Label: TERRAVITALS LOBAK- chamomilla, phosphorus, platinum metallicum, ruta graveolens, veratrum album pellet
- NDC Code(s): 74474-103-01
- Packager: Terravitals LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated June 15, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Terravitals LoBak Active Ingredients
Drug Facts
Active ingredients
Chamomilla officinalis 6C HPUS
Phosphorus 30C HPUS
Platinum Metallicum 30C HPUS
Ruta graveolens 6C HPUS
Veratrum Album 30C HPUS
'HPUS' indicates that this ingredient is officially included in the Homeopathic Pharmacopoeia of the United States.
Small inner packaging label:
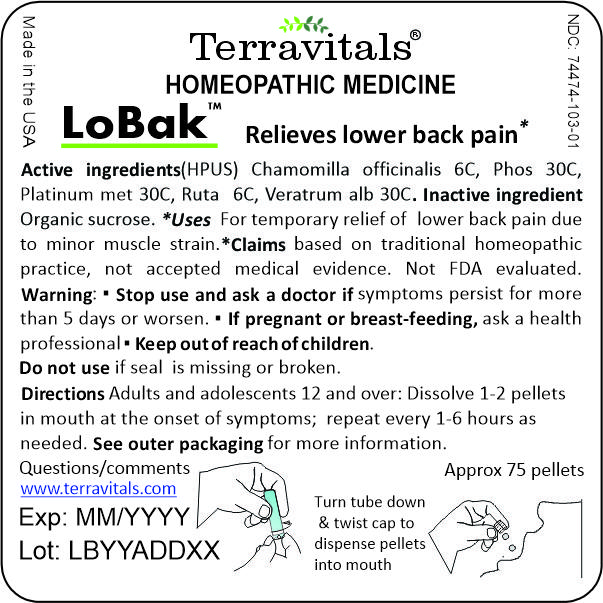
-
Terravitals LoBak Purpose
Active ingredients** ...................................................................... Purpose
Chamomilla officinalis 6C HPUS.................. lumbago and pain in sacrum
Phosphorus 30C HPUS............................................ burning pain in back
Platinum Metallicum 30C HPUS.... spasmodic pain, numbness in coccyx
Ruta graveolens 6C HPUS..................pain in sacrum, lumbar vertebreae
Veratrum album 30C HPUS...................stiffness to sacrum, small of back
'HPUS' indicates that this ingredient is officially included in the Homeopathic Pharmacopoeia of the United States
**'C' is homeopathic dilution.
Uses* For temporary relief of lower back and lumbar pain due to minor muscle strain.
*Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
- Terravitals Warnings
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- Terravitals Dosage
- Terravitals Other
- DO NOT USE
- Terravitals Inactive Ingredient
-
Terravitals LoBak main panel
TERRAVITALS LoBak
Homeopathic Medicine
Relieves lower back pain*
NDC 74474-103-01
Approx 75 Oral soluble pellets
Non-drowsy
Not habit-forming
No drug interactions
No known side effects
Made in the USA by Terravitals LLC, Gaithersburg MD 20879
www.terravitals.com
Exp: MM/YYYY
Lot: YYADXX
Outer packaging label:

-
INGREDIENTS AND APPEARANCE
TERRAVITALS LOBAK
chamomilla, phosphorus, platinum metallicum, ruta graveolens, veratrum album pelletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:74474-103 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHOSPHORUS (UNII: 27YLU75U4W) (PHOSPHORUS - UNII:27YLU75U4W) PHOSPHORUS 30 [hp_C] CHAMOMILE (UNII: FGL3685T2X) (CHAMOMILE - UNII:FGL3685T2X) CHAMOMILE 6 [hp_C] RUTA GRAVEOLENS FLOWERING TOP (UNII: N94C2U587S) (RUTA GRAVEOLENS FLOWERING TOP - UNII:N94C2U587S) RUTA GRAVEOLENS FLOWERING TOP 6 [hp_C] VERATRUM ALBUM ROOT (UNII: QNS6W5US1Z) (VERATRUM ALBUM ROOT - UNII:QNS6W5US1Z) VERATRUM ALBUM ROOT 30 [hp_C] PLATINUM (UNII: 49DFR088MY) (PLATINUM - UNII:49DFR088MY) PLATINUM 30 [hp_C] Inactive Ingredients Ingredient Name Strength SUCROSE (UNII: C151H8M554) Product Characteristics Color white (White spheroids) Score Shape Size 4mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:74474-103-01 1 in 1 TUBE 06/15/2021 1 1 in 1 BOTTLE, DISPENSING; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 06/15/2021 Labeler - Terravitals LLC (117322434) Establishment Name Address ID/FEI Business Operations Terravitals LLC 117322434 manufacture(74474-103)

