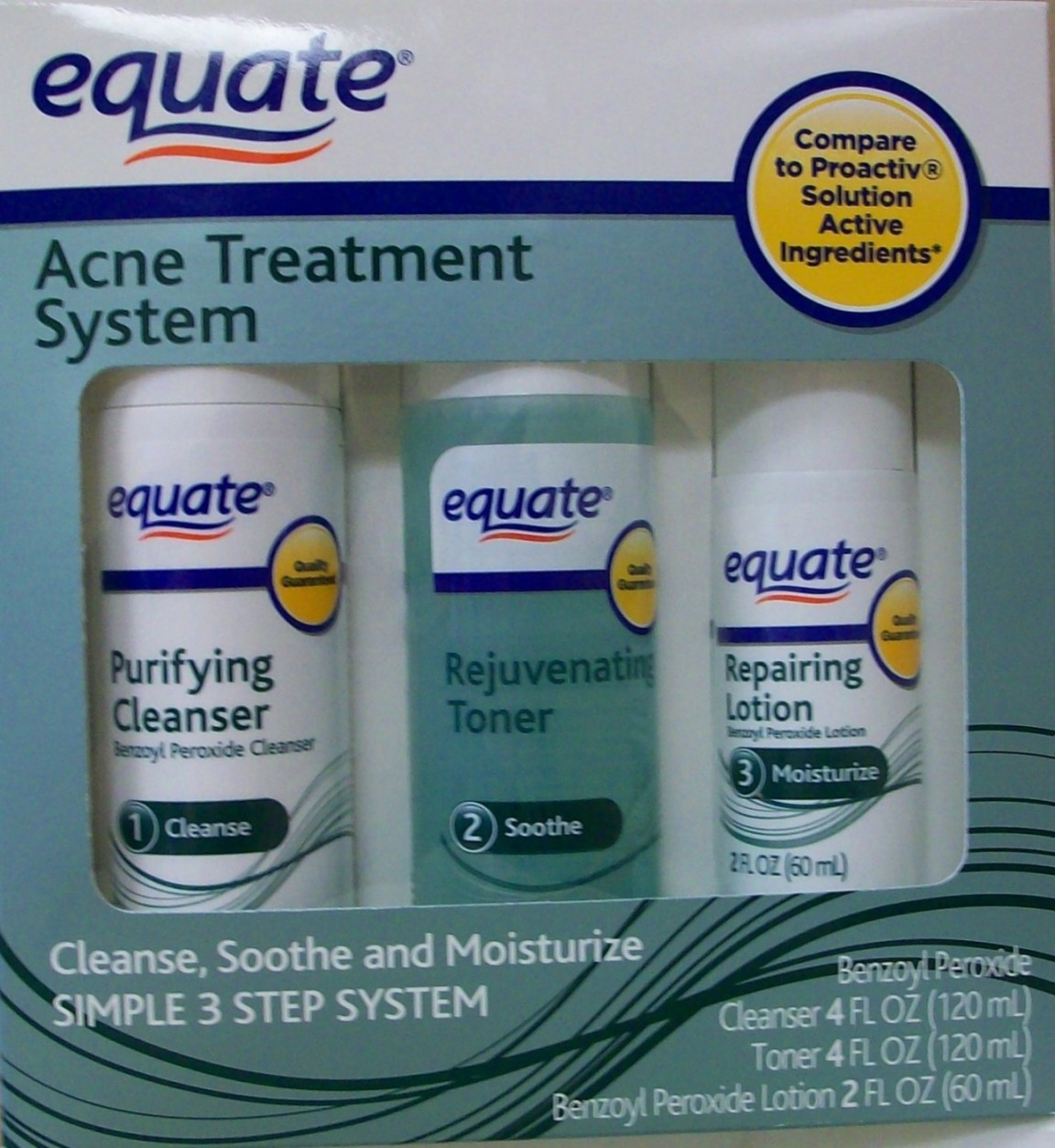
Label: BENZOYL PEROXIDE kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 49035-021-13 - Packager: Wal-Mart Stores Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 4, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
-
KEEP OUT OF REACH OF CHILDREN
Purifying Cleanser Warning Section
Warnings
For external use only
Do not use this medication if you have very sensitive
skin or if you are very sensitive to benzoyl peroxide.
Ask a doctor or pharmacist before use if you are
using other topical acne medications at the same
time or immediately following use of this product.
