Label: ABRYSVO- respiratory syncytial virus vaccine kit
ABRYSVO- respiratory syncytial virus vaccine injection, powder, lyophilized, for solution
-
NDC Code(s):
0069-0207-01,
0069-0250-01,
0069-0344-01,
0069-0344-05, view more0069-0344-10, 0069-0344-21, 0069-2465-01, 0069-2465-10, 0069-2465-19
- Packager: Pfizer Laboratories Div Pfizer Inc
- Category: VACCINE LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated December 23, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ABRYSVO safely and effectively. See full prescribing information for ABRYSVO.
ABRYSVO® (Respiratory Syncytial Virus Vaccine) for injection, for intramuscular use
Initial U.S. Approval: 2023RECENT MAJOR CHANGES
Warnings and Precautions, Guillain‑Barré Syndrome (5.1)
1/2025
INDICATIONS AND USAGE
ABRYSVO is a vaccine indicated for
- •
- Active immunization of pregnant individuals at 32 through 36 weeks gestational age for the prevention of lower respiratory tract disease (LRTD) and severe LRTD caused by respiratory syncytial virus (RSV) in infants from birth through 6 months of age. (1.1)
- •
- Active immunization for the prevention of LRTD caused by RSV in individuals 60 years of age and older. (1.2)
- •
- Active immunization for the prevention of LRTD caused by RSV in individuals 18 through 59 years of age who are at increased risk for LRTD caused by RSV. (1.3)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
For injection.
CONTRAINDICATIONS
History of severe allergic reaction (e.g., anaphylaxis) to any component of ABRYSVO. (4)
WARNINGS AND PRECAUTIONS
- •
- Guillain‑Barré Syndrome. The results of a postmarketing observational study suggest an increased risk of Guillain‑Barré syndrome during the 42 days following vaccination with ABRYSVO. (5.1, 6.2)
- •
- Potential risk of preterm birth. To avoid the potential risk of preterm birth with use of ABRYSVO before 32 weeks of gestation, administer ABRYSVO as indicated in pregnant individuals at 32 through 36 weeks gestational age. (5.2)
ADVERSE REACTIONS
- •
- The most commonly reported solicited local and systemic adverse reactions in pregnant individuals (≥10%) were pain at the injection site (40.6%), headache (31.0%), muscle pain (26.5%), and nausea (20.0%). (6.1)
- •
- The most commonly reported solicited local and systemic adverse reactions in individuals 60 years of age and older (≥10%) were fatigue (15.7%), headache (12.9%), pain at the injection site (10.6%), and muscle pain (10.2%). (6.1)
- •
- The most commonly reported solicited local and systemic adverse reactions in individuals 18 through 59 years of age (≥10%) were pain at the injection site (35.3%), muscle pain (24.4%), joint pain (12.4%), and nausea (11.8%). (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Pfizer Inc. at 1-800-438-1985 or VAERS at 1-800-822-7967 or http://vaers.hhs.gov.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 12/2025
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Immunization of Pregnant Individuals
1.2 Immunization of Individuals 60 Years of Age and Older
1.3 Immunization of Individuals 18 Through 59 Years of Age
2 DOSAGE AND ADMINISTRATION
2.1 Dose and Schedule
2.2 Presentations and Reconstitution
2.3 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Guillain-Barré Syndrome
5.2 Potential Risk of Preterm Birth
5.3 Management of Acute Allergic Reactions
5.4 Syncope
5.5 Altered Immunocompetence
5.6 Limitations of Vaccine Effectiveness
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Immunocompromised Individuals 18 Years of Age and Older
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Study in Pregnant Individuals for Efficacy in Their Infants from Birth Through 6 Months of Age
14.2 Efficacy in Individuals 60 Years of Age and Older
14.3 Immunogenicity in Individuals 18 Through 59 Years of Age Considered to be at Increased Risk of LRTD caused by RSV
14.4 Concomitant Administration of ABRYSVO with a Seasonal Inactivated Influenza Vaccine
14.5 Concomitant Administration of ABRYSVO with Tetanus Toxoid, Reduced Diphtheria Toxoid and Acellular Pertussis Vaccine, Adsorbed
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Immunization of Pregnant Individuals
ABRYSVO is a vaccine indicated for active immunization of pregnant individuals at 32 through 36 weeks gestational age for the prevention of lower respiratory tract disease (LRTD) and severe LRTD caused by respiratory syncytial virus (RSV) in infants from birth through 6 months of age.
-
2 DOSAGE AND ADMINISTRATION
2.1 Dose and Schedule
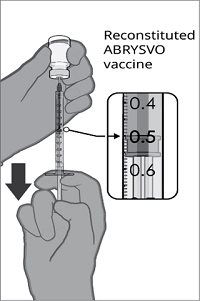
After reconstitution, a single dose of ABRYSVO is either 0.5 mL (Act-O-Vial presentation and vial and vial presentation) or approximately 0.5 mL (vial and prefilled syringe presentation) [see Dosage and Administration (2.2)].
2.2 Presentations and Reconstitution
ABRYSVO is supplied in 3 presentations as follows:
Act-O-Vial Presentation
The Act-O-Vial presentation is supplied in cartons. Each Act-O-Vial contains the Lyophilized Antigen Component (a sterile white powder) and Sterile Water Diluent Component.
Vial and Prefilled Syringe Presentation
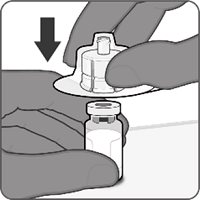
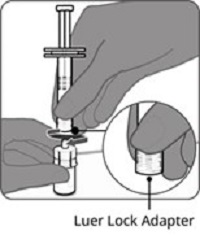
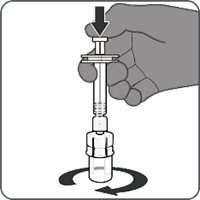
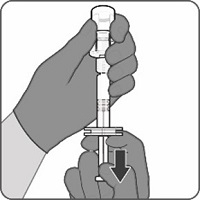
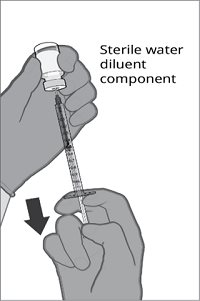
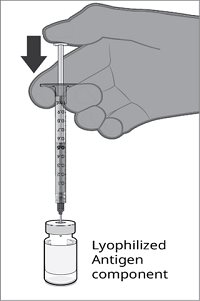
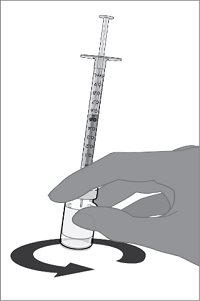
The vial and prefilled syringe presentation is supplied in cartons containing a kit(s). Each kit includes a vial of Lyophilized Antigen Component (a sterile white powder), a prefilled syringe containing Sterile Water Diluent Component, and a vial adapter.
Vial and Vial Presentation
The vial and vial presentation is supplied in cartons that include vials of Lyophilized Antigen Component (a sterile white powder) and vials containing Sterile Water Diluent Component.
For all presentations, reconstitute the Lyophilized Antigen Component with the Sterile Water Diluent Component to form ABRYSVO, as described in the instructions below.
Reconstitution Instructions for the Act-O-Vial Presentation
Reconstitution Instructions for Vial and Prefilled Syringe Presentation
Reconstitution Instructions for the Vial and Vial Presentation
2.3 Administration
For intramuscular use.
After reconstitution, ABRYSVO is a clear and colorless solution. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard if either condition is present.
Administer ABRYSVO immediately or store at room temperature [15°C to 30°C (59°F to 86°F)] and use within 4 hours. Discard reconstituted vaccine if not used within 4 hours.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Do not administer ABRYSVO to individuals with a history of a severe allergic reaction (e.g., anaphylaxis) to any component of ABRYSVO [see Description (11)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Guillain-Barré Syndrome
The results of a postmarketing observational study suggest an increased risk of Guillain‑Barré syndrome (GBS) during the 42 days following vaccination with ABRYSVO [see Adverse Reactions (6.2)].
5.2 Potential Risk of Preterm Birth
A numerical imbalance in preterm births in ABRYSVO recipients was observed compared to placebo recipients in two clinical studies [see Adverse Reactions (6.1)]. Available data are insufficient to establish or exclude a causal relationship between preterm birth and ABRYSVO. To avoid the potential risk of preterm birth with use of ABRYSVO before 32 weeks of gestation, administer ABRYSVO as indicated in pregnant individuals at 32 through 36 weeks gestational age. Pregnant individuals who were at increased risk of preterm birth were generally excluded from clinical studies of ABRYSVO.
5.3 Management of Acute Allergic Reactions
Appropriate medical treatment used to manage immediate allergic reactions must be immediately available in the event an anaphylactic reaction occurs following administration of ABRYSVO.
5.4 Syncope
Syncope (fainting) may occur in association with administration of injectable vaccines, including ABRYSVO. Procedures should be in place to avoid injury from fainting.
-
6 ADVERSE REACTIONS
In pregnant individuals, the most commonly reported (≥10%) adverse reactions were pain at the injection site (40.6%), headache (31.0%), muscle pain (26.5%), and nausea (20.0%).
In individuals 60 years of age and older, the most commonly reported (≥10%) adverse reactions were fatigue (15.7%), headache (12.9%), pain at the injection site (10.6%), and muscle pain (10.2%).
In individuals 18 through 59 years of age with chronic medical conditions, the most commonly reported (≥10%) adverse reactions and for which the rate for ABRYSVO exceeds the rate for placebo were pain at the injection site (35.3%), muscle pain (24.4%), joint pain (12.4%), and nausea (11.8%).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a vaccine cannot be directly compared to rates in the clinical trials of another vaccine and may not reflect the rates observed in practice.
Pregnant Individuals and Infants from Birth Through 6 Months of Age
The safety of ABRYSVO in maternal and infant participants was evaluated in two clinical studies in which approximately 4,000 maternal participants received a single dose of ABRYSVO.
