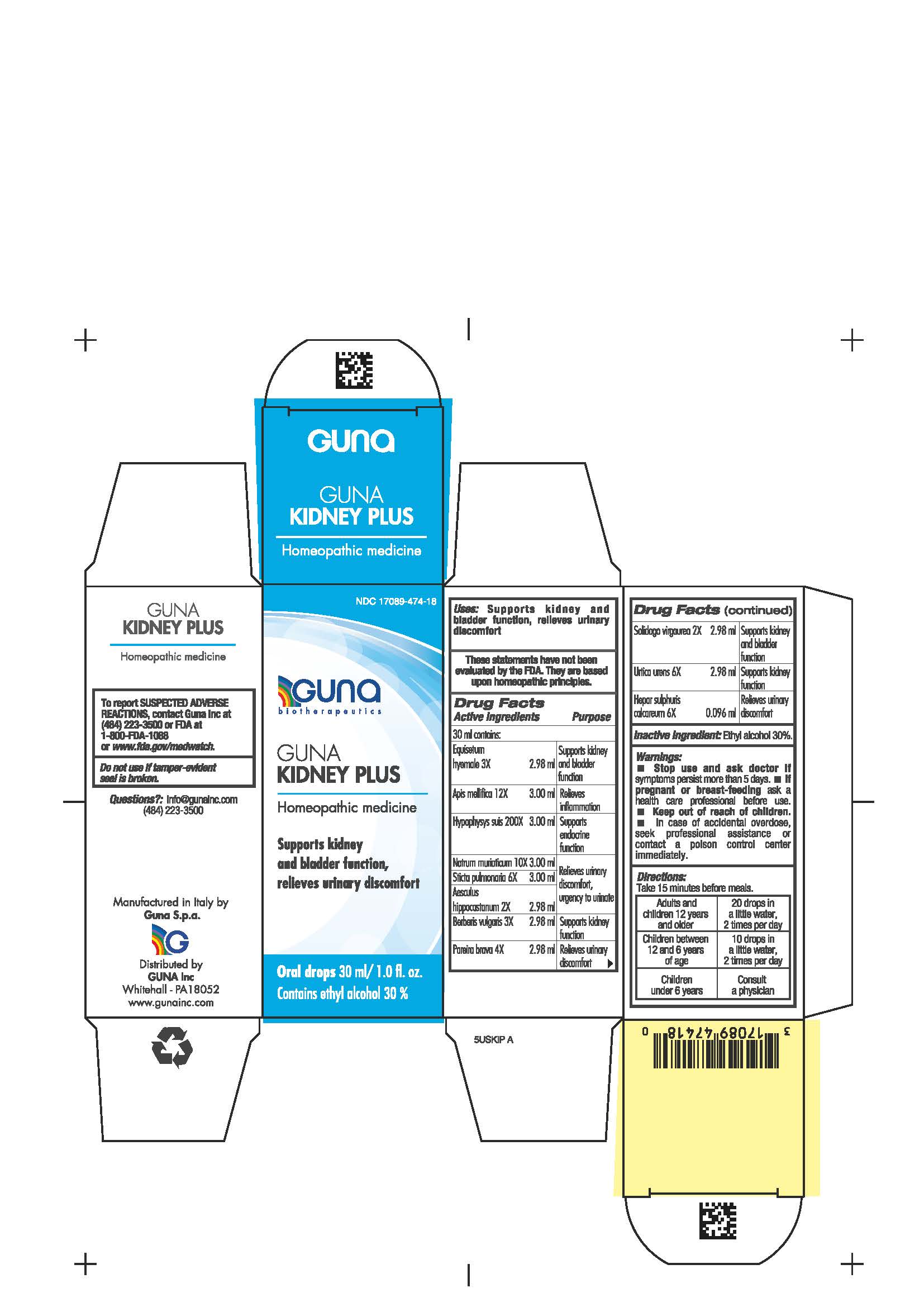
Label: GUNA KIDNEY PLUS- calcium sulfide - chondrodendron tomentosum root - equisetum hyemale - sus scrofa pituitary gland - apis mellifera - urtica urens - sodium chloride - lobaria pulmonaria - horse chestnut - berberis vulgaris fruit - solidago viragaurea flowering top - solution/ drops
- NDC Code(s): 17089-474-18
- Packager: Guna spa
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated February 4, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- INDICATIONS & USAGE
- INACTIVE INGREDIENT
- QUESTIONS
- DIRECTIONS
- WARNINGS
- PREGNANCY
- WARNINGS
- USES
-
ACTIVE INGREDIENTS/PURPOSE
Equisetum hyemale 3X Supports kidney and bladder function
Apis mellifica 12X Relieves inflammation
Hypophysys suis 200X Supports endocrine function
Natrum muriaticum 10X Relieves urinary discomfort, urgency to urinate
Sticta pulmonaria 6X Relieves urinary discomfort, urgency to urinate
Aesculus hippocastanum 2X Relieves urinary discomfort, urgency to urinate
Berberis vulgaris 3X Supports kidney function
Pareira brava 4X Relieves urinary discomfort
Solidago virgaurea 2X Supports kidney and bladder function
Urtica urens 6X Supports kidney function
Hepar sulphuris calcareum 6X Relieves urinary discomfort
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GUNA KIDNEY PLUS
calcium sulfide - chondrodendron tomentosum root - equisetum hyemale - sus scrofa pituitary gland - apis mellifera - urtica urens - sodium chloride - lobaria pulmonaria - horse chestnut - berberis vulgaris fruit - solidago viragaurea flowering top - solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:17089-474 