This may increase dryness or irritation of the skin.
If this occurs, only one medication should be used
unless directed by a doctor.
When using this product
- avoid contact with the eyes. If contact occurs,
rinse eyes thoroughly with water.
- keep away from lips and mouth
- aviod unnecessary sun exposure and use a
sunscreen
Stop use and ask a doctor if skin irritation occurs.
Keep out of the reach of children. If swallowed, get
medical help or contact a Poison Control Center right
away.
-
INDICATIONS & USAGE
Purifying Cleanser Directions Section
Directions
- Use morning and night. Apply a small (dime sized)
amount to dampened skin and gently massage for
1 to 2 minutes.
- Rinse thoroughly with warm water
- Pat dry
- If bothersome dryness or peeling occurs, reduce
application to once a day or every other day
- If going outside, use a sunscreen
Other information
- This product may bleach hair or dyed fabrics
- Avoid storing at extreme temperatures (below 40
degrees F and above 100 degrees F) -
WARNINGS
Purifying Cleanser Warning Section
Warnings
For external use only
Do not use this medication if you have very sensitive
skin or if you are very sensitive to benzoyl peroxide.
Ask a doctor or pharmacist before use if you are
using other topical acne medications at the same
time or immediately following use of this product.
This may increase dryness or irritation of the skin.
If this occurs, only one medication should be used
unless directed by a doctor.
When using this product
- avoid contact with the eyes. If contact occurs,
rinse eyes thoroughly with water.
- keep away from lips and mouth
- aviod unnecessary sun exposure and use a
sunscreen
Stop use and ask a doctor if skin irritation occurs.
Keep out of the reach of children. If swallowed, get
medical help or contact a Poison Control Center right
away.
-
DOSAGE & ADMINISTRATION
Purifying Cleanser Directions Section
Directions
- Use morning and night. Apply a small (dime sized)
amount to dampened skin and gently massage for
1 to 2 minutes.
- Rinse thoroughly with warm water
- Pat dry
- If bothersome dryness or peeling occurs, reduce
application to once a day or every other day
- If going outside, use a sunscreen
Other information
- This product may bleach hair or dyed fabrics
- Avoid storing at extreme temperatures (below 40
degrees F and above 100 degrees F)
-
INACTIVE INGREDIENT
Purifying Cleanser inactive ingredients section
Inactive Ingredients
purified water, disodium PEG-12 dimethicone
sulfosuccinate, magnesium aluminum silicate,
gyyceryl stearate, tridecyl stearate, neopentyl
glycol dicaprylate/dicaprate, tridecyl trimelliate,
PEG-100 Stearate, sorbitol, dimethyl isosorbide,
polyethylene, cetyl esters, anthemis nobilis flower
extract, xanthan gum, propylene glycol,
imidazolidinyl urea, sodium hyaluronate, sodium
PCA, methylparaben, propylparaben, fragrance - PRINCIPAL DISPLAY PANEL
- ACTIVE INGREDIENT
- PURPOSE
-
KEEP OUT OF REACH OF CHILDREN
Repairing Lotion Warning Section
Warnings
For external use only
Do not use this medication if you have very sensitive
skin or if you are very sensitive to benzoyl peroxide.
Ask a doctor or pharmacist before use if you are
using other topical acne medications at the same
time or immediately following use of this product.
This may increase dryness or irritation of the skin.
If this occurs, only one medication should be used
unless directed by a doctor.
When using this product
- avoid contact with the eyes. If contact occurs,
rinse eyes thoroughly with water.
- keep away from lips and mouth
- aviod unnecessary sun exposure and use a
sunscreen
Stop use and ask a doctor if skin irritation occurs.
Keep out of the reach of children. If swallowed, get
medical help or contact a Poison Control Center right
away. -
INDICATIONS & USAGE
Repairing Lotion Directions Section
Directions
- Cleanse the skin thoroughly before applying
medication
- Cover the entire affected area with a thin layer 1 to
3 times daily
- Because excessive drying of the skin may occur,
start with 1 application daily, then gradually
increase to 2 or 3 times daily, if needed or as
directed by a doctor. If bothersome dryness or
peeling occurs, reduce application to once a day or
every other day
- If going outside, use a sunscreen
Other information
- This product may bleach hair or dyed fabrics
- Avoid storing at extreme temperatures
(below 40 degrees F and above 100 degrees F) -
WARNINGS
Repairing Lotion Warning Section
Warnings
For external use only
Do not use this medication if you have very sensitive
skin or if you are very sensitive to benzoyl peroxide.
