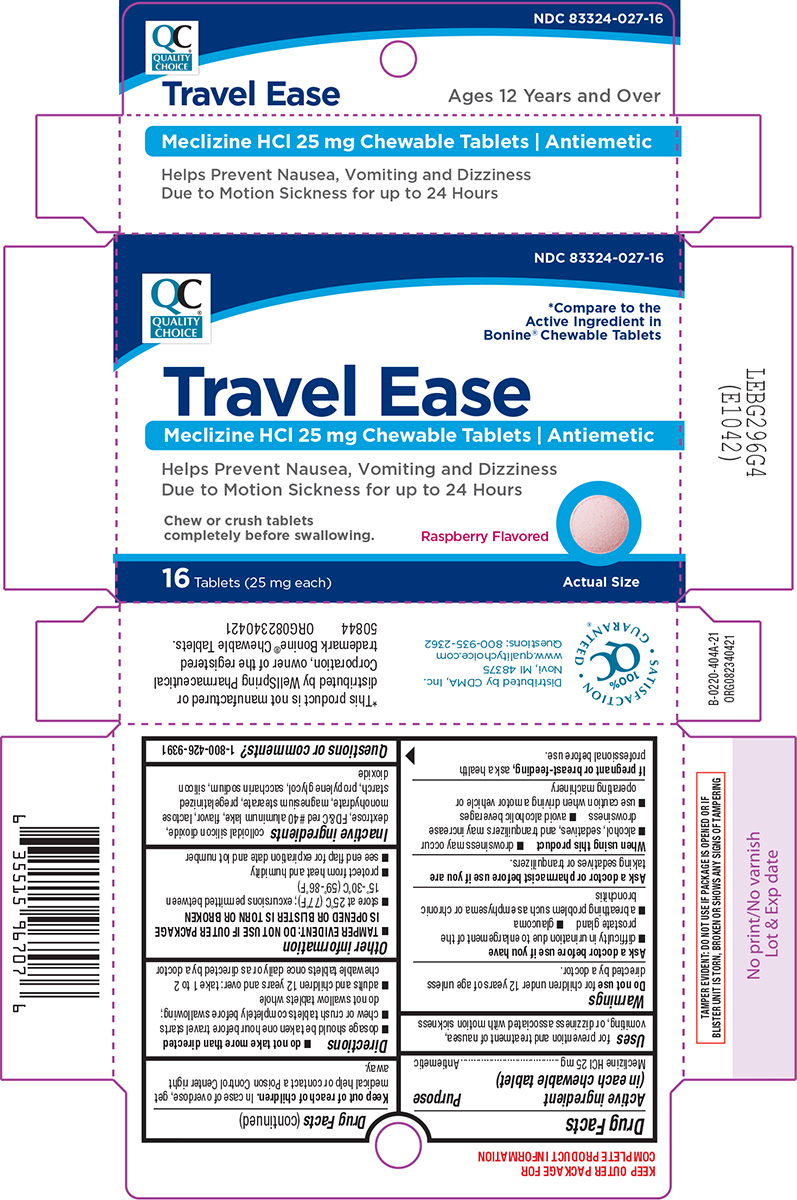
Label: TRAVEL EASE- meclizine hcl tablet, chewable
- NDC Code(s): 83324-027-16
- Packager: Chain Drug Marketing Association, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 27, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each chewable tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
- a breathing problem such as emphysema or chronic bronchitis
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
QC NDC 83324-027-16
QUALITY
CHOICE*Compare to the
Active Ingredient in
Bonine® Chewable TabletsTravel Ease
Meclizine HCl 25 mg Chewable Tablets | Antiemetic
Helps Prevent Nausea, Vomiting and Dizziness
Due to Motion Sickness for up to 24 HoursChew or crush tablets
completely before swallowing. Raspberry Flavored16 Tablets (25 mg each) Actual Size
Distributed by CDMA, Inc.
Novi, MI 48375
www.qualitychoice.com
Questions: 800-935-2362TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF
BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING*This product is not manufactured or
distributed by WellSpring Pharmaceutical
Corporation, owner of the registered
trademark Bonine® Chewable Tablets.
