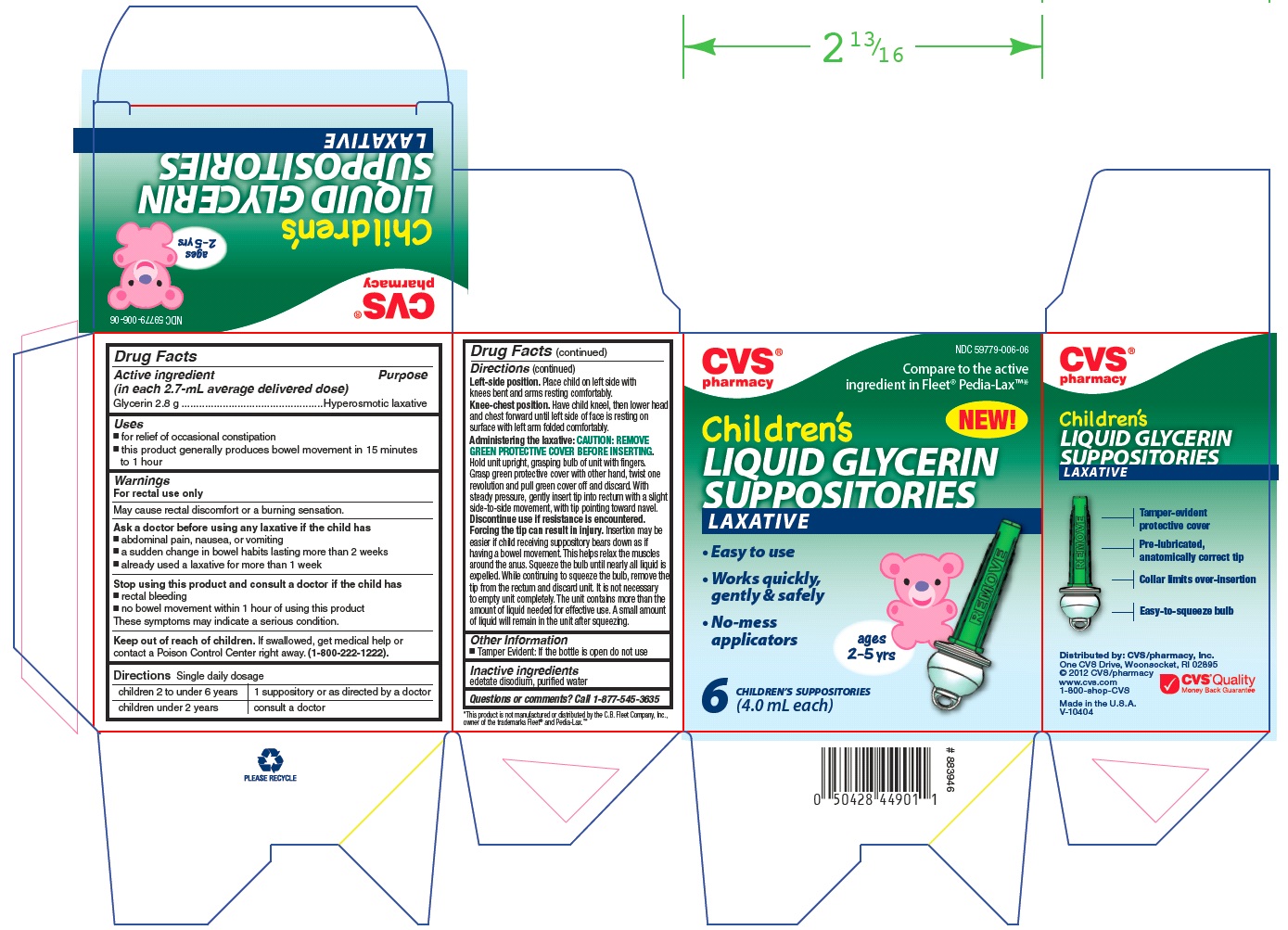
Label: CVS PHARMACY CHILDRENS GLYCERIN SUPPOSITORIES LAXATIVE- glycerin liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 59779-006-06 - Packager: CVS PHARMACY, INC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 10, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient (in each 2.7 - mL average delivered dose)
- Use
- Warnings
- Ask a doctor before using any laxative if you have
- Stop using this product and consult a doctor if you have
- Keep out of reach of children
-
Directions
Single daily dosage
children 2 to under 6 years
1 suppository or as directed by a doctor
children under 2 years
consult a doctor
Left-side position. Place child on left side with knees bent and arms resting comfortably.
Knee-chest position. Have child kneel, then lower head and chest forward until left side of face is resting on surface with left arm folded comfortably.
Administering the laxative: CAUTION: REMOVE GREEN PROTECTIVE COVER BEFORE INSERTING. Hold unit upright, grasping bulb of unit with fingers. Grasp green protective cover with other hand, twist one revolution and pull green cover off and discard. With steady pressure, gently insert tip into rectum with a slight side-to-side movement, with tip pointing toward navel.
Discontinue use if resistance is encountered. Forcing the tip can result in injury. Insertion may be easier if child receiving suppository bears down as if having a bowel movement. This helps relax the muscles around the anus. Squeeze the bulb until nearly all liquid is expelled. While continuing to squeeze the bulb, remove the tip from the rectum and discard unit. It is not necessary to empty unit completely. The unit contains more than the amount of liquid needed for effective use. A small amount of liquid will remain in the unit after squeezing.
- Other Information
- Inactive Ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CVS PHARMACY CHILDRENS GLYCERIN SUPPOSITORIES LAXATIVE
glycerin liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59779-006 Route of Administration RECTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Glycerin (UNII: PDC6A3C0OX) (Glycerin - UNII:PDC6A3C0OX) Glycerin 2.8 g Inactive Ingredients Ingredient Name Strength EDETATE DISODIUM (UNII: 7FLD91C86K) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59779-006-06 4 in 1 CARTON Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part334 06/03/2002 Labeler - CVS PHARMACY, INC. (062312574)