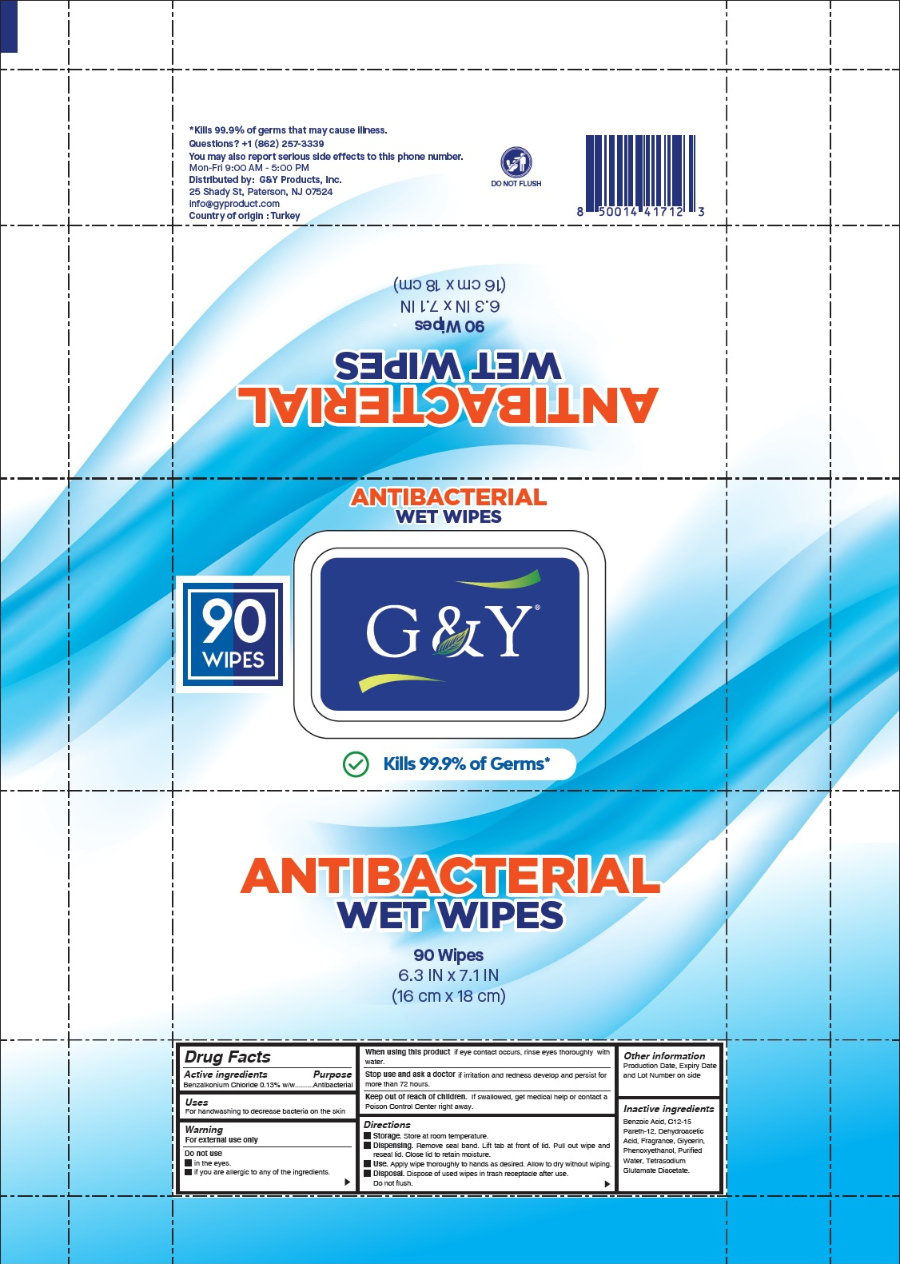
Label: G AND Y ANTIBACTERIAL WET WIPES cloth
-
Contains inactivated NDC Code(s)
NDC Code(s): 80903-013-90 - Packager: G&Y PRODUCTS INC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 1, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
- Warnings
- Do not use
- When using this product
- Keep out of reach of children.
- STOP USE
- Cloth
- Directions
- Inactive Ingredients
-
Package label
Kills 99.9% of Germs that may cause illness.
6.3 IN x 7.1 IN
(16 cm x 18 cm)
Questions? +1 (862) 257-3339
You may also report serious side effects to this phone number.
Mon-Fri 9:00 AM - 5:00 PM
Distributed by: G&Y Products, Inc.
25 Shady St, Paterson, NJ 07524
info@gyproduct.com
Country of origin: Turkey
- Packaging
-
INGREDIENTS AND APPEARANCE
G AND Y ANTIBACTERIAL WET WIPES
g and y antibacterial wet wipes clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80903-013 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.13 g in 3.5 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) TETRASODIUM GLUTAMATE DIACETATE (UNII: 5EHL50I4MY) DEHYDROACETIC ACID (UNII: 2KAG279R6R) PHENOXYETHANOL (UNII: HIE492ZZ3T) C12-15 PARETH-12 (UNII: 131316X18L) BENZOIC ACID (UNII: 8SKN0B0MIM) GLYCERIN (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80903-013-90 90 in 1 PACKAGE 07/03/2020 1 3.5 mL in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 07/03/2020 Labeler - G&Y PRODUCTS INC. (081236664) Registrant - G AND Y PRODUCTS INC. (081236664) Establishment Name Address ID/FEI Business Operations ERUSLU SAGLIK URUNLERI SANAYI VE TICARET ANONIM SIRKETI 565415460 manufacture(80903-013)