Label: THE ORGANIC PHARMACY CELLULAR PROTECTION SUNSCREEN SPF 30- titanium dioxide and zinc oxide cream
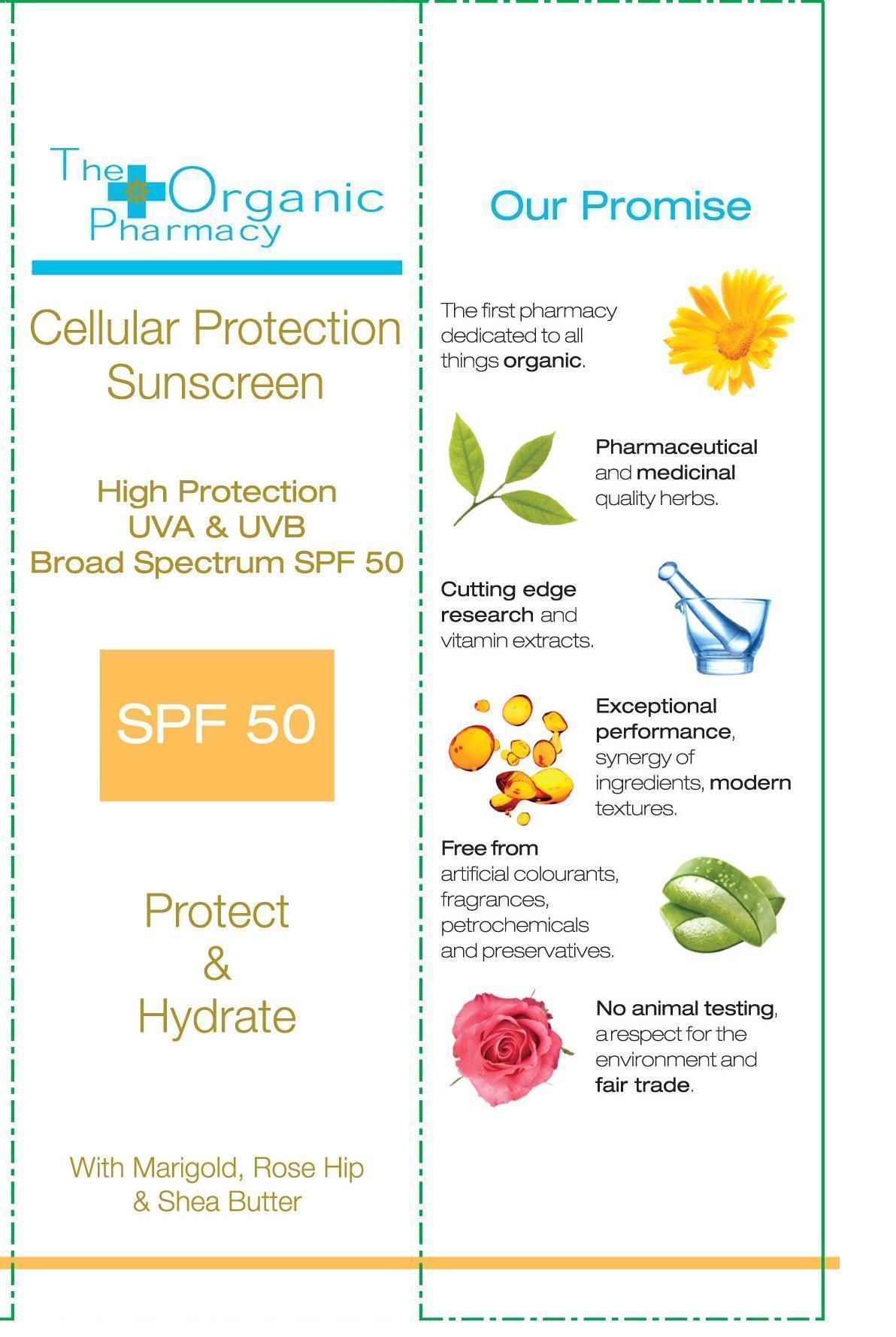
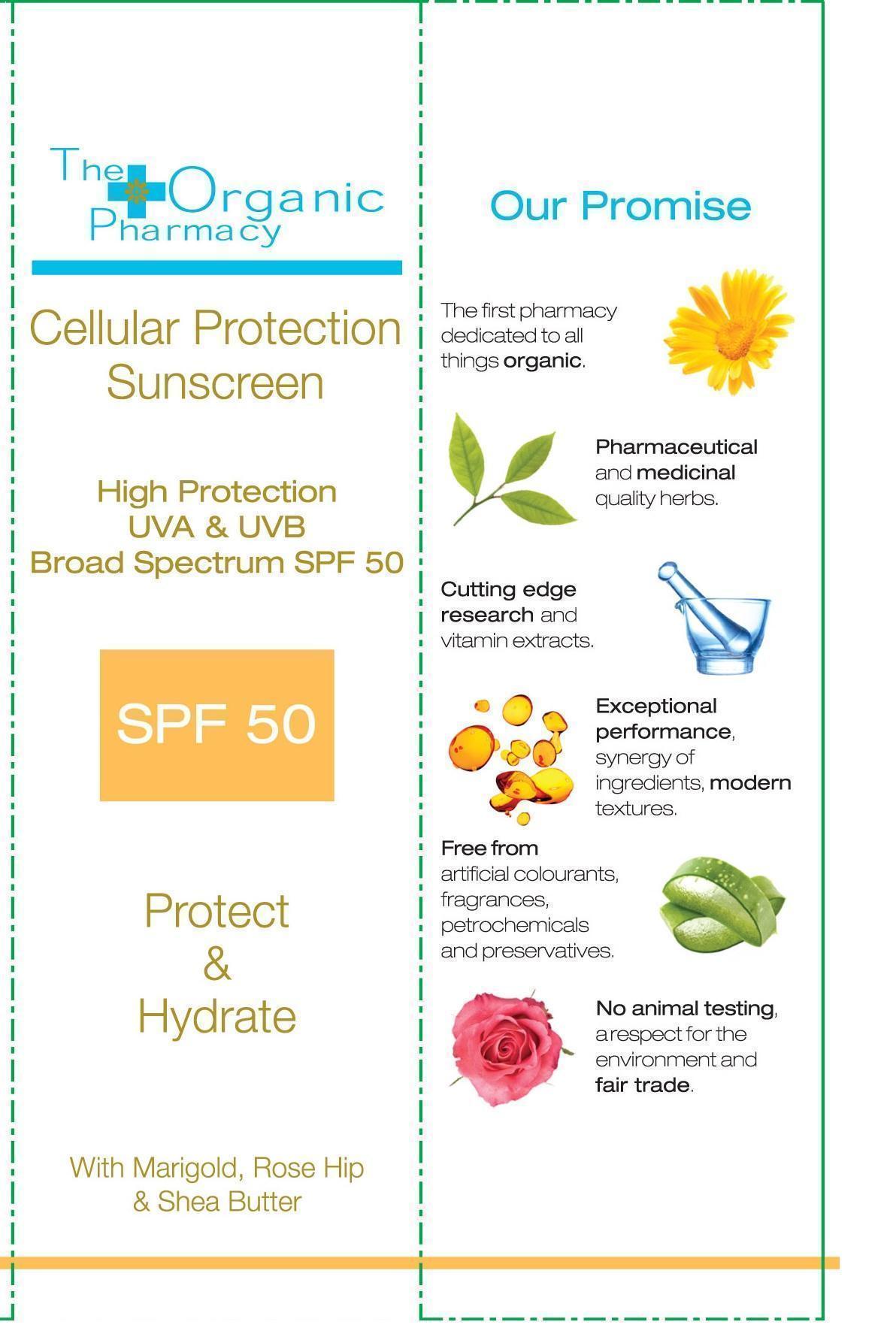
THE ORGANIC PHARMACY CELLULAR PROTECTION SUNSCREEN SPF 50- titanium dioxide and zinc oxide cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 69303-630-01, 69303-650-01 - Packager: The organic pharmacy Limited
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 17, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Carton.DrugFactsSPF50
Active ingredients
Zinc Oxide 12.5%
Titanium Dioxide 1%
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
Warnings
For external use only
Do not use on damaged or broken skin
When using this product keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if rash occurs.
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison contorl Center right away.
Directions
- apply liberally 15 minutes before sun exposure
- reapply:
- after swimming or sweating
- immediately after towel drying
- at least every 2 hours
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin ageing. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long sleeve shirts, pants, hats and sunglasses
- children under 6 months: Ask a doctor
Inactive ingredients
Aloe Barbadensis Leaf Juice, Caprylic/Capric Triglyceride, Alcohol, Aqua, Glycerin, Behenyl Alcohol, Cetearyl Alcohol, Glyceryl Stearate, Butyrospermum Parkii Butter, Calendula Officinalis Extract, Helianthus Annuus Seed Oil, Rosa Canina Fruit Oil, Phospholipids, Parfum , Xanthan Gum, Citrus Grandis Seed Extract, Lactic Acid, Glycine Soja Sterols, Silica, Tocopherol
Other information
- protect this product from excessive heat and direct sun
Questions or comments?
UK-0844-800-8399
US-310-272-7275

-
CartonSPF30.DrugFacts
Active ingredients
Zinc Oxide 10 %
Titanium Dioxide 1%
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
Warnings
For external use only
Do not use on damaged or broken skin
When using this product keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if rash occurs.
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison contorl Center right away.
