Label: SUFFUSION EAR CLEANER- salicylic acid, phytosphingosine hcl solution
- NDC Code(s): 46066-168-80, 46066-168-90, 46066-168-91
- Packager: Aspen Veterinary Resource, LTD.
- Category: OTC ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated February 25, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
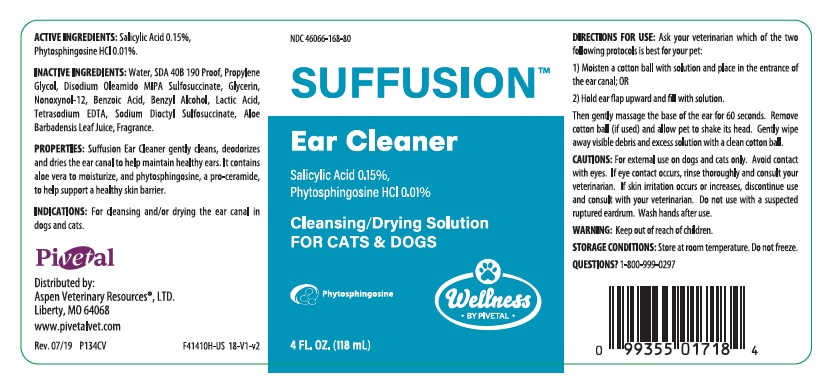
SUFFUSION Ear Cleaner
ACTIVE INGREDIENTS: Salicylic Acid 0.15%, Phytosphingosine HCl 0.01%.
INACTIVE INGREDIENTS: Water, SDA 40B 190 Proof, Propylene Glycol, Disodium Oleamido MIPA Sulfosuccinate, Glycerin, Nonoxynol-12, Benzoic Acid, Benzyl Alcohol, Lactic Acid, Tetrasodium EDTA, Sodium Dioctyl Sulfosuccinate, Aloe Barbadensis Leaf Juice, Fragrance.
PROPERTIES: Suffusion Ear Cleaner gently cleans, deodorizes and dries the ear canal to help maintain healthy ears. It contains aloe vera to moisturize, and phytosphingosine, a pro-ceramide, to help support a healthy skin barrier.
-
INDICATIONS & USAGE
INDICATION: For cleansing and/or drying the ear canal in dogs and cats.
DIRECTIONS FOR USE: Ask your veterinarian which of the two following protocols is best for your pet:
1) Moisten a cotton ball with solution and place at the entrance of the ear canal; OR
2) Hold ear flap upward and fill with solution.
Then gently massage the base of the ear for 60 seconds. Remove cotton ball (if used) and allow pet to shake its head. Gently wipe away visible debris and exceess solution with a clean cotton ball.
-
WARNINGS AND PRECAUTIONS
CAUTIONS: For external use on dogs and cats only. Avoid contact with the eyes. If eye contact occurs, rinse thoroughly and consult your veterinarian. If skin irritation occurs or increases, discontinue use and consult with your veterinarian. Do not use with a suspected ruptured eardrum. Wash hands after use.
WARNING: Keep out of reach of children.
- STORAGE AND HANDLING
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 4 OUNCE (118 mL) Bottle
- PRINCIPAL DISPLAY PANEL - 8 OUNCE (237 mL) Bottle
- PRINCIPAL DISPLAY PANEL - 16 OUNCE (473 mL) Bottle
-
INGREDIENTS AND APPEARANCE
SUFFUSION EAR CLEANER
salicylic acid, phytosphingosine hcl solutionProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:46066-168 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 1.5 mg in 1 mL PHYTOSPHINGOSINE HYDROCHLORIDE (UNII: TT871XV7TU) (PHYTOSPHINGOSINE - UNII:GIN46U9Q2Q) PHYTOSPHINGOSINE HYDROCHLORIDE 0.1 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) DISODIUM OLEAMIDO MIPA-SULFOSUCCINATE (UNII: 0MBZ20845F) NONOXYNOL-12 (UNII: 6NR43D77O6) GLYCERIN (UNII: PDC6A3C0OX) BENZOIC ACID (UNII: 8SKN0B0MIM) BENZYL ALCOHOL (UNII: LKG8494WBH) LACTIC ACID (UNII: 33X04XA5AT) EDETATE SODIUM (UNII: MP1J8420LU) DI-N-OCTYL SODIUM SULFOSUCCINATE (UNII: 4YLY5570Y0) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:46066-168-80 118 mL in 1 BOTTLE, PLASTIC 2 NDC:46066-168-90 237 mL in 1 BOTTLE, PLASTIC 3 NDC:46066-168-91 473 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/27/2019 Labeler - Aspen Veterinary Resource, LTD. (627265361) Registrant - Ceva Sante Animale (261126049)