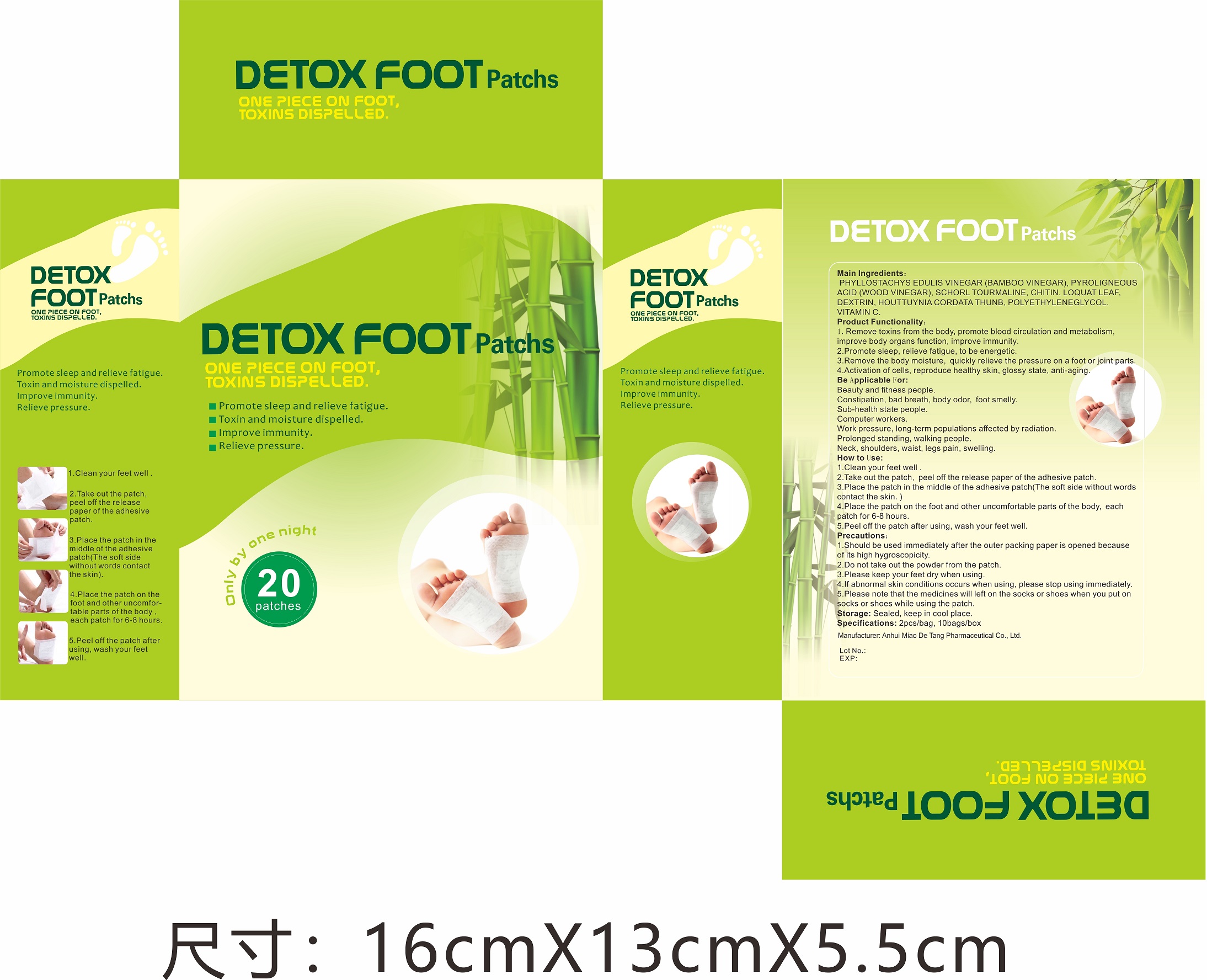
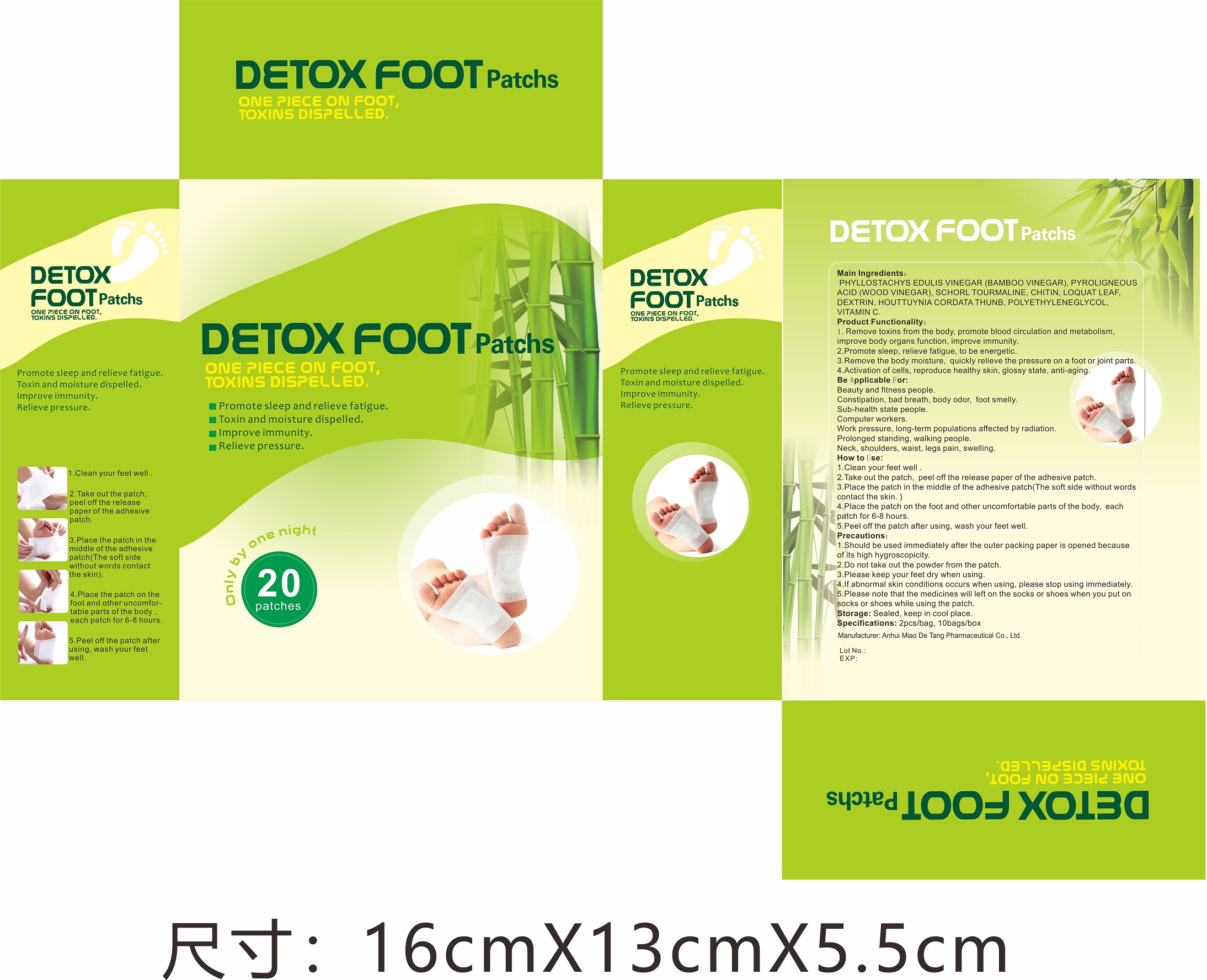
Label: DETOX FOOT PATCH (phyllostachys edulis vinegar(bamboo vinegar), pyroligneous acid- wood vinegar, schorl tourmaline, houttuynia cordata thunb patch
- NDC Code(s): 81484-101-01, 81484-101-02, 81484-101-03
- Packager: Anhui Miao De Tang Pharmaceutical Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated July 4, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient(s)
- Purpose
- Use
- Warnings
- Do not use
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
Directions
Clean your feet well.
Take out the patch, peel off the release paper of the adhesive patch.
Place the patch in the middle of the adhesive patch(The soft side without words contact the skin.)
Plase the patch on the foot and other uncomfortable parts of the body, each patch for 6-8 hours.
Peel off the patch after using, wash your fee well.
- Other information
- Inactive ingredients
- Package Label - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
DETOX FOOT PATCH
phyllostachys edulis vinegar(bamboo vinegar), pyroligneous acid(wood vinegar), schorl tourmaline, houttuynia cordata thunb patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81484-101 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOUTTUYNIA