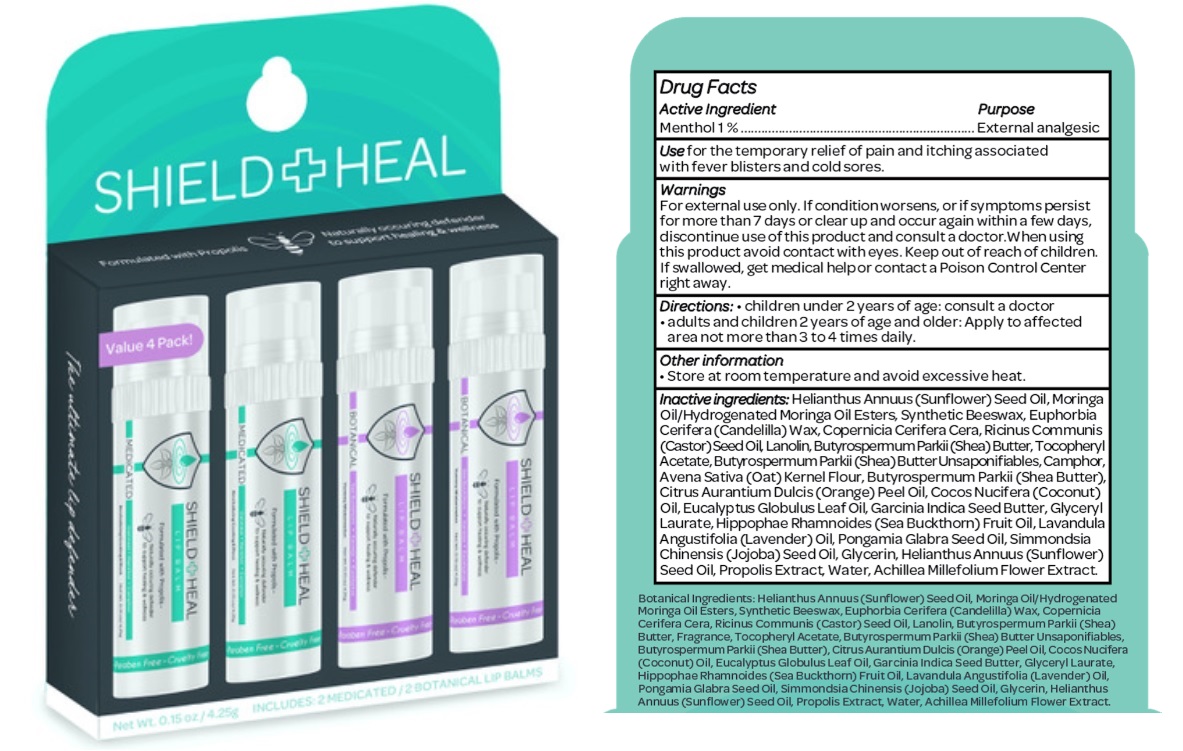
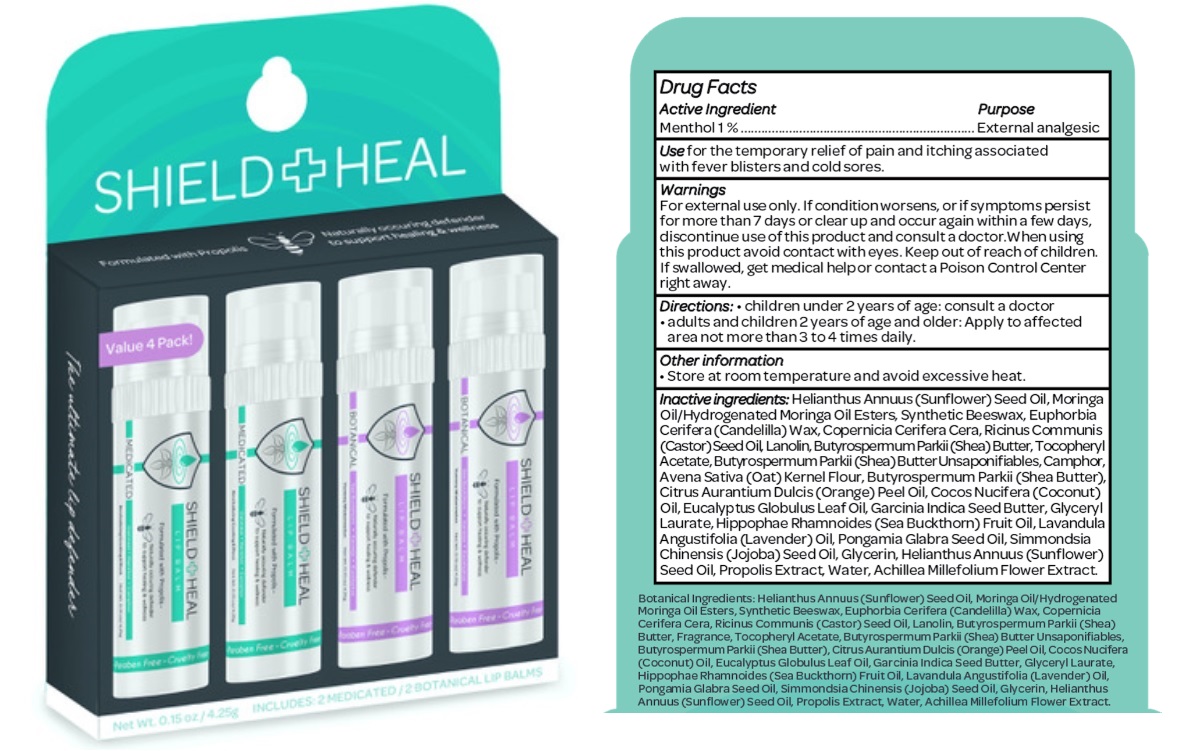
Label: SHIELD AND HEAL LIP BALM- menthol kit
- NDC Code(s): 81470-002-01
- Packager: Shield and Heal Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Purpose
- Use
- Warnings
- Directions
- Other information
-
Inactive ingredients
Helianthus Annuus (Sunflower) Seed Oil, Moringa Oil/Hydrogenated Moringa Oil Esters, Synthetic Beeswax, Euphorbia Cerifera (Candelilla) Wax, Copernicia Cerifera Cera, Ricinus Communis (Castor) Seed Oil, Lanolin, Butyrospermum Parkii (Shea) Butter, Tocopheryl Acetate, Butyrospermum Parkii (Shea) Butter Unsaponifiables, Camphor, Avena Sativa (Oat) Kernel Flour, Butyrospermum Parkii (Shea Butter), Citrus Aurantium Dulcis (Orange) Peel Oil, Cocos Nucifera (Coconut) Oil, Eucalyptus Globulus Leaf Oil, Garcinia Indica Seed Butter, Glyceryl Laurate, Hippophae Rhamnoides (Sea Buckthorn) Fruit Oil, Lavandula Angustifolia (Lavender) Oil, Pongamia Glabra Seed Oil, Simmondsia Chinensis (Jojoba) Seed Oil, Glycerin, Helianthus Annuus (Sunflower) Seed Oil, Propolis Extract, Water, Achillea Millefolium Flower Extract.
- Product Packaging
-
INGREDIENTS AND APPEARANCE
SHIELD AND HEAL LIP BALM
menthol kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81470-002 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81470-002-01 1 in 1 KIT; Type 0: Not a Combination Product 02/15/2021 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 4.25 g Part 2 1 TUBE 4.25 g Part 1 of 2 SHIELD AND HEAL MEDICATED LIP BALM
menthol stickProduct Information Item Code (Source) NDC:81470-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 10 mg in 1 g Inactive Ingredients Ingredient Name Strength SYNTHETIC BEESWAX (UNII: 08MNR5YE2R) CARNAUBA WAX (UNII: R12CBM0EIZ) SHEA BUTTER (UNII: K49155WL9Y) BUTYROSPERMUM PARKII (SHEA) BUTTER UNSAPONIFIABLES (UNII: 0C9AC7D6XU) OATMEAL (UNII: 8PI54V663Y) ORANGE OIL (UNII: AKN3KSD11B) COCONUT OIL (UNII: Q9L0O73W7L) KARUM SEED OIL (UNII: 62160PU6FJ) GARCINIA INDICA SEED