Label: NU SKIN AGELOC ME DAY S1 BROAD SPECTRUM SPF 25- homosalate, octinoxate, avobenzone, and octocrylene lotion
- NDC Code(s): 62839-1617-1
- Packager: NSE Products, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 24, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
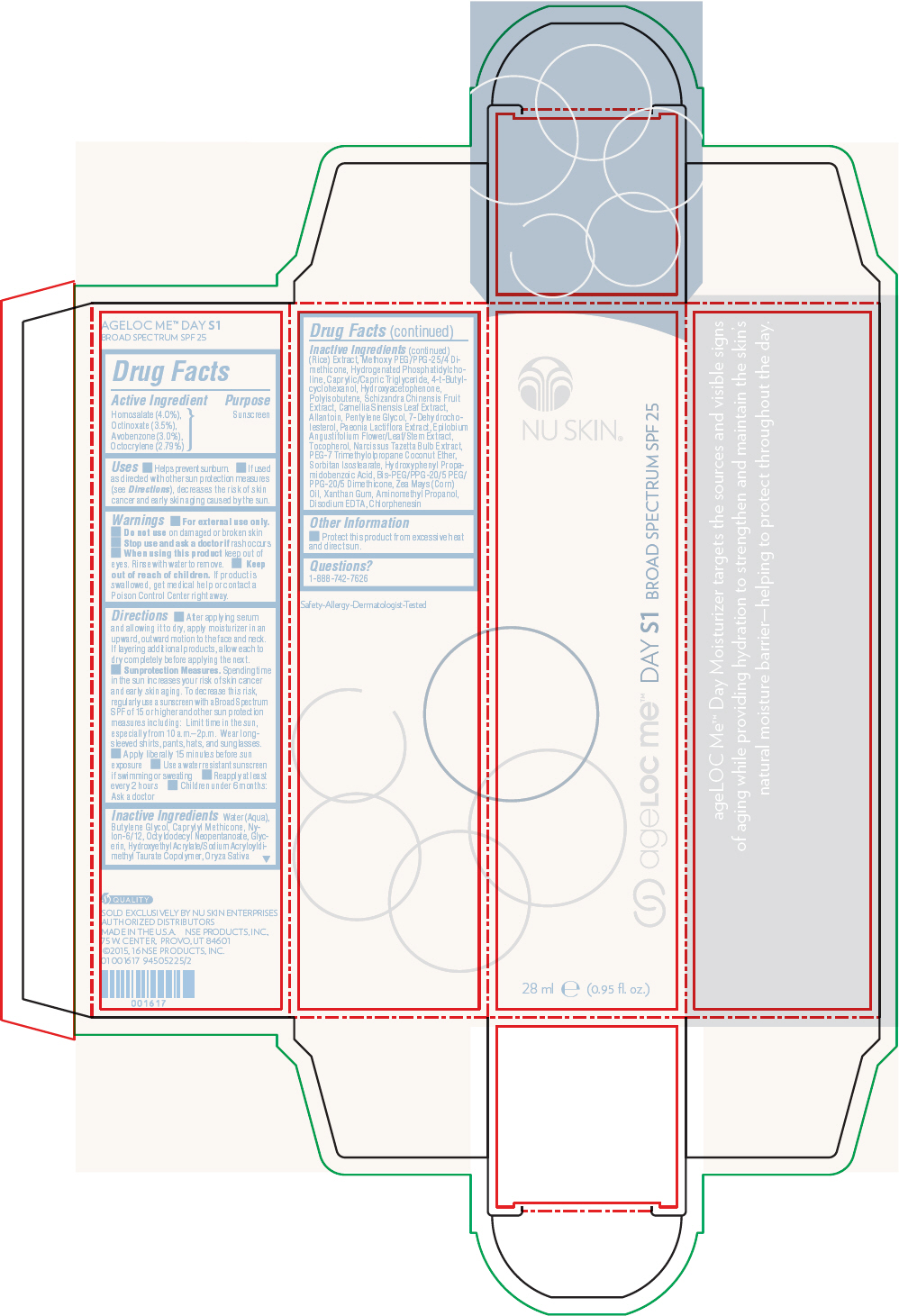
- Active Ingredient
- Purpose
-
Uses
- Helps prevent sunburn.
