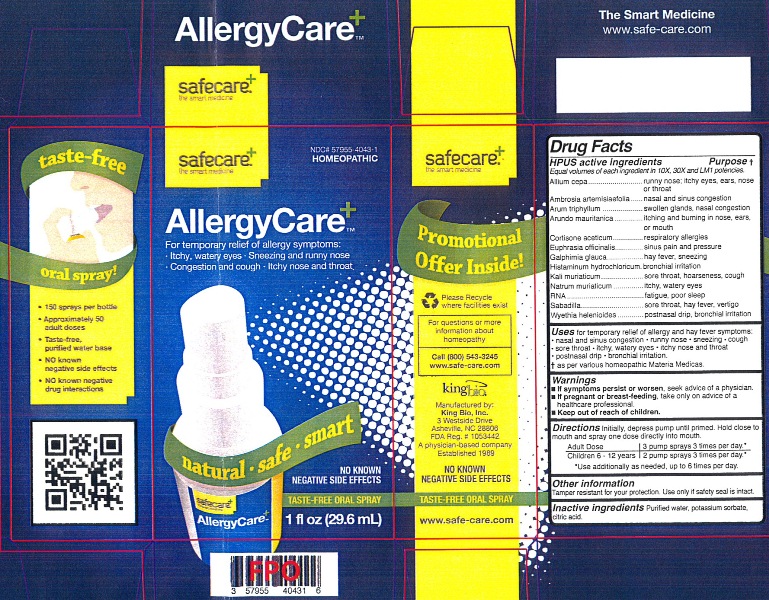
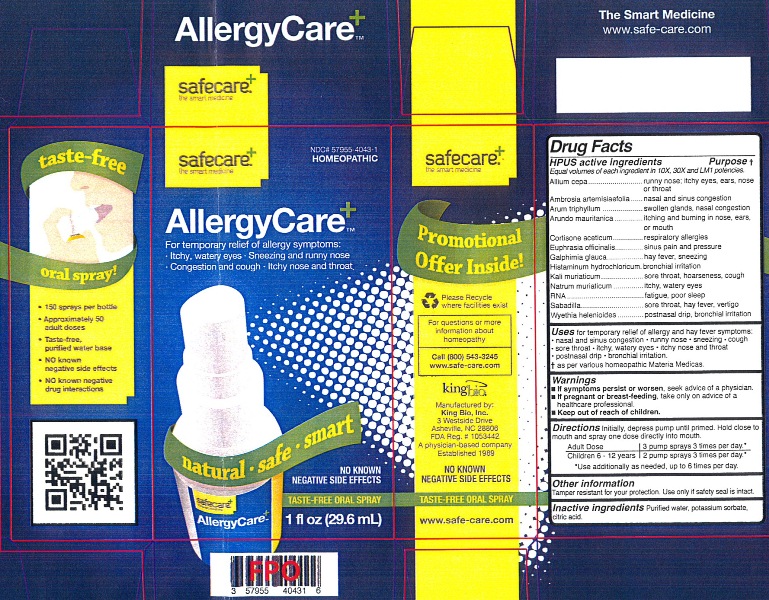
Label: ALLERGYCARE- llium cepa, ambrosia artemisiaefolia, arum triphyllum, arundo mauritanica, cortisone aceticum, euphrasia officinalis, galphimia glauca, histaminum hydrochloricum, kali muriaticum, natrum muriaticum, rna, sabadilla and wyethia helenioides liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 57955-4043-1 - Packager: King Bio Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated June 13, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Active Ingredients
Drug Facts
HPUS active ingredients
Equal volumes of each ingredient in 10X, 30X and LM1 potencies.
Allium cepa, Ambrosia artemisiaefolia, Arum triphyllum, Arundo mauritanica, Cortisone aceticum, Euphrasia officinalis, Galphimia glauca, Histaminum hydrochloricum, Kali muriaticum, Natrum muriaticum, RNA, Sabadilla and Wyethia helenioides.
Reference image allergycare.jpg
- Inactive Ingredients
-
Purpose
Drug Facts
HPUS active ingredients Purpose
Equal volumes of each ingredient in 10X, 30X and LM1 potencies.
