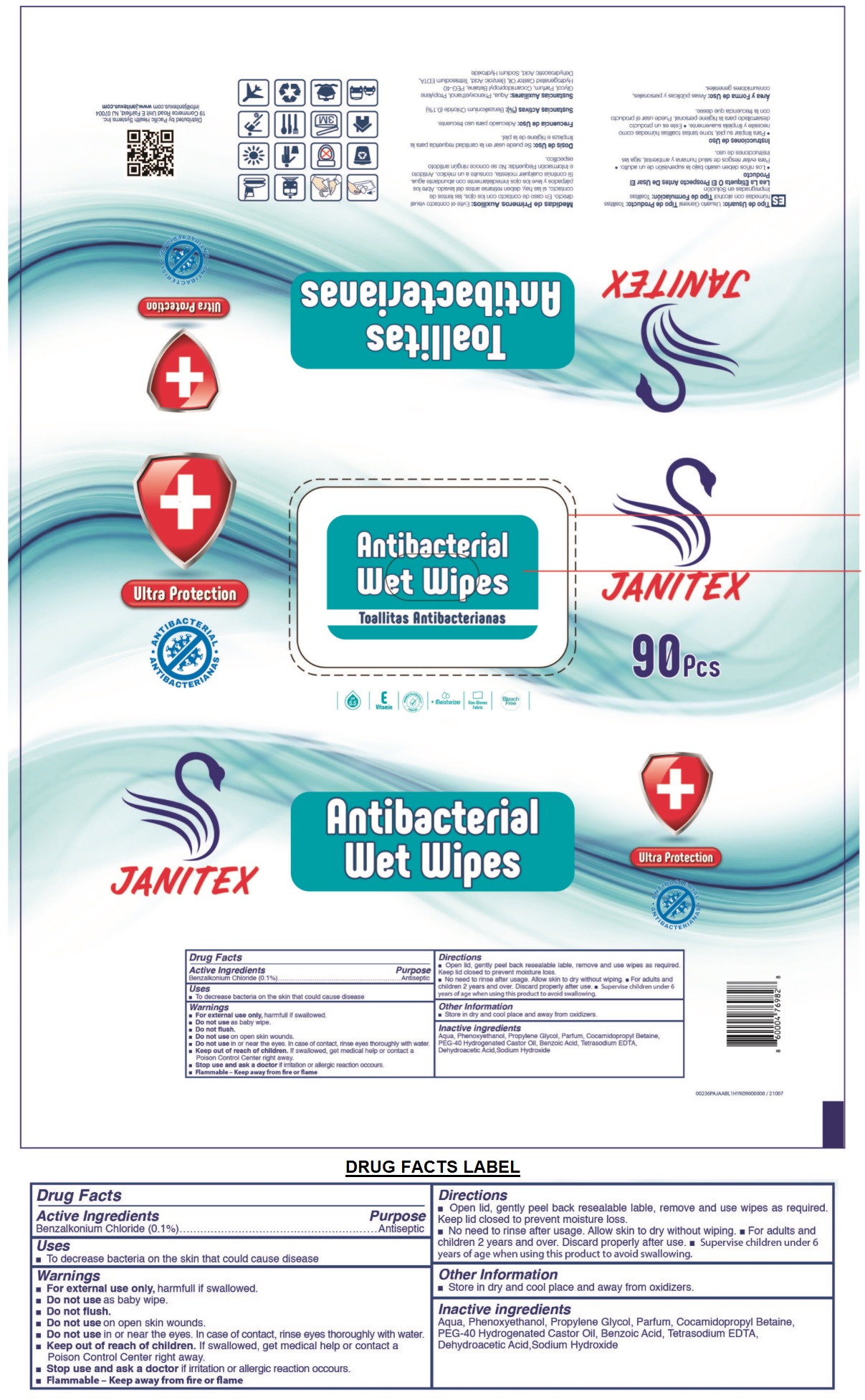
Label: JANITEX ANTIBACTERIAL WET WIPES- benzalkonium chloride cloth
-
Contains inactivated NDC Code(s)
NDC Code(s): 79519-050-01 - Packager: Pacific Health Systems Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 11, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients
- Purpose
- Uses
-
Warnings
- For external use only, harmful if swallowed.
- Do not use as baby wipe.
- Do not flush.
- Do not use on open skin wounds.
- Do not use in or near the eyes. In case of contact, rinse eyes thoroughly with water.
- Stop use and ask a doctor if irritation or allergic reaction occurs.
- Flammable - Keep away from fire or flame
-
Directions
• Open lid, gently peel back resealable label, remove and use wipes as required. Keep lid closed to prevent moisture loss.
• No need to rinse after usage. Allow skin to dry without wiping. • For adults and children 2 years and over. Discard properly after use. • Supervise children under 6 years of age when using this product to avoid swallowing.
- Other Information
- Inactive ingredients
- SPL UNCLASSIFIED SECTION
- Packaging
-
INGREDIENTS AND APPEARANCE
JANITEX ANTIBACTERIAL WET WIPES
benzalkonium chloride clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:79519-050 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.1 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PHENOXYETHANOL (UNII: HIE492ZZ3T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) POLYOXYL 40 HYDROGENATED CASTOR OIL (UNII: 7YC686GQ8F) BENZOIC ACID (UNII: 8SKN0B0MIM) EDETATE SODIUM (UNII: MP1J8420LU) DEHYDROACETIC ACID (UNII: 2KAG279R6R) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:79519-050-01 90 in 1 PACKET 08/11/2020 1 3.23 g in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 08/11/2020 Labeler - Pacific Health Systems Inc. (080638960)