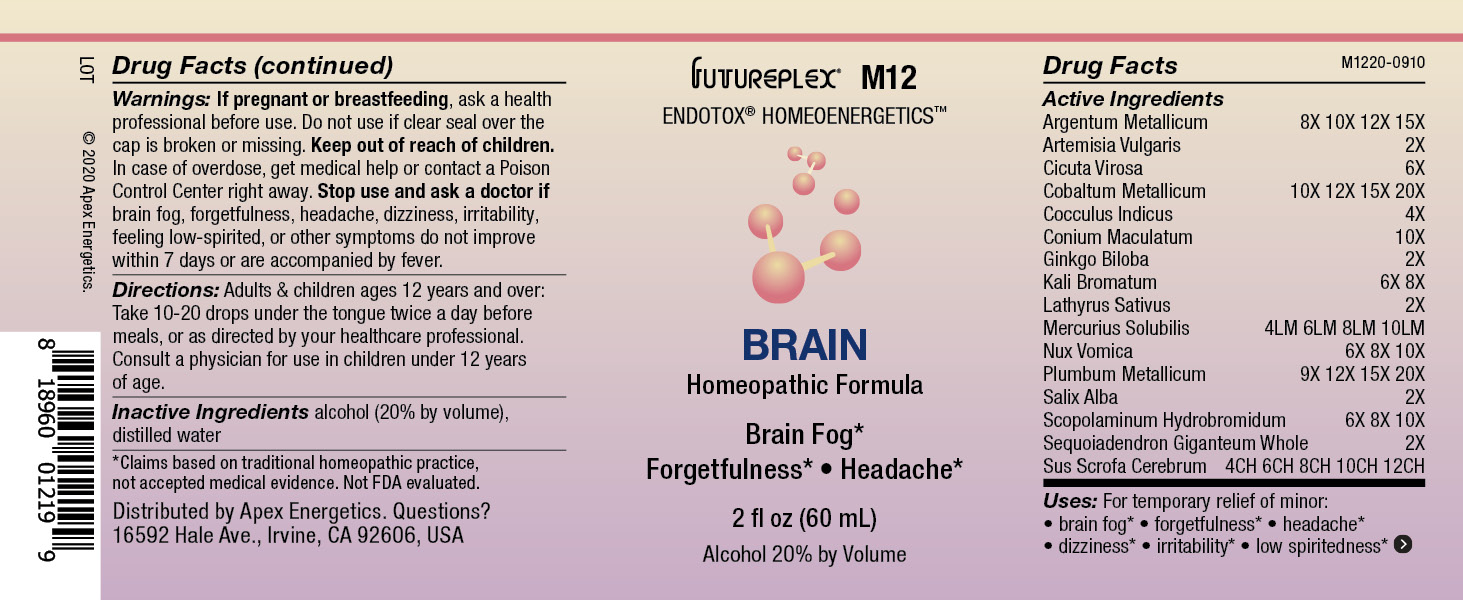
Label: M12 BRAIN- argentum metallicum, artemisia vulgaris, cicuta virosa, cobaltum metallicum, cocculus indicus, conium maculatum, ginkgo biloba, kali bromatum, lathyrus sativus, mercurius solubilis, nux vomica, plumbum metallicum, salix alba, scopolaminum hydrobromidum, sequoiadendron giganteum, sus scrofa cerebrum solution/ drops
- NDC Code(s): 63479-1312-1
- Packager: Apex Energetics Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated January 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active Ingredients
Argentum Metallicum
8X 10X 12X 15X
Artemisia Vulgaris
2X
Cicuta Virosa
6X
Cobaltum Metallicum
10X 12X 15X 20X
Cocculus Indicus
4X
Conium Maculatum
10X
Ginkgo Biloba
2X
Kali Bromatum
6X 8X
Lathyrus Sativus
2X
Mercurius Solubilis
4LM 6LM 8LM 10LM
Nux Vomica
6X 8X 10X
Plumbum Metallicum
9X 12X 15X 20X
Salix Alba
2X
Scopolaminum Hydrobromidum
6X 8X 10X
Sequoiadendron Giganteum Whole
2X
Sus Scrofa Cerebrum
4CH 6CH 8CH 10CH 12CH
- INDICATIONS & USAGE
- Warnings:
- Directions:
- Inactive Ingredients
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
M12 BRAIN
argentum metallicum, artemisia vulgaris, cicuta virosa, cobaltum metallicum, cocculus indicus, conium maculatum, ginkgo biloba, kali bromatum, lathyrus sativus, mercurius solubilis, nux vomica, plumbum metallicum, salix alba, scopolaminum hydrobromidum, sequoiadendron giganteum, sus scrofa cerebrum solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63479-1312 Route of Administration SUBLINGUAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SEQUOIADENDRON GIGANTEUM WHOLE (UNII: 6CO90XQD9O) (SEQUOIADENDRON GIGANTEUM WHOLE - UNII:6CO90XQD9O) SEQUOIADENDRON GIGANTEUM WHOLE 2 [hp_X] in 1 mL MERCURIUS SOLUBILIS (UNII: 324Y4038G2) (MERCURIUS SOLUBILIS - UNII:324Y4038G2) MERCURIUS SOLUBILIS 10 [hp_Q] in 1 mL LEAD (UNII: 2P299V784P) (LEAD - UNII:2P299V784P) LEAD 20 [hp_X] in 1 mL SALIX ALBA BARK (UNII: 205MXS71H7) (SALIX ALBA BARK - UNII:205MXS71H7) SALIX ALBA BARK 2 [hp_X] in 1 mL SCOPOLAMINE HYDROBROMIDE (UNII: 451IFR0GXB) (SCOPOLAMINE - UNII:DL48G20X8X) SCOPOLAMINE HYDROBROMIDE 10 [hp_X] in 1 mL SUS SCROFA CEREBRUM (UNII: 4GB5DQR532) (SUS SCROFA CEREBRUM - UNII:4GB5DQR532) SUS SCROFA CEREBRUM 12 [hp_C] in 1 mL CICUTA VIROSA ROOT (UNII: YEA9P21S8N) (CICUTA VIROSA ROOT - UNII:YEA9P21S8N) CICUTA VIROSA ROOT 6 [hp_X] in 1 mL SILVER (UNII: 3M4G523W1G) (SILVER - UNII:3M4G523W1G) SILVER 15 [hp_X] in 1 mL COBALT (UNII: 3G0H8C9362) (COBALT - UNII:3G0H8C9362) COBALT 20 [hp_X] in 1 mL GINKGO (UNII: 19FUJ2C58T) (GINKGO - UNII:19FUJ2C58T) GINKGO 2 [hp_X] in 1 mL ARTEMISIA VULGARIS ROOT (UNII: 32MP823R8S) (ARTEMISIA VULGARIS ROOT - UNII:32MP823R8S) ARTEMISIA VULGARIS ROOT 2 [hp_X] in 1 mL STRYCHNOS NUX-VOMICA SEED (UNII: 269XH13919) (STRYCHNOS NUX-VOMICA SEED - UNII:269XH13919) STRYCHNOS NUX-VOMICA SEED 10 [hp_X] in 1 mL ANAMIRTA COCCULUS SEED (UNII: 810258W28U) (ANAMIRTA COCCULUS SEED - UNII:810258W28U) ANAMIRTA COCCULUS SEED 4 [hp_X] in 1 mL CONIUM MACULATUM FLOWERING TOP (UNII: Q28R5GF371) (CONIUM MACULATUM FLOWERING TOP - UNII:Q28R5GF371) CONIUM MACULATUM FLOWERING TOP 10 [hp_X] in 1 mL POTASSIUM BROMIDE (UNII: OSD78555ZM) (BROMIDE ION - UNII:952902IX06) POTASSIUM BROMIDE 8 [hp_X] in 1 mL LATHYRUS SATIVAS SEED (UNII: 8VP54WOT4I) (LATHYRUS SATIVAS SEED - UNII:8VP54WOT4I) LATHYRUS SATIVAS SEED 2 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63479-1312-1 60 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 03/15/1993 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 03/15/1993 Labeler - Apex Energetics Inc (195816384)