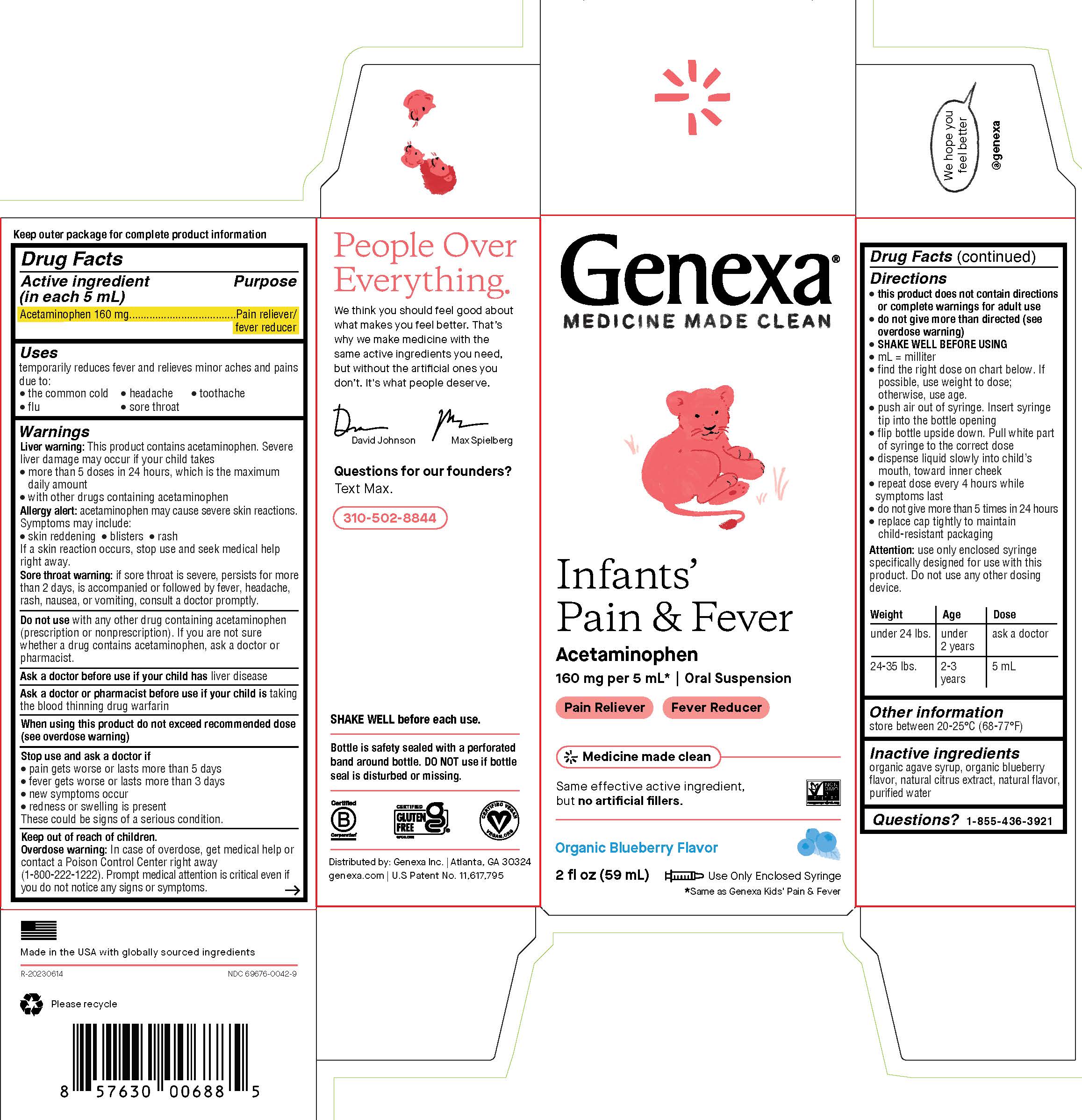
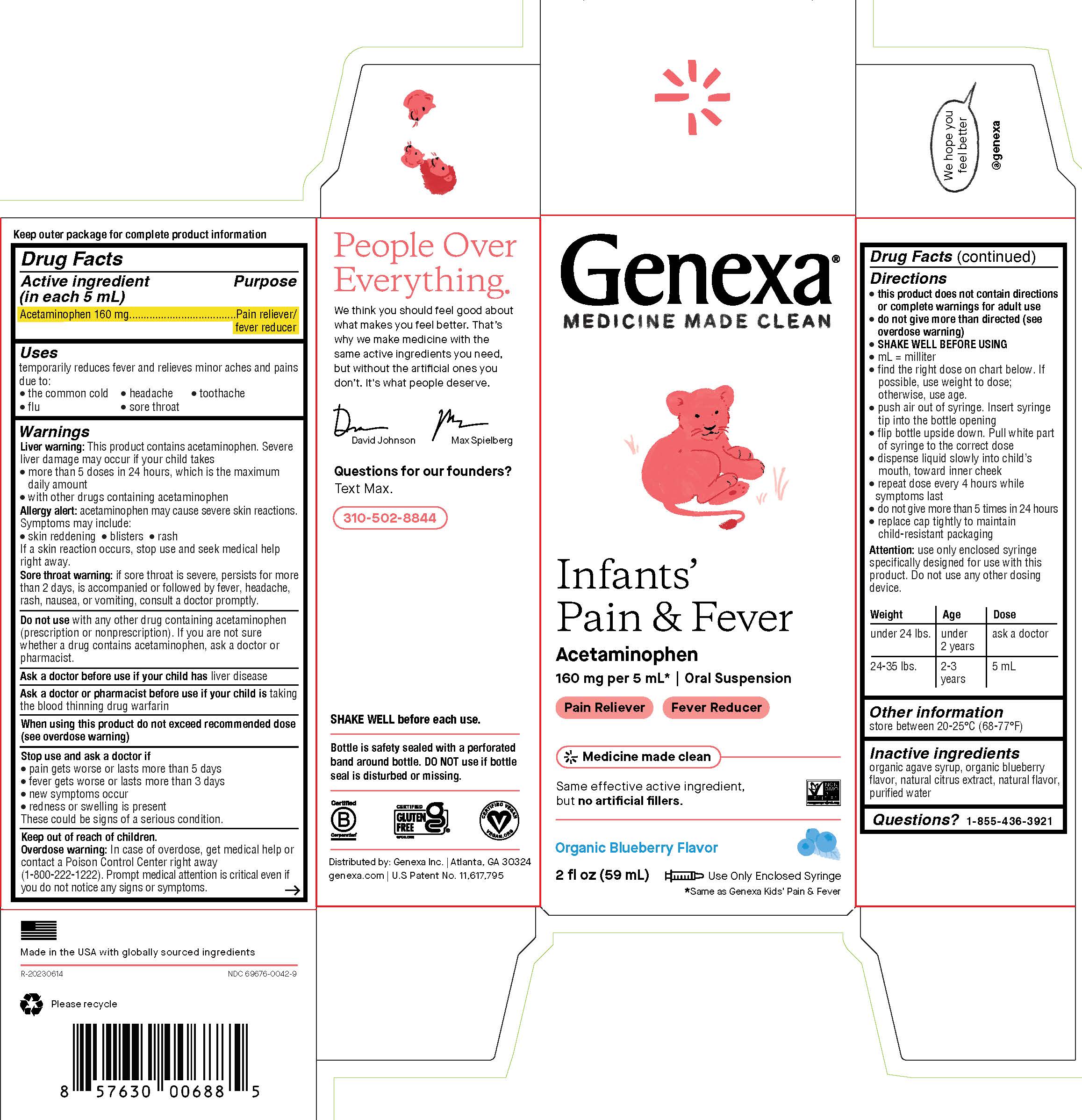
Label: GENEXA INFANTS PAIN AND FEVER- acetaminophen suspension
- NDC Code(s): 69676-0042-9
- Packager: Genexa Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 3, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if your child takes
- more than 5 doses in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
Allergy alert: acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: if sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea or vomiting, consult a doctor promptly.
Do not use with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
-
DOSAGE & ADMINISTRATION
Drug Facts (continued)
Directions
- this product does not contain directions or complete warnings for adult use
- do not give more than directed (see overdose warning)
- SHAKE WELL BEFORE USING
- mL = milliliter
- find the right dose on chart below. If possible, use weight to dose; otherwise, use age
- push air out of syringe. Insert syringe tip into the bottle opening
- flip bottle upside down. Pull white part of syringe to correct dose
- dispense liquid slowly into child's mouth, toward inner cheek
- repeat dose every 4 hours while symptoms last
- do not give more than 5 times in 24 hours
- replace cap tightly to maintain child-resistant packaging