- If used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun.
- Warnings
-
Directions
- After applying serum and allowing it to dry, apply moisturizer in an upward, outward motion to the face and neck. If layering additional products, allow each to dry completely before applying the next.
- Sunprotection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF of 15 or higher and other sun protection measures including: Limit time in the sun, especially from 10 a.m.–2p.m. Wear long-sleeved shirts, pants, hats, and sunglasses.
- Apply liberally 15 minutes before sun exposure
- Use a water resistant sunscreen if swimming or sweating
- Reapply at least every 2 hours
- Children under 6 months: Ask a doctor
-
Inactive Ingredients
Water (Aqua), Butylene Glycol, Caprylyl Methicone, Nylon-6/12, Octyldodecyl Neopentanoate, Glycerin, Hydroxyethyl Acrylate/Sodium Acryloyldimethyl Taurate Copolymer, Oryza Sativa (Rice) Extract, Methoxy PEG/PPG-25/4 Dimethicone, Hydrogenated Phosphatidylcholine, Caprylic/Capric Triglyceride, 4-t-Butylcyclohexanol, Hydroxyacetophenone, Polyisobutene, Schizandra Chinensis Fruit Extract, Camellia Sinensis Leaf Extract, Allantoin, Pentylene Glycol, 7-Dehydrocholesterol, Paeonia Lactiflora Extract, Epilobium Angustifolium Flower/Leaf/Stem Extract, Tocopherol, Narcissus Tazetta Bulb Extract, PEG-7 Trimethylolpropane Coconut Ether, Sorbitan Isostearate, Hydroxyphenyl Propamidobenzoic Acid, Bis-PEG/PPG-20/5 PEG/PPG-20/5 Dimethicone, Zea Mays (Corn) Oil, Xanthan Gum, Aminomethyl Propanol, Disodium EDTA, Chlorphenesin
- Other Information
- Questions?
- PRINCIPAL DISPLAY PANEL - 28 ml Cartridge Carton
-
INGREDIENTS AND APPEARANCE
NU SKIN AGELOC ME DAY S1 BROAD SPECTRUM SPF 25
homosalate, octinoxate, avobenzone, and octocrylene lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62839-1617 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Homosalate (UNII: V06SV4M95S) (Homosalate - UNII:V06SV4M95S) Homosalate 40 mg in 1 mL Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 35 mg in 1 mL Avobenzone (UNII: G63QQF2NOX) (Avobenzone - UNII:G63QQF2NOX) Avobenzone 30 mg in 1 mL Octocrylene (UNII: 5A68WGF6WM) (Octocrylene - UNII:5A68WGF6WM) Octocrylene 27.9 mg in 1 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Butylene Glycol (UNII: 3XUS85K0RA) Octyldodecyl Neopentanoate (UNII: X8725R883T) Caprylyl Trisiloxane (UNII: Q95M2P1KJL) Pentylene Glycol (UNII: 50C1307PZG) Glycerin (UNII: PDC6A3C0OX) Medium-Chain Triglycerides (UNII: C9H2L21V7U) Rice Germ (UNII: 7N2B70SFEZ) Xanthan Gum (UNII: TTV12P4NEE) 4-tert-Butylcyclohexanol (UNII: K0H1405S9C) Chlorphenesin (UNII: I670DAL4SZ) Allantoin (UNII: 344S277G0Z) Edetate Disodium (UNII: 7FLD91C86K) Epilobium Angustifolium Flowering Top (UNII: 08H094218D) Green Tea Leaf (UNII: W2ZU1RY8B0) Sorbitan Isostearate (UNII: 01S2G2C1E4) Hydroxyphenyl Propamidobenzoic Acid (UNII: 25KRT26H77) Schisandra Chinensis Fruit (UNII: ABS794681C) Corn Oil (UNII: 8470G57WFM) Narcissus Tazetta Bulb (UNII: K17762966S) 7-Dehydrocholesterol (UNII: BK1IU07GKF) Aminomethylpropanol (UNII: LU49E6626Q) Tocopherol (UNII: R0ZB2556P8) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62839-1617-1 1 in 1 CARTON 05/01/2016 1 28 mL in 1 CARTRIDGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 05/01/2016 Labeler - NSE Products, Inc. (803486393)