Study 1 (NCT04424316) was a Phase 3, randomized, double-blind, multicenter, placebo-controlled study to investigate the efficacy and safety of ABRYSVO administered to pregnant individuals ≤49 years of age with uncomplicated, singleton pregnancies, to protect their infants against RSV disease. Pregnant individuals with high-risk pregnancies were excluded from the study (BMI>40 kg/m2 prior to pregnancy, pregnancies resulting after in vitro fertilization, preeclampsia, eclampsia, uncontrolled gestational hypertension, placental abnormalities, polyhydramnios or oligohydramnios, significant bleeding or blood clotting disorder, unstable endocrine disorders including untreated disorders of glucose intolerance or thyroid disorders). Pregnant individuals with prior pregnancy complications (e.g., history of preterm birth ≤34 weeks gestation, prior stillbirth, neonatal death, previous infant with a known genetic disorder or significant congenital anomaly) could be included, based on the investigators’ judgment, but were generally not enrolled in the study. In this study with 1:1 randomization 3,698 participants received ABRYSVO and 3,687 received placebo (0.5 mL dose, containing the same buffer ingredients in the same quantities as in a single dose of ABRYSVO [see Description (11)]). Infants born in year 1 were followed for up to 24 months, and infants born in year 2 were followed for up to 12 months to assess safety. At the time of final data evaluation, 3,659 infants were born to the maternal participants in the ABRYSVO group and 3,646 in the placebo group, and of these, approximately 93.3% have been followed for 12 months and 86.7% for 24 months. This multicenter study was conducted in Argentina, Australia, Brazil, Canada, Chile, Denmark, Finland, Gambia, Japan, Republic of Korea, Mexico, Netherlands, New Zealand, Philippines, South Africa, Spain, Taiwan, and the US.
Demographic characteristics in Study 1 among participants who received ABRYSVO and those who received placebo were generally similar with regard to age, race, and ethnicity. Of the participants in the study, 65% were White, 20% were Black or African American, 13% were Asian, and 29% were Hispanic/Latino. The median maternal age at the time of study vaccination was 29 years (range 16 to 45 years in the ABRYSVO group, 14 to 47 years in the placebo group). The median gestational age at vaccination was 31 weeks and 2 days (range 24-36.9 weeks). ABRYSVO is approved for use for pregnant individuals at 32 through 36 weeks gestational age [see Indications and Usage (1.1)]. The median infant gestational age at birth was 39 weeks and 1 day (range 27 weeks and 3 days to 44 weeks and 2 days). Among the infants born to maternal participants 51% were male and 49% were female.
Study 2 (NCT04032093) was a Phase 2, randomized, placebo-controlled, observer-blinded study that investigated the safety of two dose levels (120 mcg and a higher dose) of ABRYSVO administered to pregnant individuals. ABRYSVO (120 mcg) was administered to 115 maternal participants, and 114 infants were born to these maternal participants. This study was conducted in the US, South Africa, Argentina, and Chile. Demographic characteristics among participants who received ABRYSVO and those who received placebo were generally similar with regard to age, race, and ethnicity. Of the participants in the study, 76% were White, 21% were Black or African American, and 28% were Hispanic/Latino. The median age of participants was 27 years (range 18-42 years). The median gestational age at vaccination was 30 weeks (range 24-36 weeks). ABRYSVO is approved for use for pregnant individuals at 32 through 36 weeks gestational age [see Indications and Usage (1.1)].
For all maternal participants in Study 1, solicited local reactions and systemic events were collected using electronic diaries for 7 days after study vaccination, adverse events for 1 month and obstetric complications, serious adverse events, and adverse events of special interest for the duration of the study. For infant participants, the collection period for nonserious adverse events was from birth to 1 month. Serious adverse events were monitored for at least 1 year for all infant participants and for up to 2 years for half of the infants in Study 1.
Solicited Local and Systemic Reactions in Study 1
The majority of solicited local and systemic reactions in maternal participants resolved within 2-3 days of onset. Severe local reactions were reported for 0.3% of maternal participants in the ABRYSVO group and none in the placebo group, and severe systemic reactions within 7 days after vaccination were reported by 2.3% of maternal participants in both groups.
Solicited local and systemic reactions reported within 7 days after vaccination in Study 1 are presented in Tables 1 and 2.
Table 1 Percentage of Maternal Participants with Local Reactions Reported, by Maximum Severity, within 7 Days after Vaccination – Study 1* Note: The presented results are based on the participants with solicited local reactogenicity data at the time of the data cut-off date for the primary analysis. - *
- NCT04424316
- †
- N = number of participants who provided e-diary data for a specific reaction after vaccination.
- ‡
- Mild: does not interfere with activity; moderate: interferes with activity; severe: prevents daily activity.
- §
- Any includes all participants who reported a reaction as mild, moderate, or severe during Day 1 to Day 7 after vaccination.
- ¶
- Mild: >2 cm to 5 cm; moderate: >5 cm to 10 cm; severe: >10 cm.
Local Reactions
ABRYSVO
N=3,663†
%
PLACEBO
N=3,639†
%
Injection site pain‡
Any§
40.6
10.1
Mild
36.1
9.3
Moderate
4.4
0.9
Severe
0.1
0
Redness¶
Any§
7.2
0.2
Mild
5.0
0.1
Moderate
2.1
0.1
Severe
0.1
0
Swelling¶
Any§
6.2
0.2
Mild
4.1
0.1
Moderate
2.0
<0.1
Severe
<0.1
0
Table 2 Percentage of Maternal Participants with Systemic Reactions Reported, by Maximum Severity, within 7 Days after Vaccination – Study 1* Note: The presented results are based on the participants with solicited systemic reactogenicity data at the time of the data cut-off date for the primary analysis. - *
- NCT04424316
- †
- N = number of participants who provided e-diary data for a specific reaction after vaccination.
- ‡
- Mild: does not interfere with activity; moderate: some interference with activity; severe: prevents daily routine activity.
- §
- Any includes all participants who reported a reaction as mild, moderate, or severe during Day 1 to Day 7 after vaccination.
- ¶
- Mild: 2 to 3 loose stools in 24 hours; moderate: 4 to 5 loose stools in 24 hours; severe: 6 or more loose stools in 24 hours.
- #
- Mild: 1 to 2 times in 24 hours; moderate: >2 times in 24 hours; severe: requires intravenous hydration.
Systemic Reactions
ABRYSVO
N=3,663†
%
PLACEBO
N=3,638-3,639†
%
Fever (≥38.0℃)
≥38.0°C
2.6
2.9
≥38.0°C to 38.4°C
1.7
1.5
>38.5°C to 38.9°C
0.8
1.2
>39.0°C to 40.0°C
<0.1
0.1
>40.0°C
<0.1
0.1
Fatigue‡
Any§
46.1
43.8
Mild
23.4
22.8
Moderate
21.4
19.6
Severe
1.3
1.4
Headache‡
Any§
31.0
27.6
Mild
20.2
17.9
Moderate
10.4
9.3
Severe
0.4
0.4
Muscle pain‡
Any§
26.5
17.1
Mild
17.6
10.0
Moderate
8.6
6.8
Severe
0.4
0.3
Nausea‡
Any§
20.0
19.2
Mild
14.4
13.8
Moderate
5.4
5.2
Severe
0.2
0.2
Joint pain‡
Any§
11.6
10.5
Mild
6.5
6.0
Moderate
4.9
4.4
Severe
0.2
<0.1
Diarrhea¶
Any
11.2
11.5
Mild
9.1
9.4
Moderate
2.0
1.9
Severe
0.1
0.2
Vomiting#
Any
7.8
7.0
Mild
6.4
5.4
Moderate
1.3
1.5
Severe
0.2
<0.1
Unsolicited Adverse Events in Study 1
Unsolicited adverse events reported within 1 month after vaccination by maternal participants were 14.0% in the ABRYSVO group and 13.2% in the placebo group.
The most frequently reported unsolicited adverse events in maternal participants from vaccination through the 1-month follow-up visit were disorders of pregnancy, puerperium and perinatal conditions (7.1% for the ABRYSVO group versus 6.3% for the placebo group).
Within 14 days of vaccination, lymphadenopathy was reported in 2 vaccine recipients and 0 placebo recipients and was considered related to ABRYSVO.
Serious Adverse Events in Study 1
In Study 1, serious adverse events in maternal participants were reported by 16.6% in the ABRYSVO group and 15.8% in the placebo group occurring any time during the study (see Table 3) with 4.3% serious adverse events in the ABRYSVO group and 3.8% in the placebo group occurring within 1 month after vaccination. Most of the serious adverse events in maternal participants were related to pregnancy complications and occurred after the 1 month period following vaccination.
Table 3 Select Pregnancy-related Serious Adverse Events in Study 1 in Pregnant Individuals Occurring at any Time Following Vaccination* - *
- Includes all SAEs from vaccination to 6 months post-delivery (up to approximately 10 months, depending on the gestational age at the time of vaccination). In Study 1, HELLP syndrome occurred in 5 participants (2 in the ABRYSVO group and 3 in the placebo group).
- †
- There was one maternal death in the ABRYSVO group due to postpartum hemorrhage that was not likely to be associated with vaccination.
- ‡
- A total of 19 intrauterine deaths were reported for the index pregnancy: 10 intrauterine deaths in the ABRYSVO group (0.3%) and 9 intrauterine deaths in the placebo group (0.2%). The intrauterine deaths represented various clinical conditions and presentations resulting in fetal demise without clear evidence of a common pathophysiology.
Serious Adverse Reaction
ABRYSVO
N=3,698
n (%)
95% CI
Placebo
N=3,687
n (%)
95% CI
All Maternal SAEs
613 (16.6)
(15.4, 17.8)
581 (15.8)
(14.6, 17.0)
Pre-eclampsia
67 (1.8)
(1.4, 2.3)
53 (1.4)
(1.1, 1.9)
Gestational hypertension
43 (1.2)
(0.8, 1.6)
41 (1.1)
(0.8, 1.5)
Eclampsia
7 (0.2)
(0.1, 0.4)
3 (<0.1)
(0.0, 0.2)
Premature rupture of membranes
14 (0.4)
(0.2, 0.6)
16 (0.4)
(0.2, 0.7)
Preterm premature rupture of membranes
15 (0.4)
(0.2, 0.7)
11 (0.3)
(0.1, 0.5)
Hypertension
13 (0.4)
(0.2, 0.6)
6 (0.2)
(0.1, 0.4)
Maternal death†
1 (<0.1)
(0.0, 0.2)
0
(0.0, 0.1)
Fetal death‡
10 (0.3)
(0.1, 0.5)
9 (0.2)
(0.1, 0.5)
Preterm Births in Study 1 and Study 2
A numerical imbalance in preterm births in ABRYSVO recipients compared to placebo recipients was observed in both Studies 1 and 2. In Study 2, preterm births occurred in 5.3% (6 out of 114) in the ABRYSVO group and 2.6% (3 out of 116) in the placebo group. In the subsequent Study 1, preterm birth events occurred in 5.7% [95% CI: 4.9, 6.5] (207 out of 3,659) in the ABRYSVO group and 4.7% [95% CI: 4.1, 5.5] (172 out of 3,646) in the placebo group. In infants born preterm, 84 infants in the ABRYSVO group and 81 infants in the placebo group remained hospitalized or were readmitted to the hospital in the neonatal period (up to 30 days after birth). Available data are insufficient to establish or exclude a causal relationship between preterm birth and ABRYSVO.