Directions
- apply liberally 15 minutes before sun exposure
- reapply:
- after swimming or sweating
- immediately after towel drying
- at least every 2 hours
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin ageing. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long sleeve shirts, pants, hats and sunglasses
- children under 6 months: Ask a doctor
Inactive ingredients
Aloe Barbadensis Leaf Juice, Aqua, Caprylic/Capric Triglyceride, Alcohol, Glycerin, Behenyl Alcohol, Cetearyl Alcohol, Glyceryl Stearate, Butyrospermum Parkii Butter, Calendula Officinalis Extract, Helianthus Annuus Seed Oil, Rosa Canina Fruit Oil, Phospholipids, Parfum , Xanthan Gum, Citrus Grandis Seed Extract, Lactic Acid, Glycine Soja Sterols, Silica, Tocopherol
Other information
- protect this product from excessive heat and direct sun
Questions or comments?
UK-0844-800-8399
US-310-272-7275

-
INGREDIENTS AND APPEARANCE
THE ORGANIC PHARMACY CELLULAR PROTECTION SUNSCREEN SPF 30
titanium dioxide and zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69303-630 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 1.0000 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 10.0000 g in 100 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) 40.0000 g in 100 g WATER (UNII: 059QF0KO0R) 12.7760 g in 100 g MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) 10.0000 g in 100 g ALCOHOL (UNII: 3K9958V90M) 10.0000 g in 100 g GLYCERIN (UNII: PDC6A3C0OX) 5.0760 g in 100 g DOCOSANOL (UNII: 9G1OE216XY) 2.1150 g in 100 g CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) 2.0000 g in 100 g GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) 1.5750 g in 100 g SHEA BUTTER (UNII: K49155WL9Y) 1.0000 g in 100 g CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) 1.0000 g in 100 g SUNFLOWER OIL (UNII: 3W1JG795YI) 1.0000 g in 100 g ROSA CANINA FRUIT OIL (UNII: CR7307M3QZ) 1.0000 g in 100 g OMEGA-3 FATTY ACIDS (UNII: 71M78END5S) 0.6750 g in 100 g XANTHAN GUM (UNII: TTV12P4NEE) 0.2000 g in 100 g LACTIC ACID (UNII: 33X04XA5AT) 0.1800 g in 100 g SOY STEROL (UNII: PL360EPO9J) 0.1350 g in 100 g CITRUS MAXIMA SEED (UNII: 083X55C543) 0.1180 g in 100 g SILICON DIOXIDE (UNII: ETJ7Z6XBU4) 0.1000 g in 100 g TOCOPHEROL (UNII: R0ZB2556P8) 0.0500 g in 100 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69303-630-01 1 in 1 CARTON 07/15/2014 1 150 g in 1 TUBE; Type 9: Other Type of Part 3 Combination Product (e.g., Drug/Device/Biological Product) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 07/15/2014 THE ORGANIC PHARMACY CELLULAR PROTECTION SUNSCREEN SPF 50
titanium dioxide and zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69303-650 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 1.0000 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 12.5000 g in 100 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) 40.0000 g in 100 g MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) 12.5000 g in 100 g ALCOHOL (UNII: 3K9958V90M) 10.0000 g in 100 g WATER (UNII: 059QF0KO0R) 7.7760 g in 100 g GLYCERIN (UNII: PDC6A3C0OX) 5.0760 g in 100 g DOCOSANOL (UNII: 9G1OE216XY) 2.1150 g in 100 g CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) 2.0000 g in 100 g GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) 1.5750 g in 100 g SHEA BUTTER (UNII: K49155WL9Y) 1.0000 g in 100 g CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) 1.0000 g in 100 g SUNFLOWER OIL (UNII: 3W1JG795YI) 1.0000 g in 100 g ROSA CANINA FRUIT OIL (UNII: CR7307M3QZ) 1.0000 g in 100 g OMEGA-3 FATTY ACIDS (UNII: 71M78END5S) 0.6750 g in 100 g XANTHAN GUM (UNII: TTV12P4NEE) 0.2000 g in 100 g LACTIC ACID (UNII: 33X04XA5AT) 0.1800 g in 100 g SOY STEROL (UNII: PL360EPO9J) 0.1350 g in 100 g CITRUS MAXIMA SEED (UNII: 083X55C543) 0.1180 g in 100 g SILICON DIOXIDE (UNII: ETJ7Z6XBU4) 0.1000 g in 100 g TOCOPHEROL (UNII: R0ZB2556P8) 0.0500 g in 100 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69303-650-01 1 in 1 CARTON 07/15/2014 1 150 g in 1 TUBE; Type 9: Other Type of Part 3 Combination Product (e.g., Drug/Device/Biological Product) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 07/15/2014 Labeler - The organic pharmacy Limited (220198688) Registrant - The organic pharmacy Limited (220198688) Establishment Name Address ID/FEI Business Operations The Organic Pharmacy Limited 220198688 manufacture(69303-630, 69303-650)