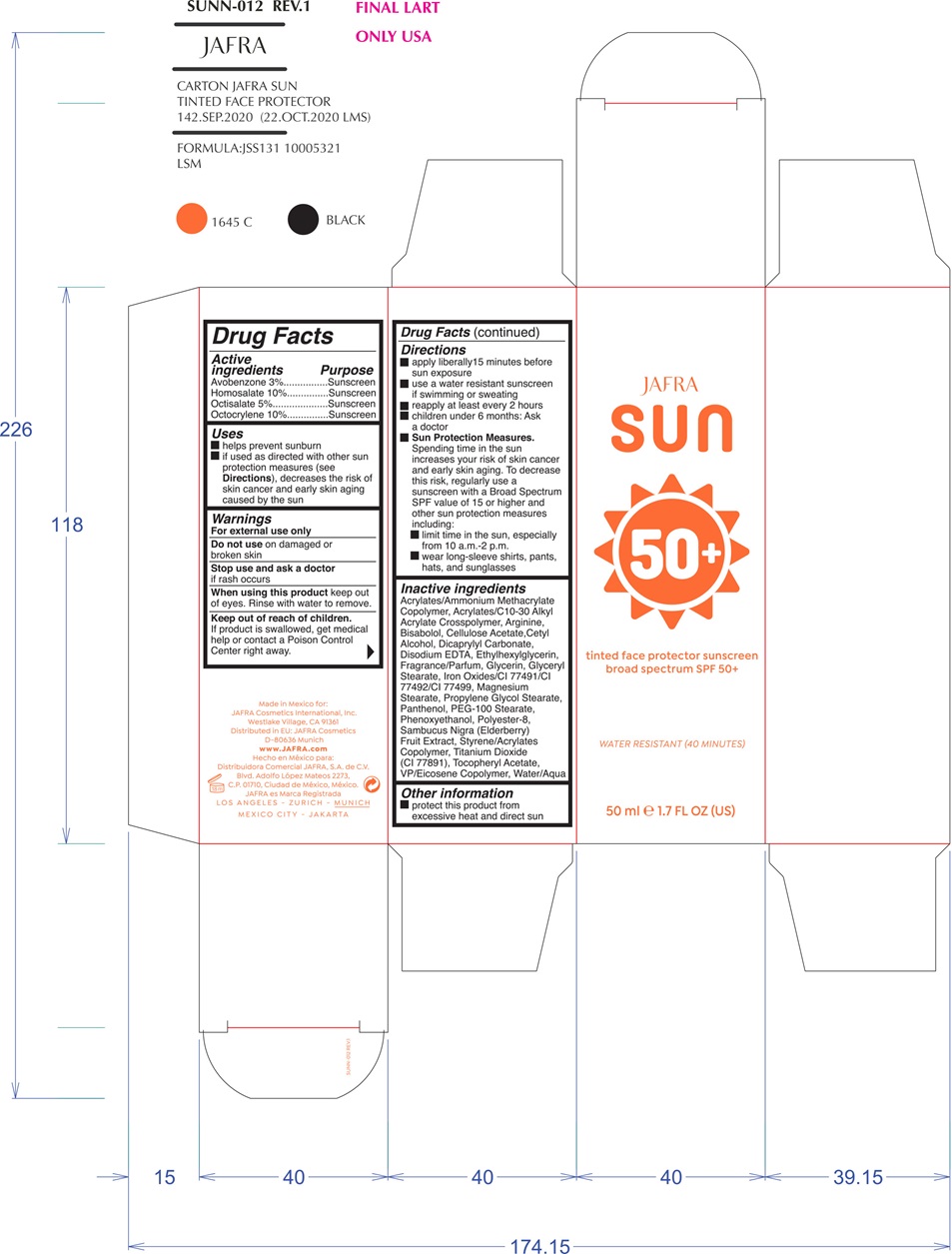
Label: JAFRA SUN TINTED FACE PROTECTOR SUNSCREEN BROAD SPECTRUM 50- avobenzone, homosalate, octisalate, octocrylene cream
- NDC Code(s): 68828-293-01, 68828-293-02
- Packager: Jafra Cosmetics Internationl
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 29, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
-
DOSAGE & ADMINISTRATION
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- children under 6 months: Ask a doctor
- Sun Protection Measures.Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
-
INACTIVE INGREDIENT
Acrylates/Ammonium Methacrylate Copolymer, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Arginine, Bisabolol, Cellulose Acetate, Cetyl Alcohol, Dicaprylyl Carbonate, Disodium EDTA, Ethylhexylglycerin, Fragrance/Parfum, Glycerin, Glyceryl Stearate, Iron Oxides/CI 77491/CI 77492/CI 77499, Magnesium Stearate, Propylene Glycol Stearate, Panthenol, PEG-100 Stearate, Phenoxyethanol, Polyester-8, Sambucus Nigra (Elderberry) Fruit Extract, Styrene/Acrylates Copolymer, Titanium Dioxide (CI 77891 ), Tocopheryl Acetate, VP/Eicosene Copolymer, Water/Aqua
- OTHER SAFETY INFORMATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
JAFRA SUN TINTED FACE PROTECTOR SUNSCREEN BROAD SPECTRUM 50
avobenzone, homosalate, octisalate, octocrylene creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68828-293 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 100 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 10 g in 100 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 10 g in 100 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5 g in 100 mL Inactive Ingredients Ingredient Name Strength GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) POLYESTER-8 (1400 MW, CYANODIPHENYLPROPENOYL CAPPED) (UNII: T9296U138P) EUROPEAN ELDERBERRY (UNII: BQY1UBX046) FERRIC OXIDE RED (UNII: 1K09F3G675) PEG-100 STEARATE (UNII: YD01N1999R) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE ACETATE (UNII: 3J2P07GVB6) CETYL ALCOHOL (UNII: 936JST6JCN) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) VINYLPYRROLIDONE/EICOSENE COPOLYMER (UNII: 035MV9S1C3) WATER (UNII: 059QF0KO0R) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) GLYCERIN (UNII: PDC6A3C0OX) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) CARBOMER INTERPOLYMER TYPE A (ALLYL SUCROSE CROSSLINKED) (UNII: 59TL3WG5CO) PANTHENOL (UNII: WV9CM0O67Z) PHENOXYETHANOL (UNII: HIE492ZZ3T) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) PROPYLENE GLYCOL STEARATE (UNII: MZM1I680W0) STYRENE/ACRYLAMIDE COPOLYMER (MW 500000) (UNII: 5Z4DPO246A) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) ARGININE (UNII: 94ZLA3W45F) LEVOMENOL (UNII: 24WE03BX2T) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68828-293-01 50 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/14/2021 2 NDC:68828-293-02 7.5 mL in 1 BOTTLE; Type 0: Not a Combination Product 01/14/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/14/2021 Labeler - Jafra Cosmetics Internationl (041676479) Establishment Name Address ID/FEI Business Operations Distribuidora Comercial Jafra, S.A. de C.V. 951612777 manufacture(68828-293)