Label: SCARLIGHT MD- hydroquinone liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 64269-9902-7, 64269-9902-8 - Packager: Scarguard Labs, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 29, 2009
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
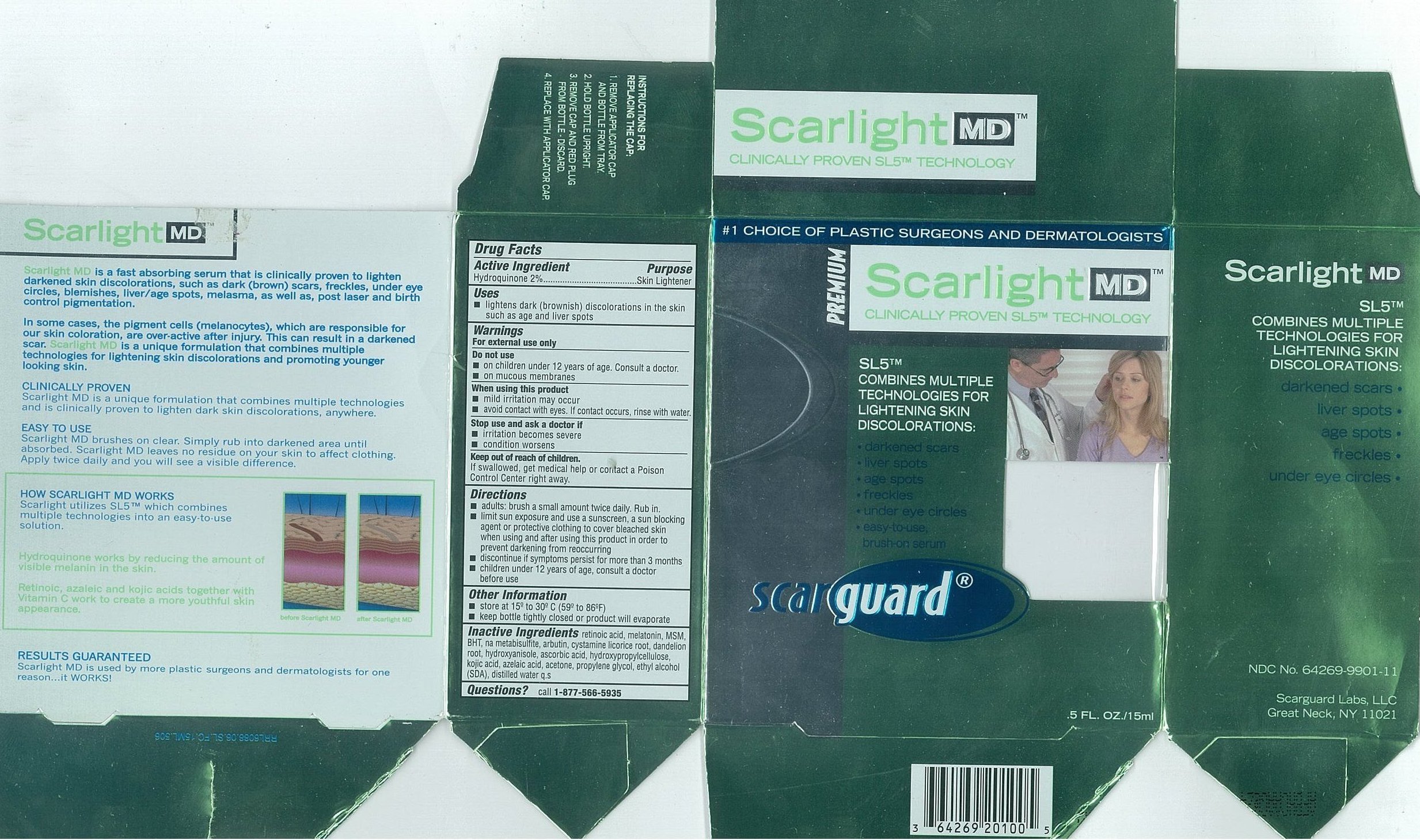
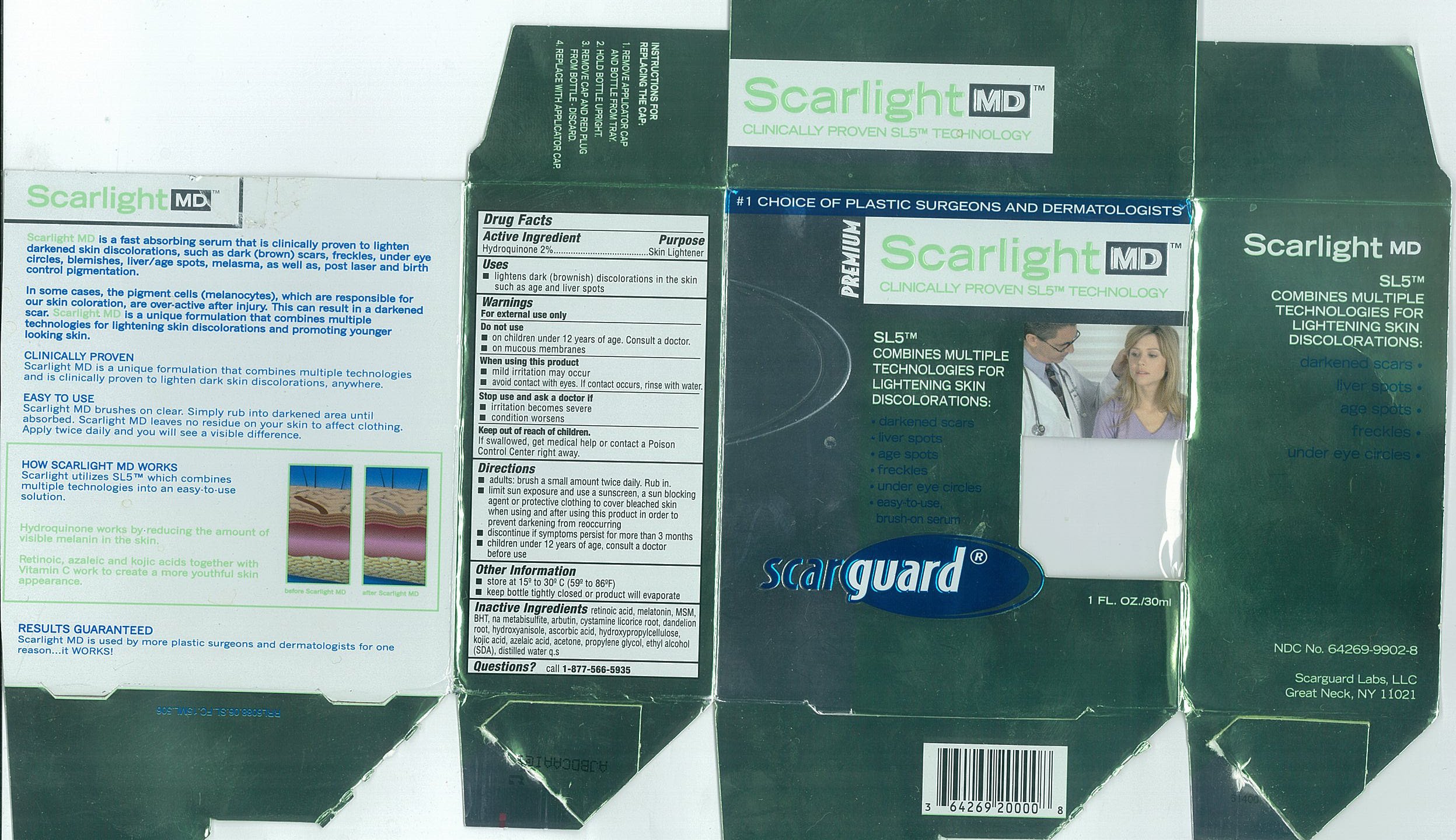
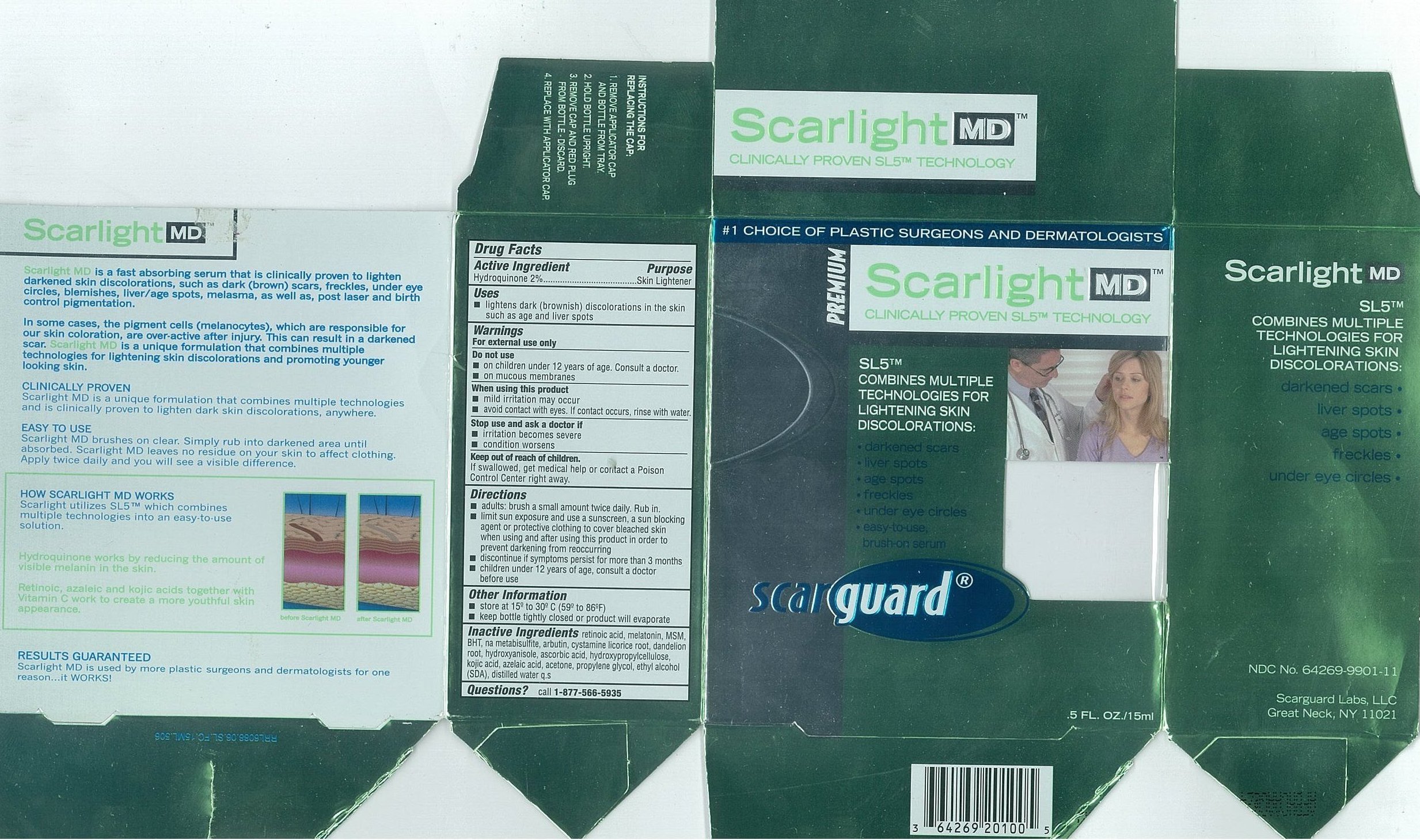
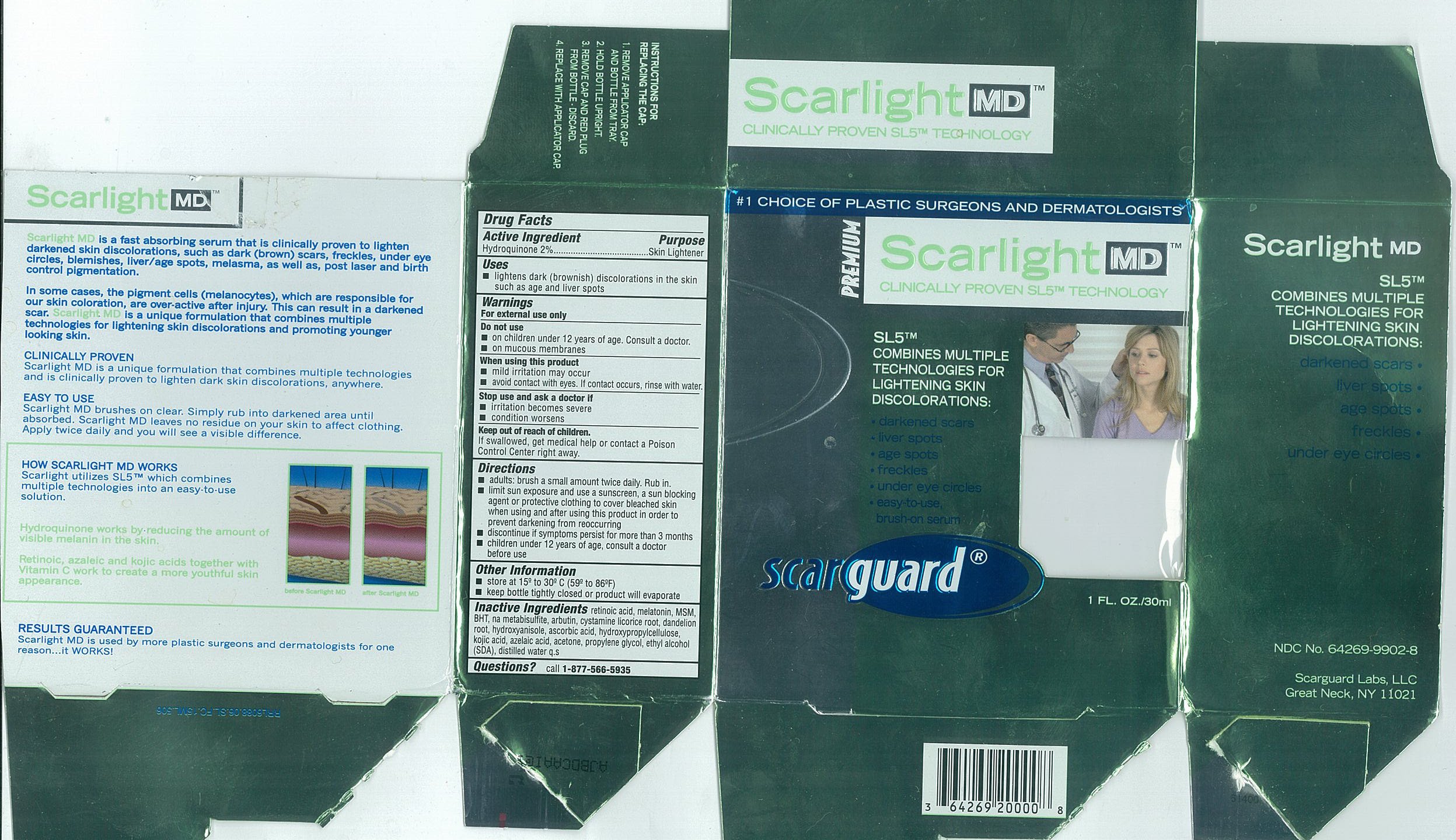
- Active Ingredients
- Purpose
- Uses
- Warnings
- Do not use
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children.
-
Directions