Allium cepa...........................................................runny nose; itchy eyes, ears, nose or throat
Ambrosia artemisiaefolia........................................nasal and sinus congestion
Arum triphyllum......................................................swollen glands, nasal congestion
Arundo mauritanica...............................................itching and burning in nose, ears, or mouth
Cortisone aceticum...............................................respiratory allergies
Euphrasia officinalis..............................................sinus pain and pressure
Galphimia glauca...................................................hay fever, sneezing
Histaminum hydrochloricum...................................bronchial irritation
Kali muriaticum.....................................................sore throat, hoarseness, cough
Natrum muriaticum...............................................itchy, watery eyes
RNA....................................................................fatigue, poor sleep
Sabadilla..............................................................sore throat, hay fever, vertigo
Wyethia helenioides............................................postnasal drip, bronchial irritation
Reference image allergycare.jpg
- Dosage and Administration
-
Warnings
Warnings:
- If symptoms persist or worsen, seek advice of a physician.
- If pregnant or breast-feeding, take only on advice of a healthcare professional.
-
Keep out of reach of children.
Reference image allergycare.jpg
- KEEP OUT OF REACH OF CHILDREN
- Indications and Usage
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALLERGYCARE
llium cepa, ambrosia artemisiaefolia, arum triphyllum, arundo mauritanica, cortisone aceticum, euphrasia officinalis, galphimia glauca, histaminum hydrochloricum, kali muriaticum, natrum muriaticum, rna, sabadilla and wyethia helenioides liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57955-4043 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ONION (UNII: 492225Q21H) (ONION - UNII:492225Q21H) ONION 10 [hp_X] in 29.6 mL AMBROSIA ARTEMISIIFOLIA (UNII: 9W34L2CQ9A) (AMBROSIA ARTEMISIIFOLIA - UNII:9W34L2CQ9A) AMBROSIA ARTEMISIIFOLIA 10 [hp_X] in 29.6 mL ARISAEMA TRIPHYLLUM ROOT (UNII: DM64K844DM) (ARISAEMA TRIPHYLLUM ROOT - UNII:DM64K844DM) ARISAEMA TRIPHYLLUM ROOT 10 [hp_X] in 29.6 mL ARUNDO PLINIANA ROOT (UNII: ZXE7LB03WC) (ARUNDO PLINIANA ROOT - UNII:ZXE7LB03WC) ARUNDO PLINIANA ROOT 10 [hp_X] in 29.6 mL CORTISONE ACETATE (UNII: 883WKN7W8X) (CORTISONE - UNII:V27W9254FZ) CORTISONE ACETATE 10 [hp_X] in 29.6 mL EUPHRASIA STRICTA (UNII: C9642I91WL) (EUPHRASIA STRICTA - UNII:C9642I91WL) EUPHRASIA STRICTA 10 [hp_X] in 29.6 mL GALPHIMIA GLAUCA FLOWERING TOP (UNII: 93PH5Q8M7E) (GALPHIMIA GLAUCA FLOWERING TOP - UNII:93PH5Q8M7E) GALPHIMIA GLAUCA FLOWERING TOP 10 [hp_X] in 29.6 mL HISTAMINE DIHYDROCHLORIDE (UNII: 3POA0Q644U) (HISTAMINE - UNII:820484N8I3) HISTAMINE DIHYDROCHLORIDE 10 [hp_X] in 29.6 mL POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152) POTASSIUM CHLORIDE 10 [hp_X] in 29.6 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM CHLORIDE 10 [hp_X] in 29.6 mL SACCHAROMYCES CEREVISIAE RNA (UNII: J17GBZ5VGX) (SACCHAROMYCES CEREVISIAE RNA - UNII:J17GBZ5VGX) SACCHAROMYCES CEREVISIAE RNA 10 [hp_X] in 29.6 mL SCHOENOCAULON OFFICINALE SEED (UNII: 6NAF1689IO) (SCHOENOCAULON OFFICINALE SEED - UNII:6NAF1689IO) SCHOENOCAULON OFFICINALE SEED 10 [hp_X] in 29.6 mL WYETHIA HELENIOIDES ROOT (UNII: J10PD1AQ0N) (WYETHIA HELENIOIDES ROOT - UNII:J10PD1AQ0N) WYETHIA HELENIOIDES ROOT 10 [hp_X] in 29.6 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57955-4043-1 1 in 1 CARTON 1 29.6 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 05/11/2012 Labeler - King Bio Inc. (617901350) Registrant - King Bio Inc. (617901350) Establishment Name Address ID/FEI Business Operations King Bio Inc. 617901350 manufacture