A numerical imbalance in preterm births was also observed in Study 1 among the subgroup of infants born to participants who were vaccinated at 32 through 36 weeks gestation, with 4.3% (71/1,667) in the ABRYSVO group and 3.7% (60/1,640) in the placebo group.
Adverse Reactions in Infants
In Study 1, adverse events in infants from birth to 1 month of age were observed in 38.0% in the ABRYSVO group compared to 35.4% in the placebo group. Low birth weight was observed in 5.1% of participants in the ABRYSVO group versus 4.3% in the placebo group, and neonatal jaundice was observed in 7.3% in the ABRYSVO group versus 6.9% in the placebo group.
Individuals 18 Years of Age and Older
Individuals 60 Years of Age and Older
The safety of ABRYSVO was evaluated in Study 3 (NCT05035212) in which 18,574 participants received ABRYSVO and 18,288 received placebo (0.5 mL dose, containing the same buffer ingredients in the same quantities as in a single dose of ABRYSVO [see Description (11)]). Study 3 was a multicenter, randomized, double-blind, placebo-controlled study to assess the efficacy and safety of ABRYSVO in individuals 60 years of age and older. This study was conducted in the US, Argentina, Japan, the Netherlands, Canada, South Africa, and Finland. Demographic characteristics among participants who received ABRYSVO and those who received placebo were generally similar with regard to age, sex, race, and ethnicity. Of the participants in the study, 51% were male and 80% were White, 12% were Black or African American, 7% were Asian, and 41% were Hispanic/Latino. The median age of participants was 67 years (range 59-97 years).
Solicited local and systemic reactions were collected using electronic diaries for 7 days after study vaccination in 7,116 participants (3,669 ABRYSVO participants and 3,447 placebo recipients) from a subset of sites. For all participants, unsolicited adverse events were collected for one month after study vaccination; serious adverse events (SAEs) are collected throughout study participation with a median duration of follow-up of 17.5 months.
Solicited Local and Systemic Reactions in Study 3
Solicited local and systemic reactions reported within 7 days after vaccination in Study 3 are presented in Tables 4 and 5.
Table 4 Percentage of Participants 60 Years of Age and Older with Local Reactions Reported, by Maximum Severity, within 7 Days after Vaccination – Study 3* Local Reactions ABRYSVO
N=3,628†
%PLACEBO
N=3,447†
%- *
- NCT05035212
- †
- N = number of participants who provided e-diary data for a specific reaction after vaccination.
- ‡
- Mild: does not interfere with activity; moderate: some interference with activity; severe: prevents daily activity.
- §
- Any includes all participants who reported a reaction as mild, moderate, or severe during Day 1 to Day 7 after vaccination.
- ¶
- Mild: 2.5 cm to 5 cm; moderate: >5 cm to 10 cm; severe: >10 cm (for data reported from e-diaries).
Injection site pain‡
Any§
10.6
6.1
Mild
9.5
5.4
Moderate
1.1
0.7
Severe
<0.1
0
Any§
2.7
0.6
Mild
1.6
0.4
Moderate
1.0
0.2
Severe
0.1
0
Any§
2.5
0.4
Mild
1.5
0.2
Moderate
0.9
0.1
Severe
0.1
<0.1
Table 5 Percentage of Participants 60 Years of Age and Older with Systemic Reactions Reported, by Maximum Severity, within 7 Days after Vaccination – Study 3* Systemic Reactions ABRYSVO
N=3,628†
%PLACEBO
N=3,447†
%- *
- NCT05035212
- †
- N = number of participants who provided e-diary data for a specific reaction after vaccination.
- ‡
- Mild: does not interfere with activity; moderate: some interference with activity; severe: prevents daily routine activity.
- §
- Any includes all participants who reported a reaction as mild, moderate, or severe during Day 1 to Day 7 after vaccination.
- ¶
- Mild: 1 to 2 times in 24 hours; moderate: >2 times in 24 hours; severe: requires intravenous hydration.
- #
- Mild: 2 to 3 loose stools in 24 hours; moderate: 4 to 5 loose stools in 24 hours; severe: 6 or more loose stools in 24 hours.
Fever (≥38.0℃)
≥38.0°C
1.4
1.5
≥38.0°C to 38.4°C
0.6
0.8
>38.4°C to 38.9°C
0.8
0.6
>38.9°C to 40.0°C
<0.1
<0.1
>40.0°C
0
<0.1
Fatigue‡
Any§
15.7
14.8
Mild
9.3
8.6
Moderate
6.0
6.0
Severe
0.3
0.1
Headache‡
Any§
12.9
12.0
Mild
9.0
8.6
Moderate
3.8
3.2
Severe
0.1
<0.1
Muscle pain‡
Any§
10.2
8.5
Mild
6.5
5.6
Moderate
3.5
2.8
Severe
0.2
<0.1
Joint pain‡
Any§
7.6
7.0
Mild
4.5
3.9
Moderate
3.0
3.0
Severe
<0.1
<0.1
Nausea‡
Any§
3.5
3.8
Mild
2.6
3.1
Moderate
0.9
0.6
Severe
0
<0.1
Vomiting¶
Any§
0.9
0.9
Mild
0.7
0.7
Moderate
0.2
0.2
Severe
0
<0.1
Diarrhea#
Any§
6.0
5.3
Mild
4.5
4.3
Moderate
1.4
0.9
Severe
0.1
0.1
Solicited local and systemic reactions had a median duration of 1-2 days.
Unsolicited Adverse Events in Study 3
Unsolicited adverse events occurring within 1 month after vaccination were similar between groups, reported in 10.8% and 10.5% of participants who received ABRYSVO and placebo, respectively.
Within 30 days after vaccination, atrial fibrillation was reported in 11 vaccine recipients and 3 placebo recipients (of which 5 in the ABRYSVO group and 2 in the placebo group were serious adverse events); the onset of symptoms was 18 to 30 days post vaccination. The currently available information on atrial fibrillation is insufficient to determine a causal relationship to the vaccine [see Use in Specific Populations (8.6)]. Within 14 days of vaccination, lymphadenopathy was reported in 6 vaccine recipients, considered related to ABRYSVO, and 3 placebo recipients. There were no other notable patterns or numerical imbalances between groups for specific categories of unsolicited adverse events.
Serious Adverse Events in Study 3
In Study 3, SAEs were reported by 6.2% of participants in the ABRYSVO group and 6.1% in the placebo group. Three participants in the ABRYSVO group had SAEs which were assessed as possibly related to study vaccination: Guillain‑Barré syndrome reported 7 days after vaccination (with diagnosis later revised to chronic inflammatory demyelinating polyneuropathy), Miller Fisher syndrome reported 8 days after vaccination, and hypersensitivity reported 8 hours after vaccination.
Individuals 18 through 59 Years of Age Considered to be at Increased Risk of LRTD caused by RSV
The safety of ABRYSVO was evaluated in Study 4 Substudy A (NCT05842967) in which 453 participants received ABRYSVO and 225 received placebo (0.5 mL dose, containing the same buffer ingredients in the same quantities as in a single dose of ABRYSVO [see Description (11)]). Study 4 was a multicenter, randomized, double-blind, placebo-controlled study to assess the safety and immunogenicity of ABRYSVO in individuals 18 through 59 years of age considered to be at increased risk of LRTD caused by RSV due to certain chronic medical conditions [see Clinical Studies (14.3)]. This study was conducted in the US. Demographic characteristics among individuals who received ABRYSVO and those who received placebo were generally similar with regard to age, race, and ethnicity; 43% and 32% of participants in the ABRYSVO and placebo groups, respectively, were male. Of the participants in the study, 68% were White, 24% were Black or African American, 5% were Asian, and 22% were Hispanic/Latino. Fifty-two percent (52%) were 18 to 49 years and 48% were 50 to 59 years. The median age of participants was 49 years. The vaccine and placebo groups were similar with regard to the prevalence of underlying medical conditions: one or more chronic pulmonary condition (52%), diabetes (43%), one or more other disease (liver, renal, neurologic, hematologic, or other metabolic disease) (31%), and one or more cardiovascular condition (8%).
Solicited local and systemic adverse reactions that occurred within 7 days following study vaccination were self‑reported in electronic diaries or were reported to an investigator. Unsolicited adverse events were collected for 1 month after study vaccination; serious adverse events (SAEs) were collected for 6 months after study vaccination.
Solicited Local and Systemic Reactions in Study 4
Solicited local and systemic reactions reported within 7 days after vaccination in Study 4 are presented in Tables 6 and 7.
Table 6 Percentage of Participants 18 through 59 Years of Age at Increased Risk of LRTD caused by RSV with Local Reactions Reported, by Maximum Severity, within 7 Days after Vaccination – Study 4* - *
- NCT05842967
- †
- N = number of participants reporting at least one response in the e-diary.
- ‡
- Mild: does not interfere with activity; moderate: some interference with activity; severe: prevents daily activity.
- §
- Any includes all participants who reported a reaction as mild, moderate, or severe during Day 1 to Day 7 after vaccination.
- ¶
- Mild: >2 cm to 5 cm; moderate: >5 cm to 10 cm; severe: >10 cm (for data reported from e-diaries).
Local Reactions
ABRYSVO
N=451†
%
PLACEBO
N=225†
%
Injection site pain‡
Any§
35.3
10.7
Mild
29.7
10.2
Moderate
5.5
0.4
Severe
0
0
Redness¶
Any§
6.0
0.4
Mild
3.8
0
Moderate
2.2
0.4
Severe
0
0
Swelling¶
Any§
7.1
0.9
Mild
4.0
0.4
Moderate
2.9
0.4
Severe
0.2
0
Table 7 Percentage of Participants 18 through 59 Years of Age at Increased Risk of LRTD caused by RSV with Systemic Reactions Reported, by Maximum Severity, within 7 Days after Vaccination – Study 4* - *
- NCT05842967
- †
- N = number of participants reporting at least one response in the e-diary.
- ‡
- Mild: does not interfere with activity; moderate: some interference with activity; severe: prevents daily routine activity.
- §
- Any includes all participants who reported a reaction as mild, moderate, or severe during Day 1 to Day 7 after vaccination.
- ¶
- Mild: 1 to 2 times in 24 hours; moderate: >2 times in 24 hours; severe: requires intravenous hydration.
- #
- Mild: 2 to 3 loose stools in 24 hours; moderate: 4 to 5 loose stools in 24 hours; severe: 6 or more loose stools in 24 hours.