50844 ORG082340421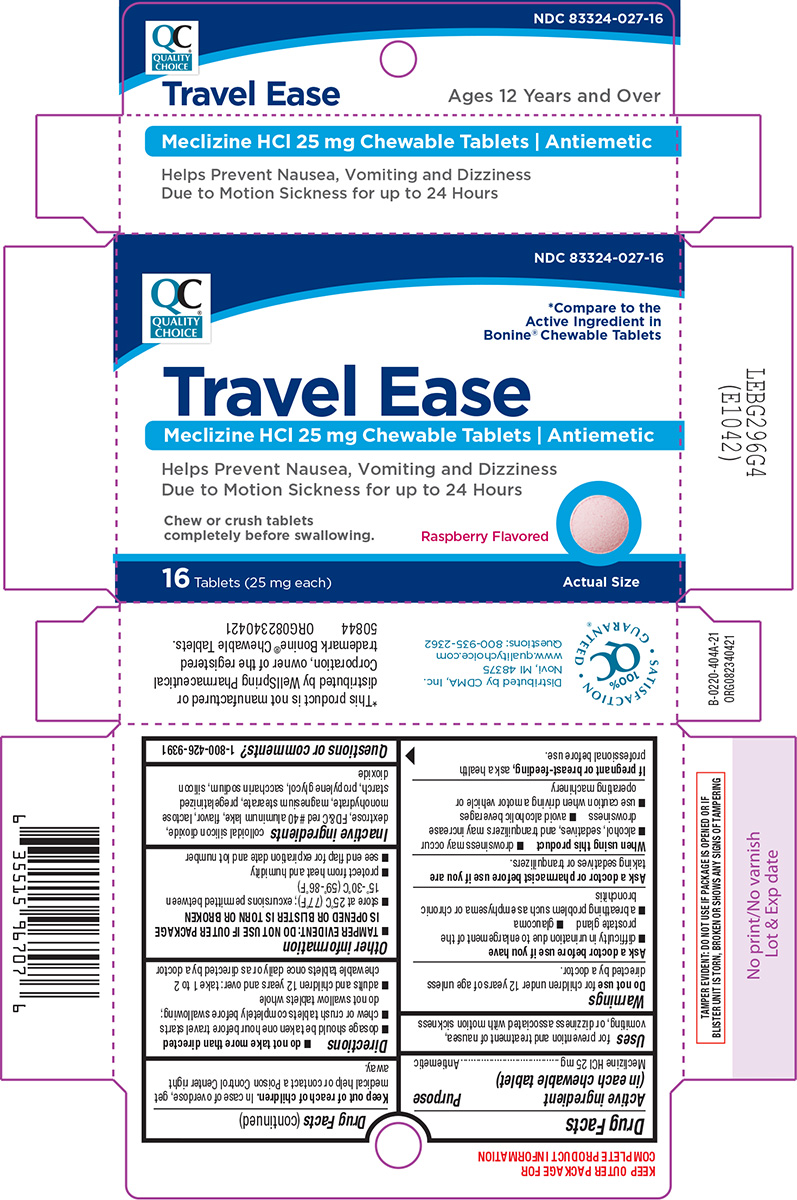
Quality choice 44-404A
-
INGREDIENTS AND APPEARANCE
TRAVEL EASE
meclizine hcl tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83324-027 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MECLIZINE HYDROCHLORIDE (UNII: HDP7W44CIO) (MECLIZINE - UNII:3L5TQ84570) MECLIZINE HYDROCHLORIDE 25 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) DEXTROSE, UNSPECIFIED FORM (UNII: IY9XDZ35W2) FD&C RED NO. 40 (UNII: WZB9127XOA) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SACCHARIN SODIUM (UNII: SB8ZUX40TY) Product Characteristics Color pink Score no score Shape ROUND Size 9mm Flavor RASPBERRY Imprint Code 44;404 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83324-027-16 2 in 1 CARTON 03/27/2024 1 8 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M009 03/27/2024 Labeler - Chain Drug Marketing Association, Inc. (011920774) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(83324-027) , pack(83324-027) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(83324-027)