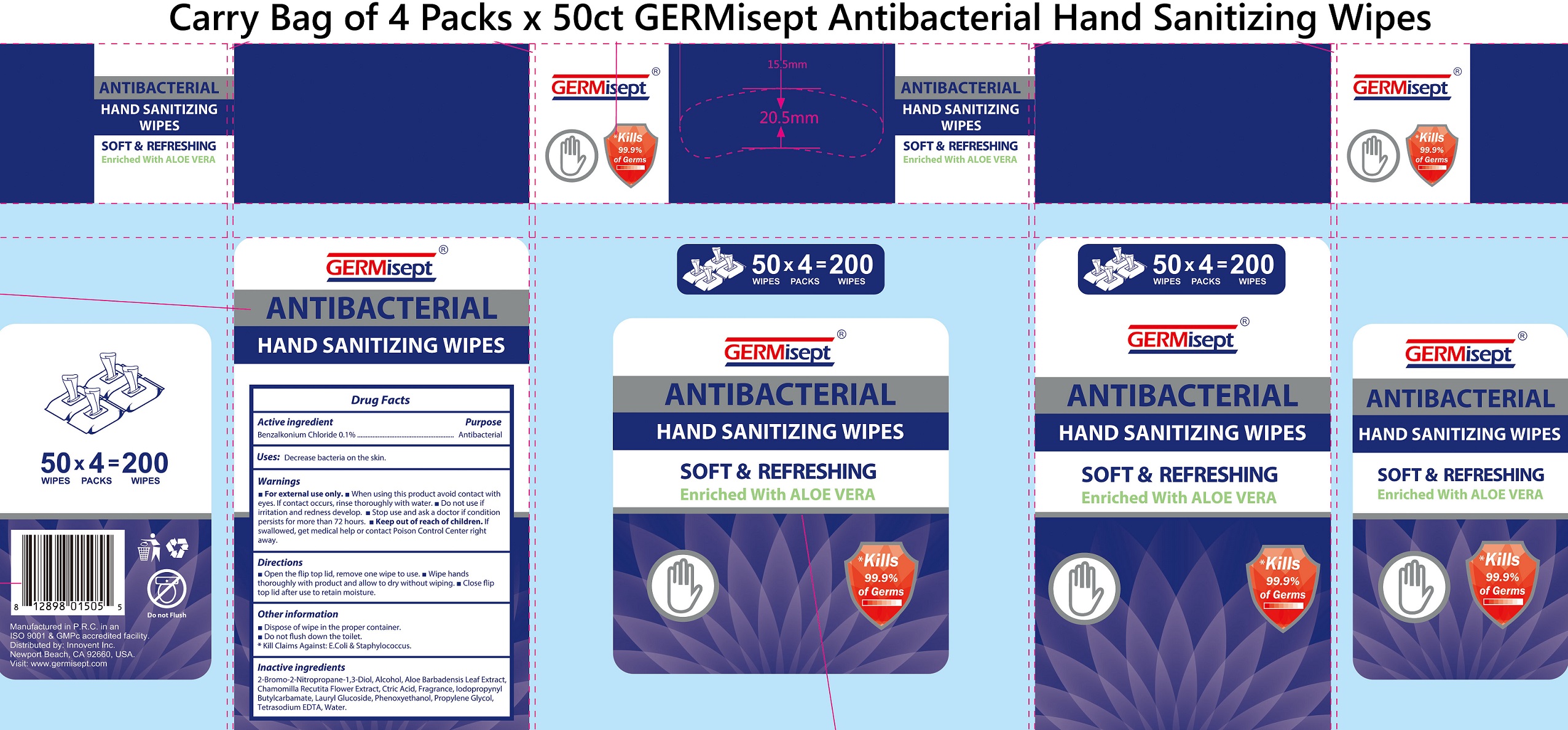
Label: GERMISEPT ANTIBACTERIAL HAND SANITIZING WIPES- benzalkonium chloride cloth
- NDC Code(s): 70335-012-01
- Packager: Innovent Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 21, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Uses:
- Warnings
- Directions
- Other information
- Inactive ingredients
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
GERMISEPT ANTIBACTERIAL HAND SANITIZING WIPES
benzalkonium chloride clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70335-012 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength BRONOPOL (UNII: 6PU1E16C9W) ALCOHOL (UNII: 3K9958V90M) ALOE VERA LEAF (UNII: ZY81Z83H0X) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) IODOPROPYNYL BUTYLCARBAMATE (UNII: 603P14DHEB) LAURYL GLUCOSIDE (UNII: 76LN7P7UCU) PHENOXYETHANOL (UNII: HIE492ZZ3T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) EDETATE SODIUM (UNII: MP1J8420LU) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70335-012-01 4 in 1 BAG 02/10/2021 1 50 in 1 PACKAGE 1 4.3 mL in 1 PATCH; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 02/10/2021 Labeler - Innovent Inc (079973489)