Systemic Reactions
ABRYSVO
N=451†
%
PLACEBO
N=225†
%
Fever (≥38.0℃)
≥38.0°C
1.6
1.3
≥38.0°C to 38.4°C
0.4
0.4
>38.4°C to 38.9°C
1.1
0.9
>38.9°C to 40.0°C
0
0
Fatigue‡
Any§
37.3
38.2
Mild
18.2
22.2
Moderate
18.2
15.6
Severe
0.9
0.4
Headache‡
Any§
28.4
30.2
Mild
20.8
18.7
Moderate
7.3
11.6
Severe
0.2
0
Muscle pain‡
Any§
24.4
16.0
Mild
15.7
9.8
Moderate
8.6
6.2
Severe
0
0
Joint pain‡
Any§
12.4
10.2
Mild
7.1
4.0
Moderate
5.1
6.2
Severe
0.2
0
Nausea‡
Any§
11.8
10.2
Mild
9.3
8.9
Moderate
2.4
0.9
Severe
0
0.4
Vomiting¶
Any§
2.0
1.3
Mild
1.6
0.4
Moderate
0.4
0.9
Severe
0
0
Diarrhea#
Any§
14.9
16.9
Mild
11.1
12.4
Moderate
3.1
3.6
Severe
0.7
0.9
Solicited local and systemic reactions had a median duration of 1-2 days.
Unsolicited Adverse Events in Study 4
Unsolicited adverse events occurring within 1 month after vaccination were reported in 7.1% and 7.6% of participants who received ABRYSVO and placebo, respectively. One case of urticaria occurred on the same day as vaccine administration and was considered related to ABRYSVO.
Serious Adverse Events in Study 4
In Study 4, SAEs were reported by 1.1% of participants in the ABRYSVO group and 3.1% in the placebo group. No SAEs were assessed as related to study vaccination.
Serious Adverse Events Reported from Other Studies
Anaphylaxis was reported in a participant enrolled in a study (NCT06473519) in the US, with onset of symptoms 10 minutes after vaccination with ABRYSVO. The study enrolled healthy female participants 18 years through 49 years of age (n=450).
Concomitant Administration of ABRYSVO with a Seasonal Inactivated Influenza Vaccine
Study 5 (NCT05301322) was a Phase 3, multicenter, parallel group, placebo-controlled, randomized, double-blind study conducted in Australia in adults ≥65 years of age. The study enrolled healthy adults and adults with stable chronic medical conditions not requiring significant change in therapy or hospitalization for worsening disease 6 weeks before enrollment. Participants were enrolled into one of two groups. Participants in the concomitant administration group (n=703) received ABRYSVO and Influenza Vaccine, Adjuvanted (FLUAD® QUADRIVALENT) concomitantly and placebo one month later. Participants in the sequential administration group (n=691) received FLUAD QUADRIVALENT and placebo concomitantly and ABRYSVO one month later.
Within 7 days following vaccine administration, fatigue was reported by 30.0% of participants who received ABRYSVO administered concomitantly with FLUAD QUADRIVALENT, 19.1% who received ABRYSVO administered alone, and 27.1% who received FLUAD QUADRIVALENT and placebo concomitantly. There were no other notable differences in reported solicited local and systemic adverse reactions within 7 days following ABRYSVO administered concomitantly with FLUAD QUADRIVALENT compared to ABRYSVO administered alone. Participants were followed for SAEs from administration of the first dose of vaccine through 1 month following the last vaccination. No SAEs were considered related to vaccination.
6.2 Postmarketing Experience
The following adverse reactions have been identified from spontaneous reports during postmarketing use of ABRYSVO. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to the vaccine.
Immune System Disorders: Anaphylaxis and other hypersensitivity reactions including rash and urticaria.
Nervous System Disorders: Guillain‑Barré syndrome.
Postmarketing Observational Study of the Risk of Guillain‑Barré Syndrome following Vaccination with ABRYSVO
The association between vaccination with ABRYSVO and Guillain‑Barré syndrome (GBS) was evaluated among Medicare beneficiaries 65 years of age and older. Using Medicare claims data, between May 2023 through July 2024, vaccinations with ABRYSVO were identified through Current Procedural Terminology (CPT)/Healthcare Common Procedure Coding System (HCPCS) codes and National Drug Codes, and potential cases of hospitalized GBS among recipients of ABRYSVO were identified through International Classification of Diseases (ICD) codes. GBS diagnoses in claims data were confirmed by medical record review when available.
The risk of GBS following vaccination with ABRYSVO was assessed in self-controlled case series analyses using a risk window of 1 to 42 days post-vaccination and a control window of 43 to 90 days post-vaccination. The analyses of all GBS cases based on claims data suggest an increased risk of GBS during the 42 days following vaccination with ABRYSVO, with an incidence rate ratio (GBS cases in the risk window/control window) of 2.02 (95% CI: 0.93, 4.40) and an estimated 9 excess cases of GBS per million doses administered to individuals 65 years of age and older. The background risk of GBS in a study population influences the excess GBS case estimate and may differ between studies, precluding direct comparison to excess GBS case estimates from other vaccine studies or populations.
The analyses of GBS diagnoses in claims data were supported by analyses of GBS cases confirmed by medical record review and by analyses of GBS cases in individuals who received ABRYSVO alone, without other concomitantly administered vaccines. While the results of this observational study suggest an increased risk of GBS with ABRYSVO, available evidence is insufficient to establish a causal relationship.
-
7 DRUG INTERACTIONS
In Study 6 (NCT04071158) in a concomitant administration study of ABRYSVO and a Tetanus Toxoid, Reduced Diphtheria Toxoid and Acellular Pertussis Vaccine, Adsorbed (Tdap) in non-pregnant women, no safety concerns were identified. Immune responses to RSV A, RSV B, diphtheria, and tetanus were non-inferior to those after separate administration. Lower geometric mean antibody concentrations (GMCs) to the acellular pertussis antigens (pertussis toxin [PT], filamentous hemagglutinin (FHA), and pertactin [PRN]) were measured when ABRYSVO was administered concomitantly with Tdap compared to pertussis GMCs when Tdap was administered alone [see Clinical Studies (14.5)].
Concomitant administration of Tdap with ABRYSVO in pregnant individuals has not been studied.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in individuals exposed to ABRYSVO during pregnancy. Individuals who received ABRYSVO during pregnancy are encouraged to contact, or have their healthcare provider contact, 1-800-616-3791 to enroll in or obtain information about the registry.
Risk Summary
All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects and miscarriages in clinically recognized pregnancies is 2% to 4%, and 15% to 20%, respectively, and the estimated background risk of fetal deaths after 20 weeks is 0.6%.
Study 1 enrolled 7,386 pregnant individuals who were randomized 1:1 and received ABRYSVO or placebo (0.5 mL dose, containing the same buffer ingredients in the same quantities as in a single dose of ABRYSVO [see Description (11)]) revealed no evidence for vaccine-associated increase in the risk of congenital anomalies or fetal deaths. Study 2 evaluated 115 pregnant individuals who received ABRYSVO and 117 who received placebo. A numerical imbalance in preterm births in ABRYSVO recipients was observed compared to placebo recipients in these two clinical studies. Available data are insufficient to establish or exclude a causal relationship between preterm birth and ABRYSVO [see Warnings and Precautions (5.2), Adverse Reactions (6.1), Clinical Considerations (8.1), Data (8.1) and Clinical Studies (14.1)].
A developmental toxicity study was performed in female rabbits administered a vaccine formulation containing two times the antigen content of a single human dose of ABRYSVO prior to and during gestation. The study showed no evidence of harm to the fetus or to postnatal survival, growth, or development (see Animal Data).
Maternal Adverse Reactions
In Study 1, 3,698 pregnant individuals received ABRYSVO and 3,687 received placebo. Local and systemic adverse reactions occurred with greater frequency in the ABRYSVO group. Serious adverse reactions observed in pregnant individuals at a higher rate in the ABRYSVO group compared to the placebo group included pre-eclampsia (1.8% versus 1.4%) and gestational hypertension (1.2% versus 1.1%) [see Adverse Reactions (6.1)].
ABRYSVO has not been studied in pregnant individuals less than 24 weeks gestational age, and those at increased risk for preterm birth.
Fetal/Neonatal Adverse Reactions
The infant safety population included 3,659 and 3,646 infants born to individuals in the ABRYSVO or placebo group, respectively. There were 10 (0.3%) fetal deaths in the ABRYSVO group and 9 (0.2%) in the placebo group. Among the infants born to individuals in the ABRYSVO group and in the placebo group, 207 (5.7%) and 172 (4.7%), respectively, were born preterm [see Warnings and Precautions (5.2), Adverse Reactions (6.1) and Clinical Studies (14.1)]. Low birth weight was observed in 5.1% of participants in the ABRYSVO group versus 4.3% in the placebo group, and neonatal jaundice was observed in 7.3% in the ABRYSVO group versus 6.9% in the placebo group. [see Adverse Reactions (6.1)]. For mortality in the neonatal period among infants born to pregnant individuals in Study 1, there were 3 deaths in the ABRYSVO group and 5 in the placebo group, and for overall mortality including after the neonatal period there were 8 deaths in the ABRYSVO group and 14 in the placebo group. Congenital abnormalities were reported in 5.6% in the ABRYSVO group and 6.7% in the placebo group.
Available data are insufficient to establish or exclude a causal relationship between preterm birth and ABRYSVO. To avoid the potential risk of preterm birth with use of ABRYSVO before 32 weeks of gestation, administer ABRYSVO as indicated in pregnant individuals at 32 through 36 weeks gestational age.
Data
Human Data
In Study 1, 3,698 pregnant individuals received ABRYSVO and 3,687 received placebo at 24 through 36 weeks’ gestation. The infant safety population included 3,659 and 3,646 infants born to individuals in the ABRYSVO or placebo group, respectively. Among the infants born to individuals in the ABRYSVO group and in the placebo group, 207 (5.7%) and 172 (4.7%), respectively, had adverse events of preterm birth and 205 (5.6%) and 245 (6.7%), respectively, had reported congenital malformations or anomalies. There were 10 (0.3%) fetal deaths in the ABRYSVO group and 9 (0.2%) in the placebo group.
A pre- and post-natal developmental toxicity study with an embryo-fetal developmental toxicity phase was performed in female New Zealand White rabbits. Rabbits were administered 4 doses by intramuscular injection: at 3 weeks and at 1 week prior to mating, and on gestation days 10 and 24. On each occasion, rabbits received 0.5 mL of a vaccine formulation containing twice the antigen content of F glycoproteins of RSV A and RSV B (120 mcg RSV preF A and 120 mcg RSV preF B), stabilized in prefusion conformation as contained in a single human dose of ABRYSVO [see Description (11)]. No adverse effects on mating, female fertility, or on embryo/fetal or post-natal survival, growth, or development were observed. There were no vaccine-related fetal malformations or variations.
