Label: KROGER- polyethylene glycol 3350 powder, for solution
- NDC Code(s): 30142-526-07, 30142-526-30
- Packager: KROGER COMPANY
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 19, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
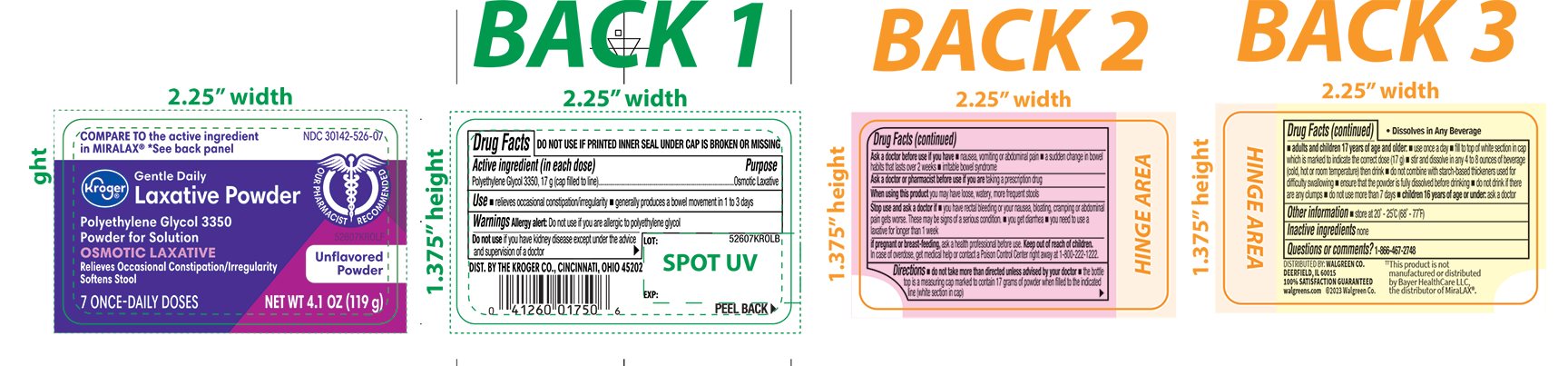
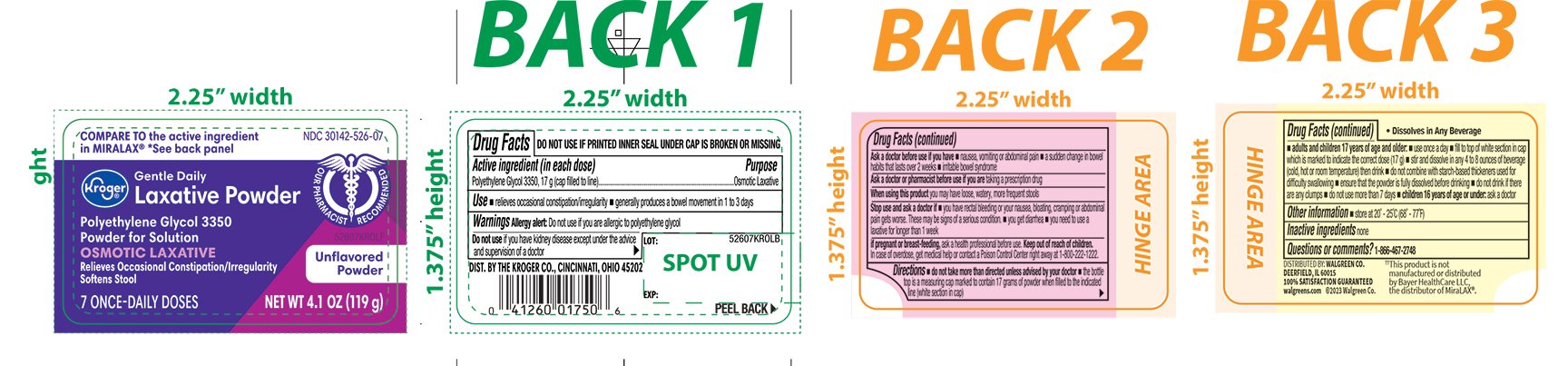
- Active ingredient (in each dose)
- Purpose
- Use
-
Warnings
Ask a doctor before use if you have
- ▪
- nausea, vomiting or abdominal pain
- ▪
- a sudden change in bowel habits that lasts over 2 weeks
- ▪
- irritable bowel syndrome
-
Directions
- ▪
- do not take more than directed unless advised by your doctor
- ▪
- the bottle top is a measuring cap marked to contain 17 grams of powder when filled to the indicated line (white section in cap)
- ▪
-
adults and children 17 years of age and older:
- ▪
- use once a day
- ▪
- fill to top of white section in cap which is marked to indicate the correct dose (17 g)
- ▪
- stir and dissolve in any 4 to 8 ounces of beverage (cold, hot or room temperature) then drink
- ▪
- do not combine with starch-based thickeners used for difficulty swallowing
- ▪
- ensure that the powder is fully dissolved before drinking
- ▪
- do not drink if there are any clumps
- ▪
- do not use no more than 7 days
- ▪