- adults: brush a small amount twice daily. Rub in.
- limit sun exposure and use a sunscreen, a sun blocking agent or protective clothing to cover bleached skin when using and after using this product in order to prevent darkening from reoccurring
- discontinue if symptoms persist for more than 3 months
- children under 12 years of age, consult a doctor before use
- Other Information
- Inactive Ingredients
- Questions
- Carton 15mL
- Carton 30mL
-
INGREDIENTS AND APPEARANCE
SCARLIGHT MD
hydroquinone liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:64269-9902 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROQUINONE (UNII: XV74C1N1AE) (Hydroquinone - UNII:XV74C1N1AE) HYDROQUINONE 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength Propylene Glycol (UNII: 6DC9Q167V3) Alcohol (UNII: 3K9958V90M) Acetone (UNII: 1364PS73AF) Azelaic Acid (UNII: F2VW3D43YT) Hydroxypropyl Cellulose (UNII: RFW2ET671P) Ascorbic Acid (UNII: PQ6CK8PD0R) Butylated Hydroxyanisole (UNII: REK4960K2U) TARAXACUM OFFICINALE ROOT (UNII: 9DE5YCO0RU) Licorice (UNII: 61ZBX54883) Arbutin (UNII: C5INA23HXF) Sodium Metabisulfite (UNII: 4VON5FNS3C) Butylated Hydroxytoluene (UNII: 1P9D0Z171K) Dimethyl Sulfone (UNII: 9H4PO4Z4FT) Melatonin (UNII: JL5DK93RCL) Tretinoin (UNII: 5688UTC01R) Water (UNII: 059QF0KO0R) Kojic Acid (UNII: 6K23F1TT52) Cystamine (UNII: R110LV8L02) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64269-9902-8 30 mL in 1 BOTTLE, WITH APPLICATOR 2 NDC:64269-9902-7 15 mL in 1 BOTTLE, WITH APPLICATOR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part358 11/01/2002 Labeler - Scarguard Labs, LLC (842204575) Registrant - Scarguard Labs, LLC (842204575)