8.2 Lactation
Risk Summary
It is not known whether ABRYSVO is excreted in human milk. Data are not available to assess the effects of ABRYSVO on the breastfed infant or on milk production/excretion. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for ABRYSVO and any potential adverse effects on the breastfed child from ABRYSVO or from the underlying maternal condition. For preventative vaccines, the underlying maternal condition is susceptibility to disease prevented by the vaccine.
8.4 Pediatric Use
The safety and effectiveness of ABRYSVO to prevent RSV LRTD and severe RSV LRTD in infants born to individuals vaccinated at younger than 10 years of age have not been established.
The safety and effectiveness of ABRYSVO to prevent RSV LRTD in non-pregnant individuals younger than 18 years of age via active immunization have not been established.
8.5 Geriatric Use
ABRYSVO is approved for use in individuals 60 years of age and older. In Study 3 and Study 7, of the 18,681 recipients who received ABRYSVO 63% (n=11,695) were aged 60-69 years of age, 32% (n=5,958) were 70-79 years of age and 5% (n=1,027) were ≥80 years of age [see Adverse Reactions (6.1), Use in Specific Populations (8.6), and Clinical Studies (14.2)].
8.6 Immunocompromised Individuals 18 Years of Age and Older
Study 7 Substudy B (NCT05842967) was a Phase 3 single‑arm, open‑label, multicenter study to assess the safety and immunogenicity of ABRYSVO in immunocompromised individuals ≥18 years of age. A total of 203 participants received one dose of ABRYSVO and 201 participants received a second dose of ABRYSVO one month later (an unapproved dosing regimen).
In Study 7, 46% of participants were male, 74% were White, 20% were Black or African American, 4% were Asian, and 5% were Hispanic/Latino. Forty‑seven percent (47%) were 18 through 59 years of age and 53% were ≥60 years of age. The median age of participants was 60 years. In this study, 48% had an autoimmune inflammatory disorder on active immunomodulator therapy, 37% had a history of solid organ transplant on maintenance immunosuppressive therapy, 15% had end‑stage renal disease on hemodialysis, and 3% were on therapy for advanced non‑small cell lung cancer.
Solicited local and systemic reactions that occurred within 7 days following study vaccination were collected. Unsolicited adverse events were collected for 1 month after dose 2 of study vaccination, and serious adverse events were collected for 6 months following dose 2 of study vaccination.
The reactogenicity profile in immunocompromised participants was similar to those observed in other studies [see Adverse Reactions (6.1)], with the following exceptions: Following vaccination with ABRYSVO, pain at the injection site (in up to 46.8% of participants) was the most frequently reported solicited local reaction. The percentages of participants 18 through 59 years of age and ≥60 years of age reporting any solicited local reaction were higher after dose 2 (46.8% and 34.3%, respectively) than dose 1 (26.0% and 19.6%, respectively). There were no severe solicited local reactions. In this study, at least one severe solicited systemic reaction, defined as those preventing daily routine activity (predominantly fatigue as well as headache, muscle pain, and joint pain), was reported in 2.1% and 4.7% of participants 18 through 59 years and ≥60 years of age, respectively, after dose 1 and reported in <1% of participants after dose 2.
One 66 year old participant reported atrial fibrillation and atrial flutter with onset of symptoms 15 days after dose 1. The currently available information on atrial fibrillation and atrial flutter is insufficient to determine a causal relationship to the vaccine [see Adverse Reactions (6.1)].
After a single dose of ABRYSVO, the seroresponse rates [defined as achieving a ≥4‑fold rise in neutralizing titers for RSV A and RSV B from baseline if the baseline measurement is above the lower limit of quantitation (LLOQ) or a post‑vaccination assay result ≥4 × LLOQ if the baseline measurement is below the LLOQ] in the overall study population were 70.2% (95% CI 63.1%, 76.6%) and 71.3% (95% CI 64.2%, 77.6%) for RSV A and RSV B, respectively, 1 month after vaccination. Seroresponse rates in the overall study population did not further increase with a second dose of ABRYSVO 1 month after the first dose [70.2% (95% CI 63.1%, 76.6%) and 71.3% (95% CI 64.2%, 77.6%) for RSV A and RSV B, respectively]. In the subgroup with a history of solid organ transplant, seroresponse rates were 58.2% (95% CI 45.5%, 70.2%) 1 month after the first dose for both RSV A and RSV B, and 65.7% (95% CI 53.1%, 76.8%) and 64.2% (95% CI 51.5%, 75.5%) 1 month after the second dose for RSV A and RSV B, respectively. The data from this study do not establish the effectiveness of ABRYSVO in immunocompromised individuals.
-
11 DESCRIPTION
ABRYSVO (Respiratory Syncytial Virus Vaccine) is a sterile solution for intramuscular use. The vaccine is supplied as a vial of Lyophilized Antigen Component that is reconstituted at the time of use with a Sterile Water Diluent Component. The antigen component contains recombinant RSV preF A and RSV preF B.
The RSV preF A and RSV preF B recombinant proteins are expressed in genetically engineered Chinese Hamster Ovary cell lines grown in suspension culture using chemically-defined media, without antibiotics or animal-derived components. The recombinant proteins are purified through a series of column chromatography and filtration steps followed by formulation, filling into vials, and lyophilization.
After reconstitution, a single dose of ABRYSVO is formulated to contain 120 mcg of RSV stabilized prefusion F proteins (60 mcg RSV preF A and 60 mcg RSV preF B) per 0.5 mL. ABRYSVO also contains the following buffer ingredients: 0.11 mg tromethamine, 1.04 mg tromethamine hydrochloride, 11.3 mg sucrose, 22.5 mg mannitol, 0.08 mg polysorbate 80, and 1.1 mg sodium chloride per 0.5 mL. ABRYSVO is a sterile, clear, and colorless solution.
ABRYSVO contains no preservatives. Each dose may also contain residual amounts of host cell proteins (≤0.1% w/w) and DNA (<0.4 ng/mg of total protein) from the manufacturing process.
The Act-O-Vial stoppers, vial stoppers and tip cap and rubber plunger of the prefilled syringe are not made with natural rubber latex.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Active Immunization
ABRYSVO induces an immune response against RSVpreF that protects against lower respiratory tract disease caused by RSV.
Passive Immunization
Antibodies to RSV antigens from individuals vaccinated in pregnancy are transferred transplacentally to protect infants younger than 6 months of age against LRTD and severe LRTD caused by RSV.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
ABRYSVO has not been evaluated for the potential to cause carcinogenicity, genotoxicity, or impairment of male fertility. A developmental toxicity study in female rabbits revealed no evidence of impaired female fertility after administration of a vaccine formulation containing two times the antigen content of a single human dose of ABRYSVO [see Use in Specific Populations (8.1)].
-
14 CLINICAL STUDIES
14.1 Study in Pregnant Individuals for Efficacy in Their Infants from Birth Through 6 Months of Age
Study 1 (NCT04424316) was a Phase 3 study that assessed the efficacy of ABRYSVO in the prevention of RSV-associated lower respiratory tract disease (LRTD) in infants born to individuals vaccinated during pregnancy. The study evaluated the efficacy of ABRYSVO to prevent RSV-associated LRTD and severe RSV-LRTD in infants within 90, 120, 150, and 180 days after birth. Participants were randomized (1:1) to receive ABRYSVO (0.5 mL dose) or placebo (0.5 mL dose containing the same buffer ingredients in the same quantities as in a single dose of ABRYSVO [see Description (11)]). This study included sites in both the northern and southern hemispheres. Vaccine efficacy (VE) was defined as the relative risk reduction of the endpoints of severe LRTD caused by RSV and LRTD cause by RSV in infants born to individuals who received ABRYSVO compared to infants born to individuals who received placebo. The demographic characteristics of Study 1 are described in Clinical Trials Experience Section 6.1.
Maternal participants were randomized (1:1) to receive ABRYSVO (3,711) or placebo (3,709). RSV-associated LRTD in infants was defined as a medically attended visit with a reverse transcription-polymerase chain reaction (RT-PCR) confirmed RSV illness with one or more of the following respiratory symptoms: tachypnea (respiratory rate ≥60 breaths/minute [<2 months of age], ≥50 breaths/minute [≥2 to 12 months of age], or ≥40 breaths/minute [≥12-24 months of age]); SpO2 measured in room air <95%; chest wall indrawing. RSV-associated severe LRTD was a subset defined as meeting the LRTD RSV criteria plus at least one of the following: tachypnea (respiratory rate ≥70 breaths per minute [<2 months of age], ≥60 breaths per minute [≥2 to 12 months of age], or ≥50 bpm [≥12 to 24 months of age]); SpO2 measured in room air <93%; high-flow nasal cannula or mechanical ventilation (invasive or noninvasive), ICU admission for >4 hours and/or failure to respond/unconscious. Secondary efficacy endpoints included hospitalizations due to RSV.
The VE results met the statistical criterion for success (a CI lower bound >20%) for reducing severe LRTD due to RSV, at all timepoints to within 180 days. The VE results did not meet the statistical criterion for success (a CI lower bound >20%) for reducing LRTD due to RSV; however, clinically meaningful efficacy was observed after 90 days through 180 days after birth.
Vaccine efficacy information is presented in Tables 8 to 12.
