Label: LAXATIVE PILLS MAXIMUM STRENGTH- sennosides tablet
- NDC Code(s): 42507-492-24
- Packager: Hy-Vee
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 10, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
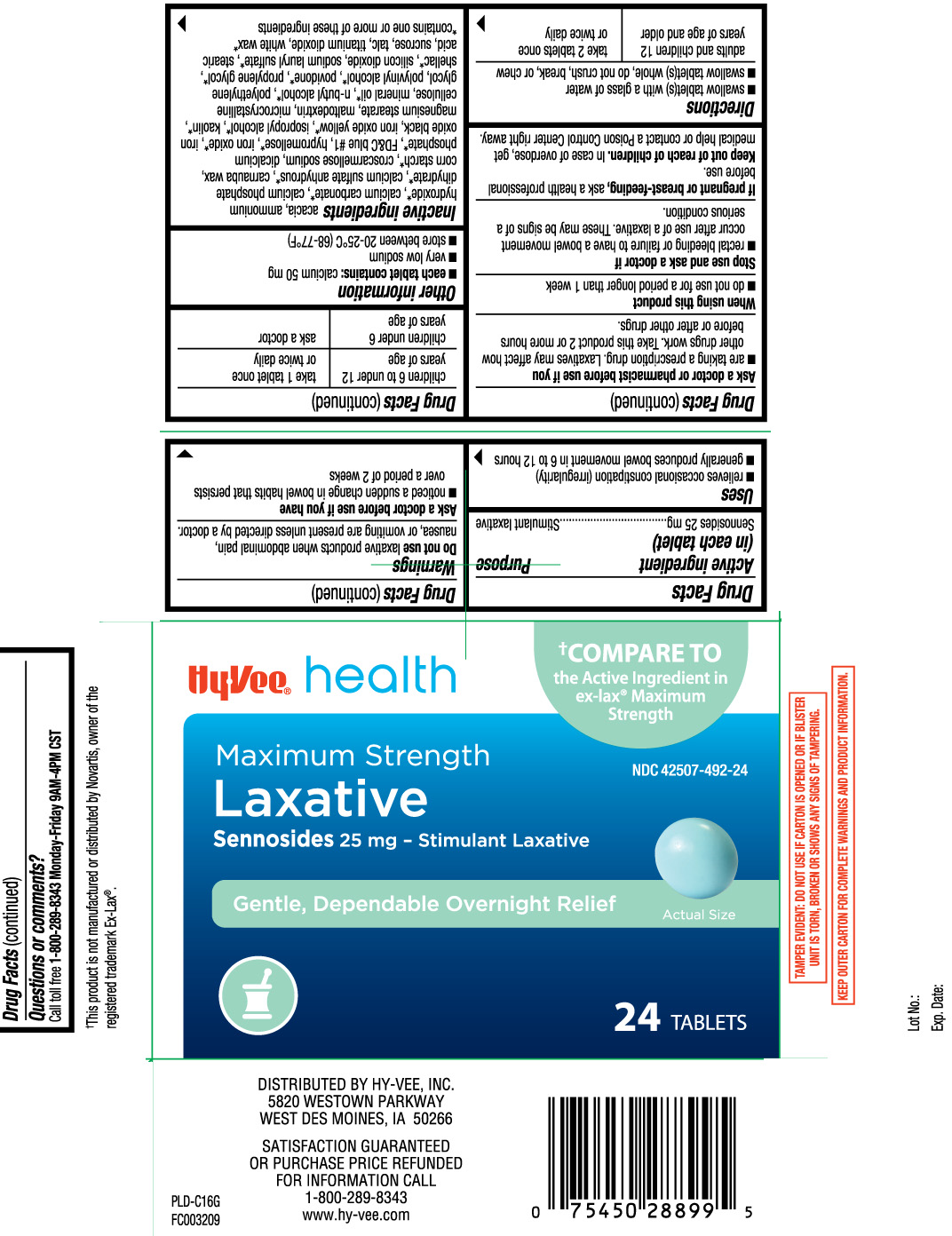
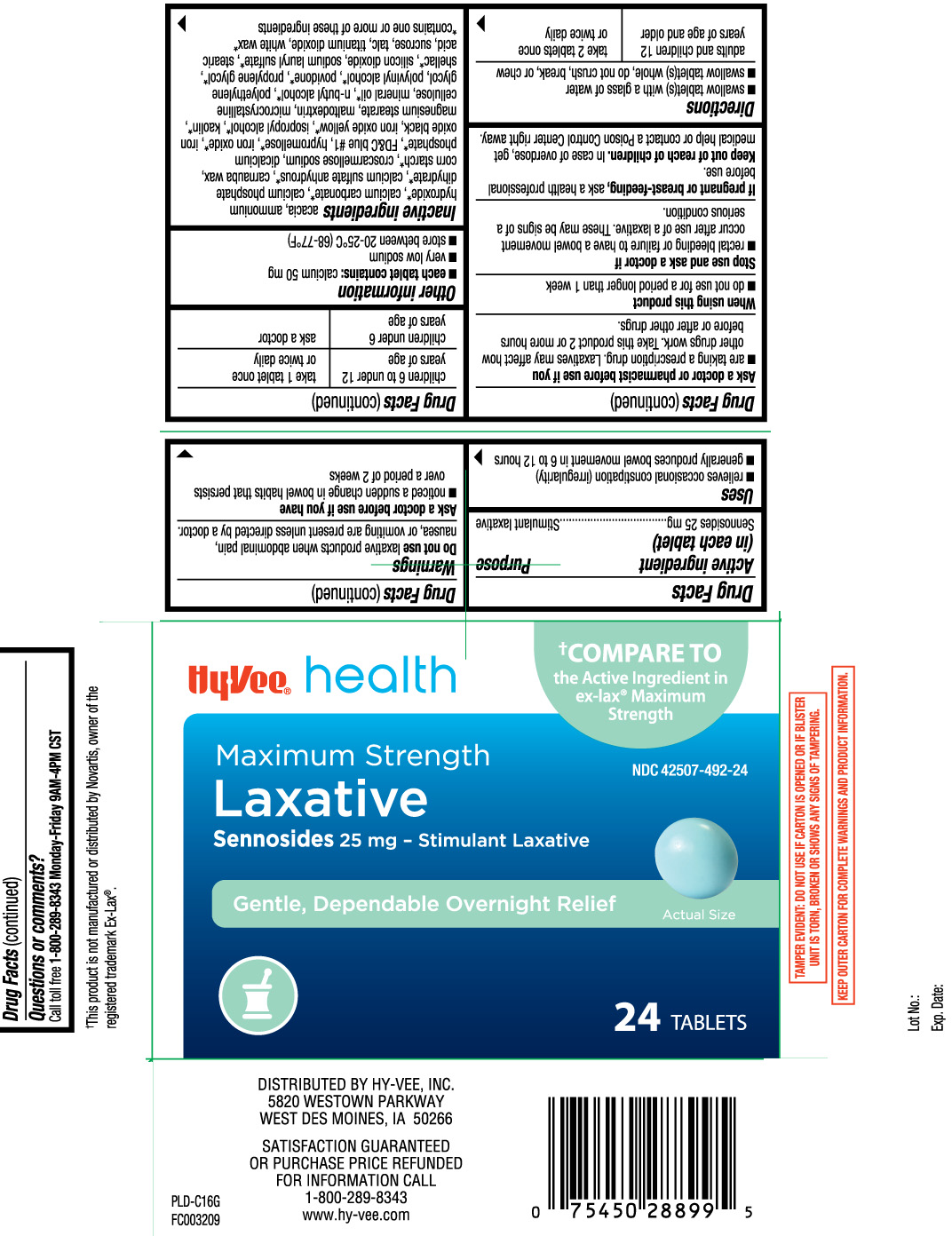
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Do not use
laxative products when abdominal pain, nausea, or vomiting are present unless directed by a doctor.
Ask a doctor before use if you have
- noticed a sudden change in bowel habits that persists over a period of 2 weeks
Ask a doctor or pharmacist before use if you
- are taking a prescription drug. Laxatives may affect how other drugs work. Take this product 2 or more hours before or after other drugs.
- Directions
- Other information
-
Inactive ingredients
acacia, ammonium hydroxide*, calcium carbonate*, calcium phosphate dihydrate*, calcium sulfate anhydrous*, carnauba wax, corn starch*, croscarmellose sodium, dicalcium phosphate*, FD&C blue #1 aluminum lake, hypromellose*, iron oxide*, iron oxide black, iron oxide yellow*, isopropyl alcohol*, kaolin*, magnesium stearate, Maltodextrin, microcrystalline cellulose, mineral oil*, n-butyl alcohol*, polyethylene glycol, polyvinyl alcohol*, povidone*, propylene glycol*, shellac, silicon dioxide*, sodium lauryl sulfate*, stearic acid*, sucrose, talc, titanium dioxide, white wax*
*contains one or more of these ingredients
- Questions or comments?
-
Principal Display Panel
†COMPARE TO the active ingredient in ex-lax® Maximum Strength
Maximum Strength
Laxative
Sennosides 25 mg - Stimulant Laxative
Gentle, Dependable Overnight Relief
Tablets