Attention: use only enclosed syringe specifically designed for use with this product. Do not use any other dosing device.
Weight Age Dose under 24 lbs. under 2 years ask a doctor 24-35 lbs. 2-3 years 5 mL - STORAGE AND HANDLING
- INACTIVE INGREDIENT
- QUESTIONS
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL
Genexa®
MEDICINE MADE CLEAN
Infants' Pain & Fever
Acetaminophen
160 mg per 5 mL* | Oral Suspension
Pain Reliever - Fever Reducer
Medicine made clean
Same effective active ingredient, but no artificial fillers.
Organic Blueberry Flavor
2 fl oz (59 mL)
Use Only Enclosed Syringe
*Same as Genexa Kids' Pain & Fever
-
INGREDIENTS AND APPEARANCE
GENEXA INFANTS PAIN AND FEVER
acetaminophen suspensionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69676-0042 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 160 mg in 5 mL Inactive Ingredients Ingredient Name Strength BLUEBERRY (UNII: 253RUG1X1A) AGAVE TEQUILANA JUICE (UNII: GVG8G0207O) WATER (UNII: 059QF0KO0R) CITRUS FRUIT (UNII: XDK00Z8012) Product Characteristics Color brown (Light brown) Score Shape Size Flavor BLUEBERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69676-0042-9 1 in 1 CARTON 12/29/2023 1 59 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 12/29/2023 Labeler - Genexa Inc. (079751024)