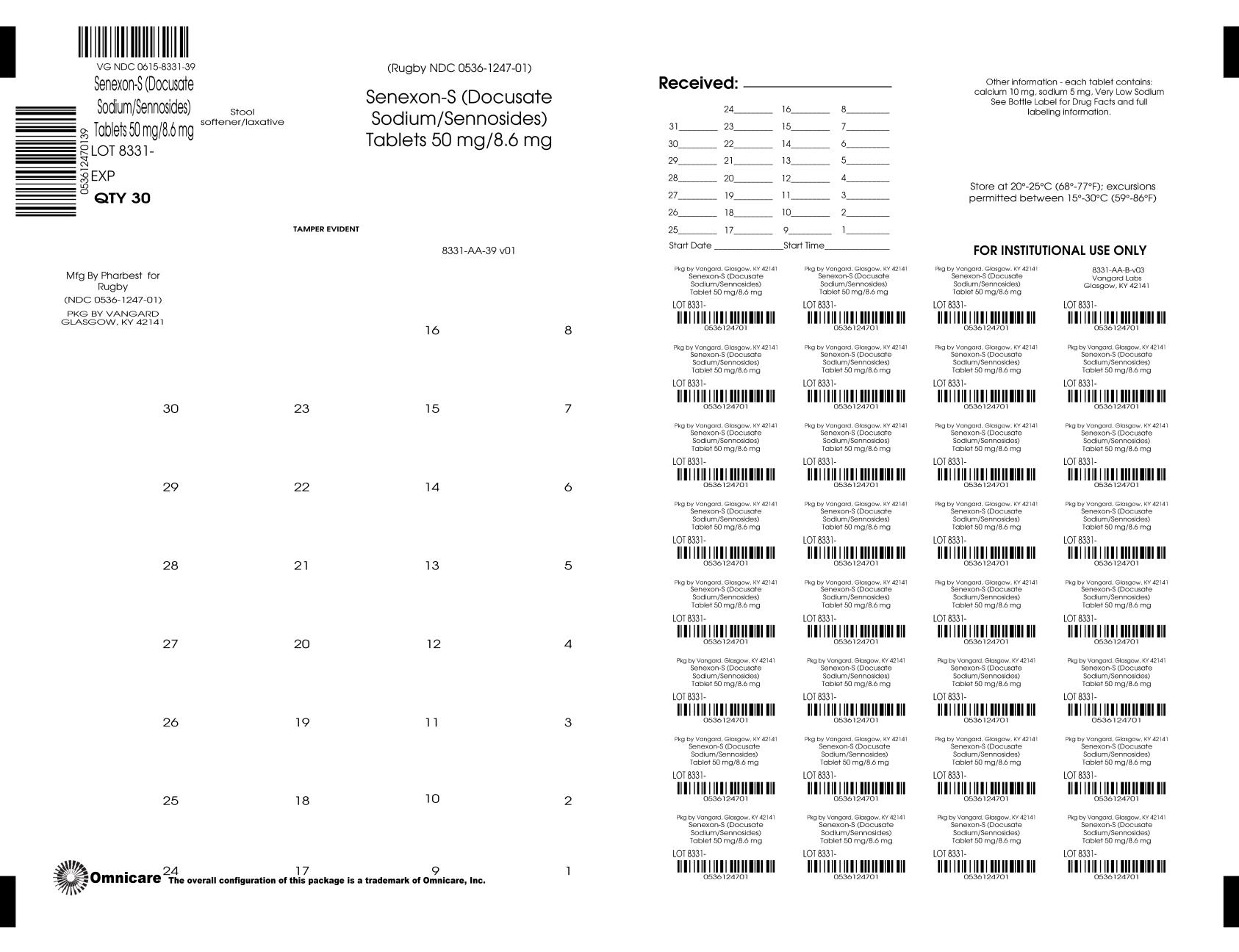
Label: SENEXON-S- docusate sodium 50mg and sennosides 8.6mg tablet, film coated
- NDC Code(s): 0615-8331-39
- Packager: NCS HealthCare of KY, LLC dba Vangard Labs
- This is a repackaged label.
- Source NDC Code(s): 0536-1247
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 24, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients (in each tablet)
- Purpose
- Uses
-
Warnings
Do not use
- if you are now taking mineral oil, unless directed by a doctor
- laxative products for longer than 1 week unless directed by a doctor
Ask a doctor before use if you have
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that continues over a period of 2 weeks
-
Directions
- take preferably at bedtime or as directed by a doctor
age
starting dosage
maximum dosage
adults and children 12 years and over
2 tablets once a day
4 tablets twice a day
children 6 to under 12 years
1 tablet once a day
2 tablets twice a day
children 2 to under 6 years
1/2 tablet once a day
1 tablet twice a day
children under 2 years
ask a doctor
ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- Principal Display Section
-
INGREDIENTS AND APPEARANCE
SENEXON-S
docusate sodium 50mg and sennosides 8.6mg tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0615-8331(NDC:0536-1247) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SENNOSIDES (UNII: 3FYP5M0IJX) (SENNOSIDES - UNII:3FYP5M0IJX) SENNOSIDES 8.6 mg DOCUSATE SODIUM (UNII: F05Q2T2JA0) (DOCUSATE - UNII:M7P27195AG) DOCUSATE SODIUM 50 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) CALCIUM PHOSPHATE, DIBASIC, DIHYDRATE (UNII: O7TSZ97GEP) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) SODIUM BENZOATE (UNII: OJ245FE5EU) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color orange (ORANGE COLOR) Score no score Shape ROUND (ROUND TABLET) Size 10mm Flavor Imprint Code PH32 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0615-8331-39 30 in 1 BLISTER PACK; Type 0: Not a Combination Product 01/07/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 10/02/2019 Labeler - NCS HealthCare of KY, LLC dba Vangard Labs (050052943) Establishment Name Address ID/FEI Business Operations NCS HealthCare of KY, LLC dba Vangard Labs 050052943 repack(0615-8331)