Rev. March 2019 - PRINCIPAL DISPLAY PANEL - 51662-1567-1 BAG AND SERIALIZED BAG LABELING
-
INGREDIENTS AND APPEARANCE
DOBUTAMINE HYDROCHLORIDE
dobutamine hydrochloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:51662-1566(NDC:0338-1073) Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOBUTAMINE HYDROCHLORIDE (UNII: 0WR771DJXV) (DOBUTAMINE - UNII:3S12J47372) DOBUTAMINE 100 mg in 100 mL Inactive Ingredients Ingredient Name Strength HYDROCHLORIC ACID (UNII: QTT17582CB) DEXTROSE MONOHYDRATE (UNII: LX22YL083G) 5 g in 100 mL SODIUM BISULFITE (UNII: TZX5469Z6I) 0.5 meq in 100 mL SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51662-1566-1 250 mL in 1 BAG; Type 0: Not a Combination Product 07/10/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020255 07/10/2021 Labeler - HF Acquisition Co LLC, DBA HealthFirst (045657305) Registrant - HF Acquisition Co LLC, DBA HealthFirst (045657305) Establishment Name Address ID/FEI Business Operations HF Acquisition Co LLC, DBA HealthFirst 045657305 relabel(51662-1566)