- children 16 years of age or under: ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
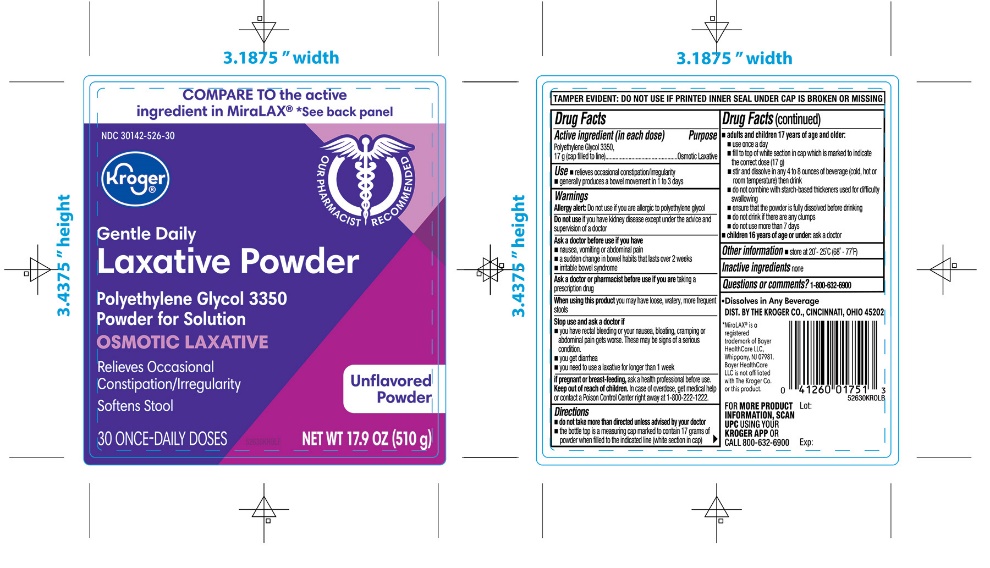
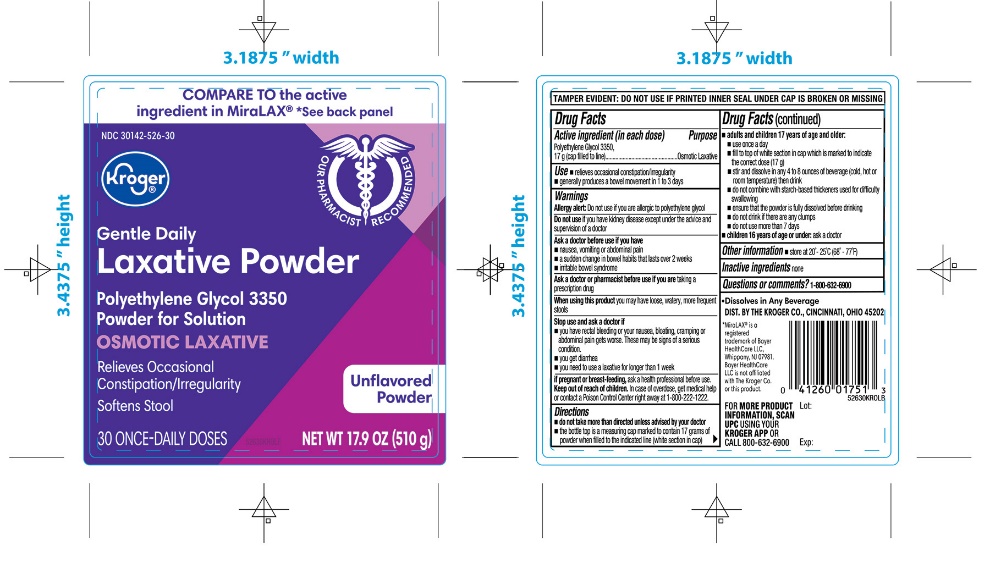
PRINCIPAL DISPLAY PANEL
*Compare to the Active Ingredient in MiraLAX®
NDC 30142-526-30
Polyethylene Glycol 3350,
Powder for Solution,
Osmotic Laxative
- •
- Relieves Occasional Constipation/Irregularity
- •
- Softens Stool
UNFLAVORED POWDER
30 ONCE-DAILY DOSESNET WT 17.9 OZ (510g)
- •
- Dissolves in ANY Beverage
*This product is not manufactured or distributed by Bayer HealthCare LLC, the distributer of MiraLAX®
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
KROGER
polyethylene glycol 3350 powder, for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:30142-526 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) (POLYETHYLENE GLYCOL 3350 - UNII:G2M7P15E5P) POLYETHYLENE GLYCOL 3350 17 g in 17 g Product Characteristics Color white (Colorless upon dissolution) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:30142-526-30 510 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 10/03/2022 2 NDC:30142-526-07 119 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 10/03/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA202071 10/03/2022 Labeler - KROGER COMPANY (006999528)