CORDATA WHOLE (UNII: O3E12ZLW5T) (HOUTTUYNIA CORDATA WHOLE - UNII:O3E12ZLW5T) HOUTTUYNIA CORDATA WHOLE 0.7875 g PYROLIGNEOUS ACID (UNII: N4G9GAT76C) (PYROLIGNEOUS ACID - UNII:N4G9GAT76C) PYROLIGNEOUS ACID 1.75 g SCHORL TOURMALINE (UNII: 173O8XLY6T) (SCHORL TOURMALINE - UNII:173O8XLY6T) SCHORL TOURMALINE 0.2625 g PHYLLOSTACHYS EDULIS VINEGAR (UNII: MR94DK8ZSM) (PHYLLOSTACHYS EDULIS VINEGAR - UNII:MR94DK8ZSM) PHYLLOSTACHYS EDULIS VINEGAR 2.625 g Inactive Ingredients Ingredient Name Strength POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) ASCORBIC ACID (UNII: PQ6CK8PD0R) CHITIN (UNII: 8SH93A7QWW) ERIOBOTRYA JAPONICA LEAF (UNII: Z02066SV11) DEXTRIN PALMITATE (CORN; 20000 MW) (UNII: 89B2BSF9I3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81484-101-01 20 in 1 BOX; Type 0: Not a Combination Product 01/29/2021 2 NDC:81484-101-02 2 in 1 BAG; Type 0: Not a Combination Product 01/29/2021 3 NDC:81484-101-03 10 in 1 BAG; Type 0: Not a Combination Product 01/29/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 01/29/2021 Labeler - Anhui Miao De Tang Pharmaceutical Co., Ltd. (405744102) Establishment Name Address ID/FEI Business Operations Anhui Miao De Tang Pharmaceutical Co., Ltd. 405744102 manufacture(81484-101)