Table 8 Vaccine Efficacy of ABRYSVO Against Severe LRTD Caused by RSV – Infants From Birth Through 6 Months of Age by Active Immunization of Pregnant Individuals (Study 1)* CI – confidence interval; N – number of participants; RSV – respiratory syncytial virus; VE – vaccine efficacy Time Period
ABRYSVO
Number of Cases
N=3,495†
PLACEBO
Number of Cases
N=3,480†
VE (%)
(CI)‡
90 days
6
33
81.8 (40.6, 96.3)
120 days
12
46
73.9 (45.6, 88.8)
150 days
16
55
70.9 (44.5, 85.9)
180 days
19
62
69.4 (44.3, 84.1)
Table 9 Vaccine Efficacy of ABRYSVO Against LRTD Caused by RSV – Infants From Birth Through 6 Months of Age by Active Immunization of Pregnant Individuals (Study 1)* CI – confidence interval; N – number of participants; RSV – respiratory syncytial virus; VE – vaccine efficacy Time Period
ABRYSVO
Number of Cases
N=3,495†
PLACEBO
Number of Cases
N=3,480†
VE (%)
(CI)‡
90 days
24
56
57.1 (14.7, 79.8)
120 days
35
81
56.8 (31.2, 73.5)
150 days
47
99
52.5 (28.7, 68.9)
180 days
57
117
51.3 (29.4, 66.8)
Table 10 Vaccine Efficacy of ABRYSVO Against Severe LRTD Caused by RSV – Infants From Birth Through 6 Months of Age by Active Immunization of Pregnant Individuals at 32 Through 36 Weeks Gestational Age (Study 1)* CI – confidence interval; N – number of participants; n – number of cases; RSV – respiratory syncytial virus; VE – vaccine efficacy Time Period
ABRYSVO
Number of Cases
N=1,572†
PLACEBO
Number of Cases
N=1,539†
VE (%)
(CI)‡
90 days
1
11
91.1 (38.8, 99.8)
180 days
6
25
76.5 (41.3, 92.1)
Table 11 Vaccine Efficacy of ABRYSVO Against LRTD Caused by RSV – Infants From Birth Through 6 Months of Age by Active Immunization of Pregnant Individuals at 32 Through 36 Weeks Gestational Age (Study 1)* CI – confidence interval; N – number of participants; n – number of cases; RSV – respiratory syncytial virus; VE – vaccine efficacy Time Period
ABRYSVO
Number of Cases
N=1,572†
PLACEBO
Number of Cases
N=1,539†
VE (%)
(CI)‡
90 days
14
21
34.7 (-34.6, 69.3)
180 days
24
55
57.3 (29.8, 74.7)
Table 12 Vaccine Efficacy of ABRYSVO Against Hospitalization Due to RSV – Infants From Birth Through 6 Months of Age by Active Immunization of Pregnant Individuals (Study 1)* CI – confidence interval; N – number of participants; n – number of cases; RSV – respiratory syncytial virus; VE – vaccine efficacy Time Period
ABRYSVO
Number of Cases
N=3,495†
PLACEBO
Number of Cases
N=3,480†
VE (%)
(CI)‡
90 days
10
31
67.7 (15.9, 89.5)
120 days
15
37
59.5 (8.3, 83.7)
150 days
17
39
56.4 (5.2, 81.5)
180 days
19
44
56.8 (10.1, 80.7)
14.2 Efficacy in Individuals 60 Years of Age and Older
Study 3 (NCT05035212) was a Phase 3, multicenter, randomized, double-blind, placebo-controlled study to assess the efficacy and safety of ABRYSVO in the prevention of RSV-associated lower respiratory tract disease in individuals 60 years of age and older. Participants were followed for up to two RSV seasons, approximately 25 months.
Participants were randomized (1:1) to receive ABRYSVO (n=18,487) or placebo (n=18,479). Randomization was stratified by age, 60-69 years (n=23,151, 63%), 70-79 years (n=11,782, 32%), and ≥80 years (n=2,031, 6%). Healthy adults and adults with stable chronic diseases were included. Among enrolled participants 16% had stable chronic cardiopulmonary conditions such as chronic obstructive pulmonary disease (COPD), asthma, or congestive heart failure (CHF).
Starting 14 days after study vaccination (study Day 15), all participants were actively monitored for onset of acute respiratory illness (ARI) symptoms: new or increased sore throat, nasal congestion, nasal discharge, cough, wheezing, sputum production, or shortness of breath. If the participant experienced 1 or more ARI symptoms, a mid-turbinate nasal swab was collected within 7 days of onset of symptoms and tested by reverse transcriptase polymerase chain reaction (RT-PCR) for RSV.
RSV-associated lower respiratory tract disease (RSV-LRTD) was evaluated in Study 3. A case of RSV-LRTD was defined as an RT-PCR confirmed RSV illness with two or more, or three or more, of the following respiratory symptoms within 7 days of symptom onset and lasting more than 1 day during the same illness: new or increased cough, wheezing, sputum production, shortness of breath, or tachypnea (≥25 breaths/min or 15% increase from resting baseline). A case of RSV-associated severe lower respiratory tract disease was defined as a case meeting the RSV-LRTD criteria plus at least one of the following: hospitalization due to RSV-LRTD, new or increased oxygen supplementation, or mechanical ventilation including Continuous Positive Airway Pressure (CPAP).
Vaccine efficacy (VE) against RSV-LRTD, defined as the relative risk reduction of first episode of RSV-LRTD in the ABRYSVO group compared to the placebo group in the first RSV season, was assessed.
The primary analysis of efficacy was conducted when 44 first-episode RSV-LRTD cases with ≥2 symptoms had accrued in the first RSV season. The study met the pre-specified success criteria for demonstration of efficacy of ABRYSVO for the primary objectives of prevention of RSV-LRTD with ≥2 symptoms and prevention of RSV-LRTD with ≥3 symptoms.
Vaccine efficacy information for the primary analysis is presented in Table 13.
Table 13 Vaccine Efficacy of ABRYSVO Against RSV-LRTD - Individuals 60 Years of Age and Older – Primary Analysis (Study 3)* CI – confidence interval; N – number of participants; n = number of cases; RSV – respiratory syncytial virus; VE – vaccine efficacy (VE based on case count ratio is calculated as 1-(P/[1-P]), where P is the number of cases in the ABRYSVO group divided by the total number of cases combined ABRYSVO and placebo) Efficacy Endpoint
7 months median follow-up
ABRYSVO
N=16,306†
n
Placebo
N=16,308†
n
VE (%)
(96.66% CI)
First episode of RSV-associated lower respiratory tract disease with ≥2 symptoms‡
11
33
66.7 (28.8, 85.8)
First episode of RSV-associated lower respiratory tract disease with ≥3 symptoms‡
2
14
85.7 (32.0, 98.7)
There were 2 cases of RSV-associated severe lower respiratory tract disease in the placebo group and no cases in the ABRYSVO group during the first RSV season. In the second RSV season, 1 case was reported in each group.
Descriptive vaccine efficacy analyses at the end of the first RSV season and combined across the two RSV seasons are presented in Tables 14 and 15.
Table 14 Descriptive Vaccine Efficacy of ABRYSVO Against RSV LRTD - Individuals 60 Years of Age and Older, End of First RSV Season (Study 3)* CI – confidence interval; N – number of participants; n = number of cases; RSV – respiratory syncytial virus; VE – vaccine efficacy (VE based on case count ratio is calculated as 1-(P/[1-P]), where P is the number of cases in the ABRYSVO group divided by the total number of cases) - *
- NCT05035212
- †
- RSV-associated LRTD symptoms included the following: new or increased cough, wheezing, sputum production, shortness of breath, or tachypnea (≥25 breaths/min or 15% increase from resting baseline).
- ‡
- Evaluable efficacy population.
- §
- VE cannot be reliably estimated due to the small number of accrued cases in the subgroup.
- ¶
- Subgroup of evaluable efficacy population with ≥1 significant underlying condition i.e. heart disease (including congestive heart failure), lung disease (including chronic obstructive pulmonary disease), asthma, diabetes mellitus, liver disease, renal disease, and current tobacco use.
- #
- Subgroup analyses by age are not presented for RSV-LRTD with ≥3 symptoms due to the limited number of accrued cases in the age subgroups during the first RSV season.
Efficacy Endpoint
Analysis Population
ABRYSVO
n/N
Placebo
n/N
VE (%)
(95% CI)
7.4 months median follow-up
First episode of RSV-associated lower respiratory tract disease with ≥2 symptoms†
Overall‡
15/18,050
43/18,074
65.1 (35.9, 82.0)
Age 60-69 years
10/11,305
25/11,351
60.0 (13.8, 82.9)
Age 70-79 years
4/5,750
12/5,742
66.7 (-10.0, 92.2)§
Age ≥ 80 years
1/995
6/981
83.3 (-37.4, 99.6)§
With ≥1 significant underlying condition¶
8/9,387
22/9,448
63.6 (15.2, 86.0)
First episode of RSVassociated lower respiratory tract disease with ≥3 symptoms†
2/18,050
18/18,074
88.9 (53.6, 98.7)
With ≥1 significant underlying condition¶
2/9,387
11/9,448
81.8 (16.7, 98.0)
For RSV-A associated LRTD at the end of the first season, descriptive VE was 81.3% (95% CI 34.5, 96.5) for cases with ≥2 LRTD symptoms and 80.0% (95% CI -78.7, 99.6) for cases with ≥3 LRTD symptoms. For RSV-B associated LRTD at the end of the first season, descriptive VE was 53.8% (95% CI 5.2, 78.8) for cases with ≥2 LRTD symptoms and 91.7% (95% CI 43.7, 99.8) for cases with ≥3 LRTD symptoms.
In the second RSV season, VE against RSV-associated LRTD with ≥2 symptoms was 55.7% (95% CI 34.7, 70.4; 39 cases in the ABRYSVO group and 88 cases in the placebo group) and with ≥3 symptoms was 77.8% (95% CI 51.4, 91.1; 8 cases in the ABRYSVO group and 36 cases in the placebo group). The median duration of follow-up was 7.7 months in the second season.
Table 15 Descriptive Vaccine Efficacy of ABRYSVO Against RSV LRTD - Individuals 60 Years of Age and Older, Across Two RSV Seasons (Study 3)* CI – confidence interval; N – number of participants; n = number of cases; RSV – respiratory syncytial virus; VE – vaccine efficacy (VE based on case count ratio is calculated as 1-(P/[1-P]), where P is the number of cases in the ABRYSVO group divided by the total number of cases) - *
- NCT05035212
- †
- RSV-associated LRTD symptoms included the following: new or increased cough, wheezing, sputum production, shortness of breath, or tachypnea (≥25 breaths/min or 15% increase from resting baseline).
- ‡
- Evaluable efficacy population.
- §
- VE cannot be reliably estimated due to the small number of accrued cases in the subgroup.
- ¶
- Subgroup of evaluable efficacy population with ≥1 significant underlying condition i.e. heart disease (including congestive heart failure), lung disease (including chronic obstructive pulmonary disease), asthma, diabetes mellitus, liver disease, renal disease, and current tobacco use.
Efficacy Endpoint
Analysis Population
ABRYSVO
n/N
Placebo
n/N
VE (%)
(95% CI)
16.9 months median time since vaccination
First episode of RSV-associated lower respiratory tract disease with ≥2 symptoms†
Overall‡
54/18,050
131/18,074
58.8 (43.0, 70.6)
Age 60-69 years
34/11,305
80/11,351
57.5 (35.8, 72.4)
Age 70-79 years
15/5,750
40/5,742
62.5 (30.6, 80.8)
Age ≥ 80 years
5/995
11/981
54.5 (-41.9, 87.6)§
With ≥1 significant underlying condition¶
36/9,387
71/9,448
49.3 (23.2, 67.0)
First episode of RSV-associated lower respiratory tract disease with ≥3 symptoms†
Overall‡
10/18,050
54/18,074
81.5 (63.3, 91.6)
Age 60-69 years
7/11,305
38/11,351
81.6 (58.2, 93.1)
Age 70-79 years
3/5,750
11/5,742
72.7 (-3.2, 95.1)§
Age ≥ 80 years
0/995
5/981
100.0 (-9.1, 100.0)§
With ≥1 significant underlying condition¶
9/9,387
34/9,448
73.5 (43.6, 88.8)
For RSV-A associated LRTD across two RSV seasons, descriptive VE was 66.3% (95% CI 47.2, 79.0) for cases with ≥2 LRTD symptoms and 80.6% (95% CI 52.9, 93.4) for cases with ≥3 LRTD symptoms. For RSV-B associated LRTD across two RSV seasons, descriptive VE was 50.0% (95% CI 18.5, 70.0) for cases with ≥2 LRTD symptoms and 86.4% (95% CI 54.6, 97.4) for cases with ≥3 LRTD symptoms.
