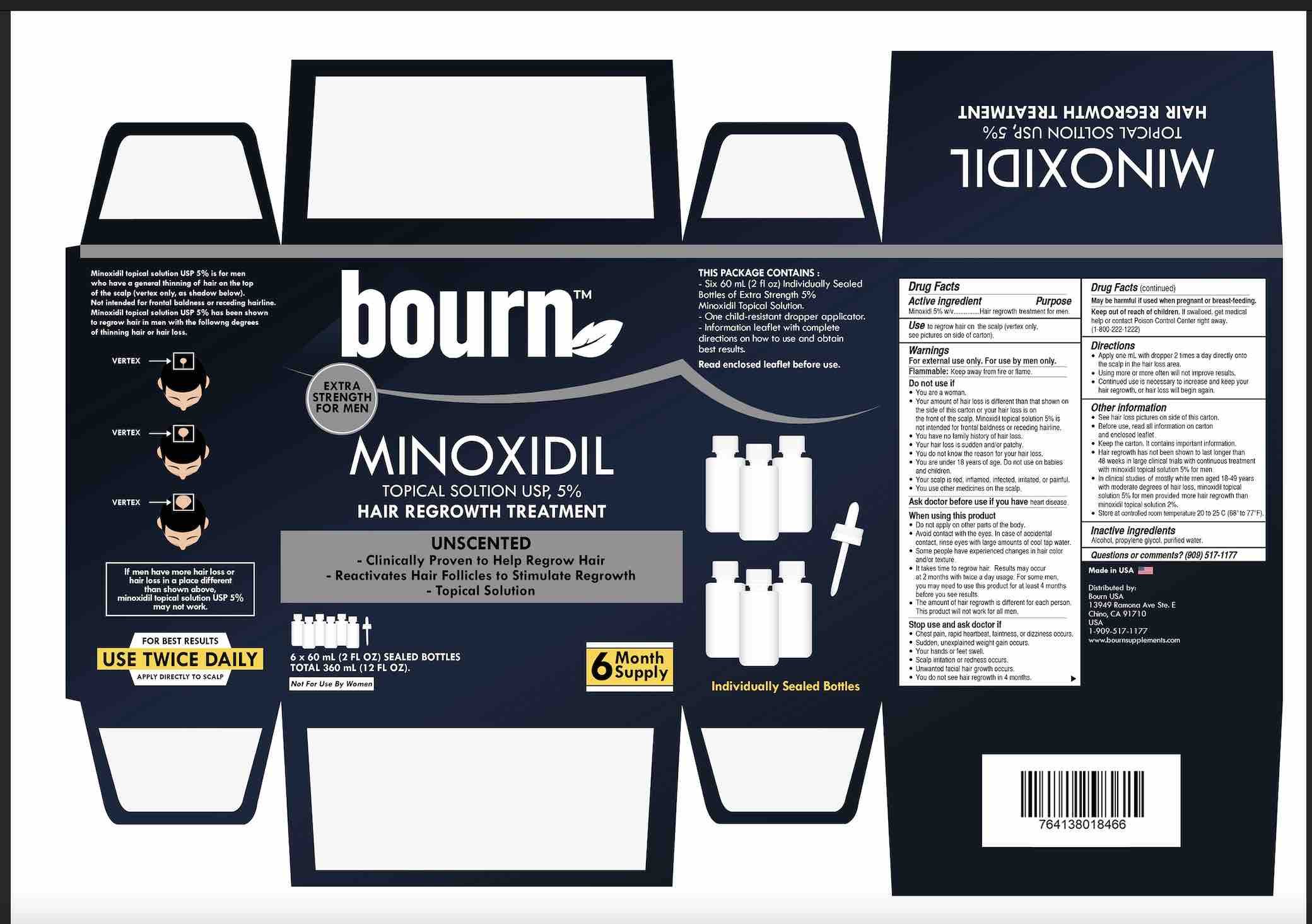
Label: BOURN HAIR REGROWTH TREATMENT FOR MEN- minoxidil 5% solution
- NDC Code(s): 81553-001-01
- Packager: Octri, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 10, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- USE
- WARNINGS
-
DO NOT USE
Do not use if
■ You are a woman
■ Your amount of hair loss is dierent than that shown on the side of this carton, or your hair loss is on the front of the scalp.
Minoxidil topical solution 5% is not intended for frontal baldness or receding hairline.
■ You have no family history of hair loss
■ Your hair loss is sudden and/or patchy
■ You do not know the reason for your hair loss
■ You are under 18 years of age. Do not use on babies and children.
■ Your scalp is red, inflamed, infected, irritated, or painful
■ You use other medicines on the scalp
- ASK DOCTOR
-
WHEN USING
When using this product
■ Do not apply on other parts of the body
■ Avoid contact with the eyes. In case of accidental contact, rinse eyes with large amounts of cool tap water.
■ Some people have experienced chances in hair color and/or texture
■ It takes time to regrow hair. Results may occur at 2 months with twice a day usage. For some men, you may need to use this product
for at least 4 months before you see results.
■ The amount of hair regrowth is different for each person. This product will not work for all men.
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
-
OTHER INFORMATION
• see hair loss pictures on side of this carton
• before use, read all information on carton and enclosed leaflet
• keep the carton. It contains important information.
• hair regrowth has not been shown to last longer than 48 weeks in large clinical trials with
continuous treatment with 5% minoxidil topical solution for men
•in clinical studies of mostly white men aged 18-49 years with moderate degrees of hair loss, minoxidil topical solution 5%
for men provided more hair regrowth than minoxidil topical solution 2%
• store at controlled room temperature 20º to 25ºC (68º to 77ºF)
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BOURN HAIR REGROWTH TREATMENT FOR MEN
minoxidil 5% solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81553-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MINOXIDIL (UNII: 5965120SH1) (MINOXIDIL - UNII:5965120SH1) MINOXIDIL 5 g in 100 mL Inactive Ingredients Ingredient Name Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81553-001-01 6 in 1 BOX 02/04/2021 1 60 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076239 02/04/2021 Labeler - Octri, Inc. (070111400)