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM SULFIDE (UNII: 1MBW07J51Q) (CALCIUM SULFIDE - UNII:1MBW07J51Q) CALCIUM SULFIDE 6 [hp_X] in 30 mL EQUISETUM HYEMALE (UNII: 59677RXH25) (EQUISETUM HYEMALE - UNII:59677RXH25) EQUISETUM HYEMALE 3 [hp_X] in 30 mL SUS SCROFA PITUITARY GLAND (UNII: L0PFEMQ1DT) (SUS SCROFA PITUITARY GLAND - UNII:L0PFEMQ1DT) SUS SCROFA PITUITARY GLAND 200 [hp_X] in 30 mL APIS MELLIFERA (UNII: 7S82P3R43Z) (APIS MELLIFERA - UNII:7S82P3R43Z) APIS MELLIFERA 12 [hp_X] in 30 mL CHONDRODENDRON TOMENTOSUM ROOT (UNII: 395A3P448Z) (CHONDRODENDRON TOMENTOSUM ROOT - UNII:395A3P448Z) CHONDRODENDRON TOMENTOSUM ROOT 4 [hp_X] in 30 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM CHLORIDE 10 [hp_X] in 30 mL LOBARIA PULMONARIA (UNII: D1YM0P5Z2T) (LOBARIA PULMONARIA - UNII:D1YM0P5Z2T) LOBARIA PULMONARIA 6 [hp_X] in 30 mL HORSE CHESTNUT (UNII: 3C18L6RJAZ) (HORSE CHESTNUT - UNII:3C18L6RJAZ) HORSE CHESTNUT 2 [hp_X] in 30 mL BERBERIS VULGARIS FRUIT (UNII: 6XEF22AHC3) (BERBERIS VULGARIS FRUIT - UNII:6XEF22AHC3) BERBERIS VULGARIS FRUIT 3 [hp_X] in 30 mL SOLIDAGO VIRGAUREA FLOWERING TOP (UNII: 5405K23S50) (SOLIDAGO VIRGAUREA FLOWERING TOP - UNII:5405K23S50) SOLIDAGO VIRGAUREA FLOWERING TOP 2 [hp_X] in 30 mL URTICA URENS (UNII: IHN2NQ5OF9) (URTICA URENS - UNII:IHN2NQ5OF9) URTICA URENS 6 [hp_X] in 30 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) 9 mL in 30 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17089-474-18 1 in 1 BOX 01/17/2021 1 30 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 01/17/2021 Labeler - Guna spa (430538264) Establishment Name Address ID/FEI Business Operations Guna spa 338587646 manufacture(17089-474)