14.3 Immunogenicity in Individuals 18 Through 59 Years of Age Considered to be at Increased Risk of LRTD caused by RSV
Study 4 was a Phase 3, multicenter, randomized, double‑blind, placebo‑controlled study to assess the safety and immunogenicity of ABRYSVO in individuals 18 through 59 years of age considered to be at increased risk of LRTD caused by RSV due to chronic medical conditions. Study 4 enrolled individuals who had chronic pulmonary (including asthma), cardiovascular (excluding isolated hypertension), renal, hepatic, neurologic, hematologic or metabolic disorders (including diabetes mellitus and hyper/hypothyroidism) [see Adverse Reactions (6.1)]. Effectiveness was assessed by comparison of the RSV neutralizing geometric mean titers (GMTs) and seroresponse rates of the evaluable immunogenicity population in Study 4 who received ABRYSVO (n=437) to those of a subgroup of individuals from select sites in the United States and Japan in Study 3 (n=410) (Tables 16 and 17). Participants in the Study 3 subgroup were 60 years of age or older; 44% had chronic medical conditions.
Non-inferiority was demonstrated for the ratio of neutralizing GMTs for RSV A and RSV B (Study 4/Study 3 subgroup; lower bounds of the 2‑sided 95% CIs >0.667) (Table 16), and the percentage difference in neutralizing titer seroresponse rates for RSV A and RSV B (Study 4 minus Study 3; lower bounds of the 2‑sided 95% CIs >-10%) (Table 17).
Table 16 Comparison of Model Adjusted RSV Neutralizing GMTs at 1 Month After Vaccination with ABRYSVO, 18 Through 59 Years at Increased Risk of LRTD caused by RSV (Study 4)* versus 60 Years and Older (Study 3)† CI – confidence interval; GMR – geometric mean ratio; GMT – geometric mean titer RSV subgroups
Study 4*
18-59 years of age at high risk
N=435-437
Adjusted GMT‡
(95% CI)
Study 3†
≥60 years
N=408
Adjusted GMT‡
(95% CI)
(95% CI)
A
41097 (37986, 44463)
26225 (24143, 28486)
1.57 (1.396, 1.759)
B
37416 (34278, 40842)
24680 (22504, 27065)
1.52 (1.333, 1.725)
Table 17 Comparison of RSV Neutralizing Titer Seroresponse* Rates 1 Month After Vaccination with ABRYSVO, 18 Through 59 Years at Increased Risk of LRTD caused by RSV (Study 4)† versus 60 Years and Older (Study 3)‡ CI - confidence interval - *
- Seroresponse is defined as achieving a ≥4-fold rise from baseline if the baseline measurement is above the lower limit of quantitation (LLOQ). If the baseline measurement is below the LLOQ, a post-vaccination assay result ≥4 × LLOQ is considered a seroresponse.
- †
- NCT05842967
- ‡
- NCT05035212
- §
- Non-inferiority was met if the lower bound of the 2-sided CI for the percentage difference (Study 4 minus Study 3) >-10% (10% NI criterion) for both RSV A and RSV B.
RSV subgroups
Study 4
18-59 years of age at high risk
N=435-437
% (95% CI)
Study 3
≥60 years
N=408
% (95% CI)
Percentage Difference§ (95% CI)
A
93 (90.3, 95.3)
88 (84.4, 91.0)
5.1 (1.2, 9.2)
B
93 (90.6, 95.5)
85 (81.2, 88.4)
8.3 (4.2, 12.6)
14.4 Concomitant Administration of ABRYSVO with a Seasonal Inactivated Influenza Vaccine
In Study 5 (NCT05301322) [see Adverse Reactions (6.1)], antibody responses to antigens contained in ABRYSVO and FLUAD QUADRIVALENT were assessed 1 month after vaccination in individuals 65 years of age and older.
Immunologic non-inferiority was demonstrated for concomitant administration of ABRYSVO and FLUAD QUADRIVALENT as compared to sequential administration. The lower limits of the 2-sided 95% CIs for the Geometric Mean Titer Ratio (GMR) (concomitant administration group versus sequential administration group) for RSV A and RSV B neutralizing titers (NT) and hemagglutination inhibition (HAI) titers against influenza strains A/Victoria, A/Darwin, B/Austria, and B/Phuket were above 0.667 (the predefined 1.5-fold non-inferiority criterion).
14.5 Concomitant Administration of ABRYSVO with Tetanus Toxoid, Reduced Diphtheria Toxoid and Acellular Pertussis Vaccine, Adsorbed
Study 6 was a Phase 2, placebo-controlled, randomized, observer-blind study to evaluate the safety, tolerability, and immunogenicity of ABRYSVO (at dose levels 120 µg and 240 µg, with or without Al(OH)3) when administered concomitantly with Tdap in non-pregnant women 18 through 49 years of age.
Antibody responses to antigens contained in ABRYSVO and Tdap were assessed 1 month after vaccination in a population of non-pregnant adult individuals. Lower geometric mean antibody concentrations (GMCs) to the acellular pertussis antigens (pertussis toxin [PT], filamentous hemagglutinin (FHA), and pertactin [PRN]) were observed when ABRYSVO was administered concomitantly with a tetanus, diphtheria and acellular pertussis vaccine (Tdap) compared to pertussis GMCs when Tdap was administered alone. The lower limit (LL) of the 2-sided 95% confidence interval of the GMC ratio (GMC ABRYSVO+Tdap /GMC Tdap) was 0.64 for PT, 0.50 for FHA, and 0.48 for PRN, which did not meet the pre-specified non-inferiority criterion (lower limit of the 95% confidence interval for the GMC ratio is >0.67). The clinical relevance of this finding is unknown. The non-inferiority criteria for tetanus, diphtheria and RSV vaccine antigens were met [see Drug Interactions (7)].
-
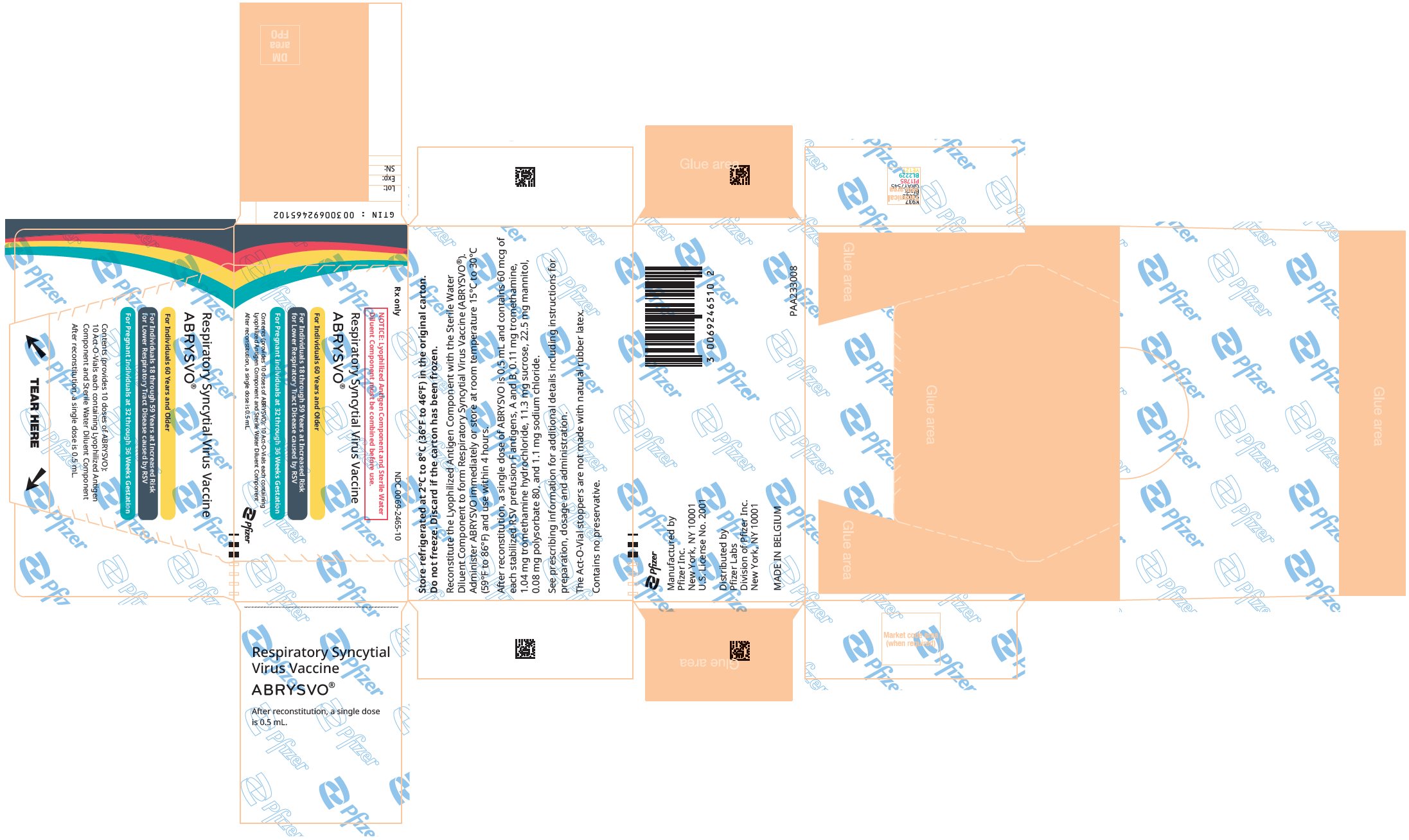
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
ABRYSVO Act-O-Vial presentation is supplied in cartons of 1 and 10 Act-O-Vials, without syringes or needles. Each Act-O-Vial contains both the Lyophilized Antigen and Sterile Water Diluent (NDC 0069-2465-19).
Carton: 1 Act-O-Vial
NDC 0069-2465-01
Carton: 10 Act-O-Vials
NDC 0069-2465-10
ABRYSVO vial and prefilled syringe presentation is supplied in cartons of 1, 5, and 10 kits, without needles. Each kit includes a vial of Lyophilized Antigen Component (NDC 0069-0207-01), a prefilled syringe containing Sterile Water Diluent Component (NDC 0069-0250-01), and a vial adapter.