BUTTER (UNII: US2H3D7800) LAVENDER OIL (UNII: ZBP1YXW0H8) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) CASTOR OIL (UNII: D5340Y2I9G) GLYCERYL LAURATE (UNII: Y98611C087) SUNFLOWER OIL (UNII: 3W1JG795YI) LANOLIN (UNII: 7EV65EAW6H) CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) EUCALYPTUS OIL (UNII: 2R04ONI662) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) CANDELILLA WAX (UNII: WL0328HX19) ACHILLEA MILLEFOLIUM FLOWER (UNII: YQR8R0SQEA) HIPPOPHAE RHAMNOIDES FRUIT OIL (UNII: TA4JCF9S1J) JOJOBA OIL (UNII: 724GKU717M) PROPOLIS WAX (UNII: 6Y8XYV2NOF) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 2 in 1 KIT 1 4.25 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 02/15/2021 Part 2 of 2 SHIELD AND HEAL BOTANICAL LIP BALM
lipstick stickProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR SYNTHETIC BEESWAX (UNII: 08MNR5YE2R) INGR CARNAUBA WAX (UNII: R12CBM0EIZ) INGR SHEA BUTTER (UNII: K49155WL9Y) INGR BUTYROSPERMUM PARKII (SHEA) BUTTER UNSAPONIFIABLES (UNII: 0C9AC7D6XU) INGR ORANGE OIL (UNII: AKN3KSD11B) INGR COCONUT OIL (UNII: Q9L0O73W7L) INGR KARUM SEED OIL (UNII: 62160PU6FJ) INGR GARCINIA INDICA SEED BUTTER (UNII: US2H3D7800) INGR LAVENDER OIL (UNII: ZBP1YXW0H8) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR WATER (UNII: 059QF0KO0R) INGR CASTOR OIL (UNII: D5340Y2I9G) INGR GLYCERYL LAURATE (UNII: Y98611C087) INGR SUNFLOWER OIL (UNII: 3W1JG795YI) INGR LANOLIN (UNII: 7EV65EAW6H) INGR EUCALYPTUS OIL (UNII: 2R04ONI662) INGR .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) INGR CANDELILLA WAX (UNII: WL0328HX19) INGR ACHILLEA MILLEFOLIUM FLOWER (UNII: YQR8R0SQEA) INGR HIPPOPHAE RHAMNOIDES FRUIT OIL (UNII: TA4JCF9S1J) INGR JOJOBA OIL (UNII: 724GKU717M) INGR PROPOLIS WAX (UNII: 6Y8XYV2NOF) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 2 in 1 KIT 1 4.25 g in 1 TUBE; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Cosmetic 02/15/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 02/15/2021 Labeler - Shield and Heal Inc. (117700988)