This product is not manufactured or distributed by Novartis, owner of the registered trademark Ex-Lax®.
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
DISTRIBUTED BY HY-VEE, INC.
5820 WESTOWN PARKWAY
WEST DES MOINES, IA 50266
- Product Label
-
INGREDIENTS AND APPEARANCE
LAXATIVE PILLS MAXIMUM STRENGTH
sennosides tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:42507-492 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SENNOSIDES (UNII: 3FYP5M0IJX) (SENNOSIDES - UNII:3FYP5M0IJX) SENNOSIDES 25 mg Inactive Ingredients Ingredient Name Strength ACACIA (UNII: 5C5403N26O) AMMONIA (UNII: 5138Q19F1X) CALCIUM PHOSPHATE, DIBASIC, DIHYDRATE (UNII: O7TSZ97GEP) CALCIUM SULFATE ANHYDROUS (UNII: E934B3V59H) CARNAUBA WAX (UNII: R12CBM0EIZ) STARCH, CORN (UNII: O8232NY3SJ) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) ALUMINUM OXIDE (UNII: LMI26O6933) HYPROMELLOSES (UNII: 3NXW29V3WO) FERRIC OXIDE RED (UNII: 1K09F3G675) FERROSOFERRIC OXIDE (UNII: XM0M87F357) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) ISOPROPYL ALCOHOL (UNII: ND2M416302) KAOLIN (UNII: 24H4NWX5CO) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) STEARIC ACID (UNII: 4ELV7Z65AP) SUCROSE (UNII: C151H8M554) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) WHITE WAX (UNII: 7G1J5DA97F) CALCIUM CARBONATE (UNII: H0G9379FGK) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) MALTODEXTRIN (UNII: 7CVR7L4A2D) MINERAL OIL (UNII: T5L8T28FGP) Product Characteristics Color blue Score no score Shape ROUND Size 11mm Flavor Imprint Code TCL083;S25 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42507-492-24 24 in 1 CARTON 02/29/2016 06/30/2024 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M007 02/29/2016 06/30/2024 Labeler - Hy-Vee (006925671)