Carton: 1 kit
NDC 0069-0344-01
Carton: 5 kits
NDC 0069-0344-05
Carton: 10 kits
NDC 0069-0344-10
ABRYSVO vial and vial presentation is supplied in cartons of 5 and 10 doses packaged without syringes or needles. Each carton includes vials of Lyophilized Antigen Component (NDC 0069-0207-01) and vials of Sterile Water Diluent Component (NDC 0069-0651-01).
Carton: 5 doses
NDC 0069-1265-10
Carton: 10 doses
NDC 0069-1265-20
The Act-O-Vial stoppers, vial stoppers and the tip cap and rubber plunger of the prefilled syringe are not made with natural rubber latex.
-
17 PATIENT COUNSELING INFORMATION
Prior to administration of this vaccine:
- •
- Inform vaccine recipient of the potential benefits and risks of vaccination with ABRYSVO.
- •
- Advise vaccine recipient to report any adverse events to their healthcare provider or to the Vaccine Adverse Event Reporting System at 1-800-822-7967 and www.vaers.hhs.gov.
This product's labeling may have been updated. For the most recent prescribing information, please visit www.pfizer.com.
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 10 Vial/Syringe Kit Carton
NDC 0069-0344-10
Rx onlyNOTICE: Lyophilized Antigen Component and Sterile
Water Diluent Component must be combined before use.Respiratory Syncytial
Virus Vaccine
ABRYSVO®For Individuals 60 Years and Older
For Individuals 18 through 59 Years at Increased Risk
for Lower Respiratory Tract Disease caused by RSVFor Pregnant Individuals at 32 through 36 Weeks Gestation
Kit Contents (provides 10 doses of ABRYSVO):
10 vials containing Lyophilized Antigen Component
10 syringes containing Sterile Water Diluent Component
10 vial adapters
After reconstitution, a single dose is approximately 0.5 mL.Pfizer
-
PRINCIPAL DISPLAY PANEL - 5 Vial/Syringe Kit Carton
NDC 0069-0344-05
Rx onlyNOTICE: Lyophilized Antigen Component and
Sterile Water Diluent Component must be
combined before use.Respiratory Syncytial
Virus Vaccine
ABRYSVO®For Individuals 60 Years and Older
For Individuals 18 through 59 Years at Increased Risk
for Lower Respiratory Tract Disease caused by RSVFor Pregnant Individuals at 32 through 36 Weeks Gestation
Kit Contents (provides 5 doses of ABRYSVO):
5 vials containing Lyophilized Antigen Component
5 syringes containing Sterile Water Diluent Component
5 vial adapters
After reconstitution, a single dose is approximately 0.5 mL.Pfizer
-
PRINCIPAL DISPLAY PANEL - 1 Vial/Syringe Kit Carton
NDC 0069-0344-01
Rx onlyNOTICE: Lyophilized Antigen Component and
Sterile Water Diluent Component must be
combined before use.Respiratory Syncytial
Virus Vaccine
ABRYSVO®For Individuals 60 Years and Older
For Individuals 18 through 59 Years at Increased Risk
for Lower Respiratory Tract Disease caused by RSVFor Pregnant Individuals at 32 through 36 Weeks Gestation
Kit Contents (provides a single dose of ABRYSVO):
1 vial containing Lyophilized Antigen Component
1 syringe containing Sterile Water Diluent Component
1 vial adapter
After reconstitution, a single dose is approximately 0.5 mL.Pfizer
- PRINCIPAL DISPLAY PANEL - 0.5 mL Vial Label
- PRINCIPAL DISPLAY PANEL - 1 Syringe Label
-
PRINCIPAL DISPLAY PANEL – 1 Act-O-Vial Carton
Rx only
NDC 0069-2465-01NOTICE: Lyophilized Antigen
Component and Sterile Water
Diluent Component must be
combined before use.Respiratory Syncytial
Virus Vaccine
ABRYSVO®For Individuals 60 Years
and OlderFor Individuals 18 through
59 Years at Increased Risk
for Lower Respiratory Tract
Disease caused by RSVFor Pregnant Individuals
at 32 through 36 Weeks
GestationContents (provides 1 dose of
ABRYSVO): 1 Act-O-Vial
containing Lyophilized Antigen
Component and Sterile Water
Diluent ComponentAfter reconstitution, a single
dose is 0.5 mL.Pfizer
-
PRINCIPAL DISPLAY PANEL – 10 Act-O-Vials Carton
Rx only
NDC 0069-2465-10NOTICE: Lyophilized Antigen Component and Sterile Water
Diluent Component must be combined before use.Respiratory Syncytial Virus Vaccine
ABRYSVO®For Individuals 60 Years and Older
For Individuals 18 through 59 Years at Increased Risk
for Lower Respiratory Tract Disease caused by RSVFor Pregnant Individuals at 32 through 36 Weeks Gestation
Contents (provides 10 doses of ABRYSVO): 10 Act-O-Vials each containing
Lyophilized Antigen Component and Sterile Water Diluent Component
After reconstitution, a single dose is 0.5 mL.Pfizer
- PRINCIPAL DISPLAY PANEL – Act-O-Vial Label
-
INGREDIENTS AND APPEARANCE
ABRYSVO
respiratory syncytial virus vaccine kitProduct Information Product Type VACCINE Item Code (Source) NDC:0069-0344 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0069-0344-01 1 in 1 CARTON 1 NDC:0069-0344-21 1 in 1 TRAY; Type 9: Other Type of Part 3 Combination Product (e.g., Drug/Device/Biological Product) 2 NDC:0069-0344-05 5 in 1 CARTON 2 NDC:0069-0344-21 1 in 1 TRAY; Type 9: Other Type of Part 3 Combination Product (e.g., Drug/Device/Biological Product) 3 NDC:0069-0344-10 10 in 1 CARTON 3 NDC:0069-0344-21 1 in 1 TRAY; Type 9: Other Type of Part 3 Combination Product (e.g., Drug/Device/Biological Product) Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL, GLASS 0.5 mL Part 2 1 SYRINGE, GLASS 0.69 mL Part 1 of 2 ABRYSVO
respiratory syncytial virus vaccine injection, powder, lyophilized, for solutionProduct Information Item Code (Source) NDC:0069-0207 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RECOMBINANT STABILIZED RSV A PREFUSION F ANTIGEN (UNII: 4PDL43Y9MR) (RECOMBINANT STABILIZED RSV A PREFUSION F ANTIGEN - UNII:4PDL43Y9MR) RECOMBINANT STABILIZED RSV A PREFUSION F ANTIGEN 0.06 mg in 0.5 mL RECOMBINANT STABILIZED RSV B PREFUSION F ANTIGEN (UNII: 34FS5XSD5Q) (RECOMBINANT STABILIZED RSV B PREFUSION F ANTIGEN - UNII:34FS5XSD5Q) RECOMBINANT STABILIZED RSV B PREFUSION F ANTIGEN 0.06 mg in 0.5 mL Inactive Ingredients Ingredient Name Strength TROMETHAMINE (UNII: 023C2WHX2V) 0.11 mg in 0.5 mL TROMETHAMINE HYDROCHLORIDE (UNII: 383V75M34E) 1.04 mg in 0.5 mL SUCROSE (UNII: C151H8M554) 11.3 mg in 0.5 mL MANNITOL (UNII: 3OWL53L36A) 22.5 mg in 0.5 mL POLYSORBATE 80 (UNII: 6OZP39ZG8H) 0.08 mg in 0.5 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 1.1 mg in 0.5 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0069-0207-01 0.5 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125769 07/12/2023 05/31/2026 Part 2 of 2 DILUENT SYRINGE FOR ABRYSVO
sterile water diluent injection, solutionProduct Information Item Code (Source) NDC:0069-0250 Route of Administration INTRAMUSCULAR Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0069-0250-01 0.69 mL in 1 SYRINGE, GLASS; Type 9: Other Type of Part 3 Combination Product (e.g., Drug/Device/Biological Product) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125769 07/12/2023 05/31/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125769 07/12/2023 05/31/2026 ABRYSVO
respiratory syncytial virus vaccine injection, powder, lyophilized, for solutionProduct Information Product Type VACCINE Item Code (Source) NDC:0069-2465 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RECOMBINANT STABILIZED RSV A PREFUSION F ANTIGEN (UNII: 4PDL43Y9MR) (RECOMBINANT STABILIZED RSV A PREFUSION F ANTIGEN - UNII:4PDL43Y9MR) RECOMBINANT STABILIZED RSV A PREFUSION F ANTIGEN 0.06 mg in 0.5 mL RECOMBINANT STABILIZED RSV B PREFUSION F ANTIGEN (UNII: 34FS5XSD5Q) (RECOMBINANT STABILIZED RSV B PREFUSION F ANTIGEN - UNII:34FS5XSD5Q) RECOMBINANT STABILIZED RSV B PREFUSION F ANTIGEN 0.06 mg in 0.5 mL Inactive Ingredients Ingredient Name Strength TROMETHAMINE (UNII: 023C2WHX2V) 0.11 mg in 0.5 mL TROMETHAMINE HYDROCHLORIDE (UNII: 383V75M34E) 1.04 mg in 0.5 mL SUCROSE (UNII: C151H8M554) 11.3 mg in 0.5 mL MANNITOL (UNII: 3OWL53L36A) 22.5 mg in 0.5 mL POLYSORBATE 80 (UNII: 6OZP39ZG8H) 0.08 mg in 0.5 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 1.1 mg in 0.5 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0069-2465-01 1 in 1 CARTON 1 NDC:0069-2465-19 0.5 mL in 1 VIAL; Type 0: Not a Combination Product 2 NDC:0069-2465-10 10 in 1 CARTON 2 NDC:0069-2465-19 0.5 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125769 07/30/2024 Labeler - Pfizer Laboratories Div Pfizer Inc (134489525) Establishment Name Address ID/FEI Business Operations Wyeth BioPharma Division of Wyeth Pharmaceuticals LLC 174350868 ANALYSIS(0069-0344, 0069-2465) , API MANUFACTURE(0069-0344, 0069-2465) Establishment Name Address ID/FEI Business Operations Pfizer Manufacturing Belgium NV 370156507 ANALYSIS(0069-0344, 0069-2465) , MANUFACTURE(0069-0344, 0069-2465) , PACK(0069-0344, 0069-2465) , LABEL(0069-0344, 0069-2465) Establishment Name Address ID/FEI Business Operations Pfizer Ireland Pharmaceuticals Unlimited Company 985586408 ANALYSIS(0069-0344, 0069-2465) Establishment Name Address ID/FEI Business Operations Hospira, Inc. 030606222 PACK(0069-0344) , LABEL(0069-0344) , MANUFACTURE(0069-0344) , ANALYSIS(0069-0344)