Ask a doctor or pharmacist before use if you are
using other topical acne medications at the same
time or immediately following use of this product.
This may increase dryness or irritation of the skin.
If this occurs, only one medication should be used
unless directed by a doctor.
When using this product
- avoid contact with the eyes. If contact occurs,
rinse eyes thoroughly with water.
- keep away from lips and mouth
- aviod unnecessary sun exposure and use a
sunscreen
Stop use and ask a doctor if skin irritation occurs.
Keep out of the reach of children. If swallowed, get
medical help or contact a Poison Control Center right
away. -
DOSAGE & ADMINISTRATION
Repairing Lotion Directions Section
Directions
- Cleanse the skin thoroughly before applying
medication
- Cover the entire affected area with a thin layer 1 to
3 times daily
- Because excessive drying of the skin may occur,
start with 1 application daily, then gradually
increase to 2 or 3 times daily, if needed or as
directed by a doctor. If bothersome dryness or
peeling occurs, reduce application to once a day or
every other day
- If going outside, use a sunscreen
Other information
- This product may bleach hair or dyed fabrics
- Avoid storing at extreme temperatures
(below 40 degrees F and above 100 degrees F) -
INACTIVE INGREDIENT
Repairing Lotion inactive ingredients section
Inactive ingredients
purified water, cetearyl alcohol, ceteareth-20,
ethoxydiglycol, cyclotetrasiloxane, propylene
glycol, dimethicone, glyceryl stearate,
cyclopentasiloxane diazolidinyl urea, methylparaben,
propylparaben, PEG-100 stearate, panthenol,
allantoin, triethanolamine, carbomer, fragrance,
xanthan gum
-
INGREDIENTS AND APPEARANCE
BENZOYL PEROXIDE
benzoyl peroxide kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49035-021 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49035-021-13 1 in 1 KIT Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE, PLASTIC 120 mL Part 2 1 BOTTLE, PLASTIC 60 mL Part 1 of 2 BENZOYL PEROXIDE
benzoyl peroxide creamProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOYL PEROXIDE (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) BENZOYL PEROXIDE 36.9 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) POLYOXYL 100 STEARATE (UNII: YD01N1999R) PEG-12 STEARATE (UNII: 9KU38O8CEK) SORBITOL (UNII: 506T60A25R) DIMETHYL ISOSORBIDE (UNII: SA6A6V432S) TRIDECYL STEARATE (UNII: A8OE252M6L) NEOPENTYL GLYCOL (UNII: QI80HXD6S5) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) CETYL ESTERS WAX (UNII: D072FFP9GU) CHAMAEMELUM NOBILE FLOWER (UNII: O2T154T6OG) HYALURONATE SODIUM (UNII: YSE9PPT4TH) SODIUM PYRROLIDONE CARBOXYLATE (UNII: 469OTG57A2) XANTHAN GUM (UNII: TTV12P4NEE) IMIDUREA (UNII: M629807ATL) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 120 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333D 02/26/2010 Part 2 of 2 BENZOYL PEROXIDE
benzoyl peroxide lotionProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOYL PEROXIDE (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) BENZOYL PEROXIDE 36.9 mg in 1 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) DIETHYLENE GLYCOL MONOETHYL ETHER (UNII: A1A1I8X02B) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) DIMETHICONE (UNII: 92RU3N3Y1O) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) METHYLPARABEN (UNII: A2I8C7HI9T) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) PROPYLPARABEN (UNII: Z8IX2SC1OH) PANTHENOL (UNII: WV9CM0O67Z) ALLANTOIN (UNII: 344S277G0Z) XANTHAN GUM (UNII: TTV12P4NEE) CARBOMER HOMOPOLYMER TYPE C (UNII: 4Q93RCW27E) POLYOXYL 100 STEARATE (UNII: YD01N1999R) TROLAMINE (UNII: 9O3K93S3TK) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 60 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333D 02/26/2010 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333D 02/26/2010 Labeler - Wal-Mart Stores Inc (051957769) Registrant - Pharma Pac, LLC (140807475) Establishment Name Address ID/FEI Business Operations Pharma Pac, LLC 140807475 manufacture