Label: STOMACH RELIEF- bismuth subsalicylate tablet, chewable
- NDC Code(s): 21130-241-30
- Packager: SAFEWAY
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 14, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each chewable tablet)
- Purpose
- Uses
-
Warnings
Reye's syndrome
Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea or vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert
Do not use if you are
- ▪
- allergic to salicylate (including aspirin)
- ▪
- taking other salicylate products
Ask a doctor or pharmacist before use if you are taking any drug for
- ▪
- anticoagulation (thinning of the blood)
- ▪
- diabetes
- ▪
- gout
- ▪
- arthritis
-
Directions
- ▪
- chew completely or dissolve tablets in mouth. Do not swallow tablets whole.
- ▪
- adults and children 12 years and over:
- ▪
- 2 tablets (1 dose) every ½ hour to 1 hour or 4 tablets (2 doses) every hour as needed for diarrhea
- ▪
- 2 tablets (1 dose) every ½ hour as needed for overindulgence (upset stomach, heartburn, indigestion, nausea)
- ▪
- do not exceed 8 doses (16 tablets) in 24 hours
- ▪
- use until diarrhea stops but not more than 2 days
- ▪
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- ▪
- children under 12 years: ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
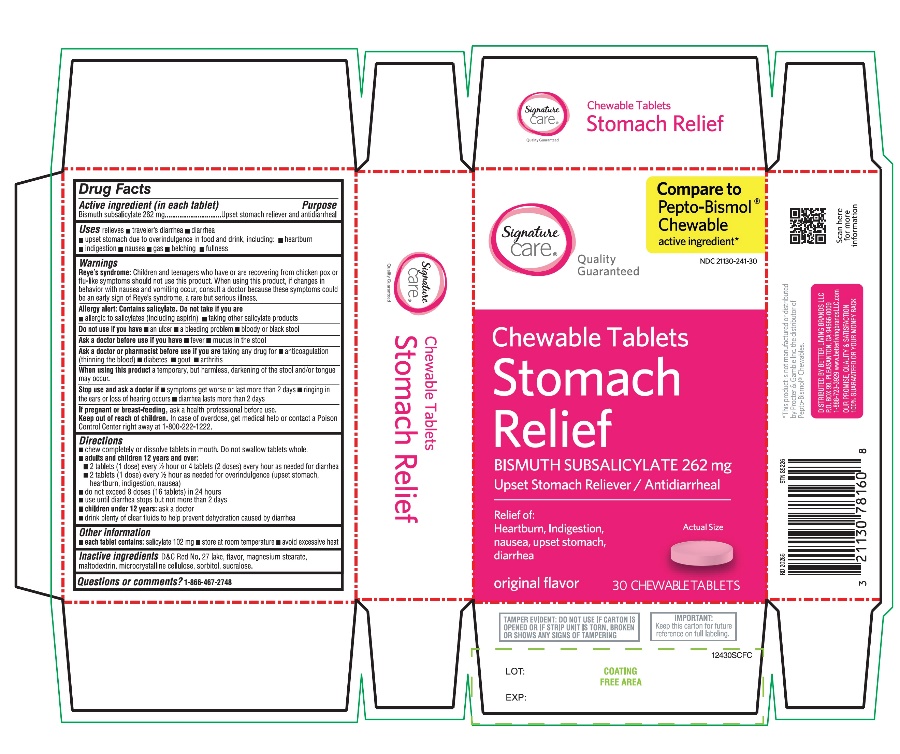
Principal Display Panel
Compare to the active ingredient in Pepto-Bismol® Chewables*
Stomach Relief
Chewable Tablets
Upset Stomach Reliever/Antidiarrheal
Original flavor
5 Symptoms Digestive Relief:
- •
- Heartburn
- •
- Indigestion
- •
- Nausea
- •
- Upset Stomach
- •
- Diarrhea
30 Chewable Tablets
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR OF STRIP UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed by Procter & Gamble Inc., the distributor of Pepto-Bismol® Chewables.
Distributed by:
-
INGREDIENTS AND APPEARANCE
STOMACH RELIEF
bismuth subsalicylate tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21130-241 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BISMUTH SUBSALICYLATE (UNII: 62TEY51RR1) (BISMUTH CATION - UNII:ZS9CD1I8YE, SALICYLIC ACID - UNII:O414PZ4LPZ) BISMUTH SUBSALICYLATE 262 mg Inactive Ingredients Ingredient Name Strength D&C RED NO. 27 (UNII: 2LRS185U6K) magnesium stearate (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SORBITOL (UNII: 506T60A25R) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color PINK (light-pink) Score no score Shape ROUND Size 16mm Flavor Imprint Code RP124 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21130-241-30 5 in 1 CARTON 10/01/2020 1 6 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M008 10/01/2020 Labeler - SAFEWAY (009137209)