Label: BLISS TRIPLE OXYGEN ULTIMATE PROTECTION UV MOISTURIZER SPF 33- avobenzone, ensulizole, octocrylene, oxybenzone lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 54111-131-50 - Packager: Bentley Laboratories, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 19, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
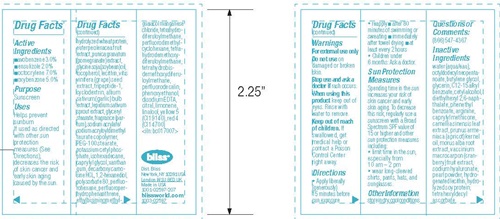
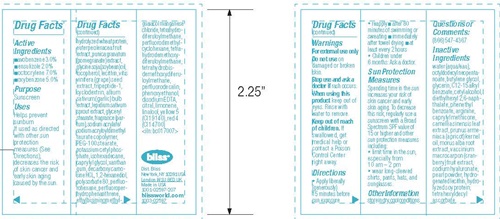
- Drug FactsActive ingredients
- Purpose
- Uses
- Warnings
- Keep out of reach of children.
- Directions
-
Sun Protection Measures
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 am - 2 pm
- wear long sleeved shirts, pants, hats, and sunglasses.
- Other information
-
Inactive Ingredients
water(aqua/eau), octyldodecyl neopentanoate, Butylene glycol, glycerin. C12-15 alkyl benzoate, cetyl alcohol, diethylhexyl 2,6-naphthalate, phenethyl benzoate, arginine, caprylyl methicone, camellia sinensis leaf extract, prunus armeniaca (apricot) kernel oil, morus alba root extract, vaccinium macrocarpon (cranberry) fruit extract, sodium hyaluronate, pearl powder, hydrogenated lecithin, hydrolyzed soy protein, tetrahexyldecyl ascorbate, hydrolyzed wheat protein, euterpe oleracea fruit extract, punica granatum (pomegranate) extract, glycine soja (soybean) oil, tocoherol, lecithin, vitis vinifera (grape) seed extract, tripeptide-1, cyclodextrin, allium sativum (garlic) bulb extract, lepidium sativum sprout extract, glycerly stearate, fragrance (parfum), sodium acrylate/sodium acryloyldimethyl taurate copolymer, PEG-100 stearate, potassium cetyl phosphate, isohexadecane, caprylyl glycol, xanthan gum, decarboxy carnosine HCL, 1,2-hexanediol, polysorbate 80, perfluorohexane, perfluoroperhydrophenanthrene, ethylbisiminomethylguaiacol manganese chloride, tetrahydrodemethoxydiferuloylmethane, tetrahydrobisdemethoxydiferuloylmethane, perfluorodecalin, phenoxyethanol, disodium EDTA, citral, limonene, linallo, yellow 5 (Cl 19140), red 4 (Cl 14700) <iln: bc017007>
-
Bliss Triple Oxygen Ultimate Protection UV Moisturizer SPF 33 product label
bliss®
from our spa to your skin™
triple oxygen®ultimate protection
uv moisturizerSPF 33
broad spectrum sunscreen
UVA/UVB/PA+++
50 mL 1.7 fl oz
"spa"-tacular results
We've poured the expertise behind our spa's triple oxygen facial™ treatment into this bottle, so you know you'll get spa-worthy results at home.
To book an appointment at Bliss Spa click: blissworld.com or call: 877 TO BLISS
For QuickBliss and Bliss Spa booking information in the UK click: bliss world.co.uk or call: 0808 100 4151
bliss®
Dist. Bliss
New York, NY 10281 USA
London W1U 8ED UK
Made in the USA • 1001-02597-208
blissworld.com
1003-02597

-
INGREDIENTS AND APPEARANCE
BLISS TRIPLE OXYGEN ULTIMATE PROTECTION UV MOISTURIZER SPF 33
avobenzone, ensulizole, octocrylene, oxybenzone lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:54111-131 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 1.5 g in 50 mL ENSULIZOLE (UNII: 9YQ9DI1W42) (ENSULIZOLE - UNII:9YQ9DI1W42) ENSULIZOLE 1 g in 50 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 3.5 g in 50 mL OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 2.5 g in 50 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) OCTYLDODECYL NEOPENTANOATE (UNII: X8725R883T) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) GLYCERIN (UNII: PDC6A3C0OX) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) CETYL ALCOHOL (UNII: 936JST6JCN) DIETHYLHEXYL 2,6-NAPHTHALATE (UNII: I0DQJ7YGXM) PHENETHYL BENZOATE (UNII: 0C143929GK) ARGININE (UNII: 94ZLA3W45F) CAPRYLYL TRISILOXANE (UNII: Q95M2P1KJL) GREEN TEA LEAF (UNII: W2ZU1RY8B0) APRICOT KERNEL OIL (UNII: 54JB35T06A) MORUS ALBA ROOT (UNII: CST1G9BZGD) CRANBERRY (UNII: 0MVO31Q3QS) HYALURONATE SODIUM (UNII: YSE9PPT4TH) PEARL (HYRIOPSIS CUMINGII) (UNII: A75L5FZ40U) HYDROGENATED SOYBEAN LECITHIN (UNII: H1109Z9J4N) SOY PROTEIN (UNII: R44IWB3RN5) TETRAHEXYLDECYL ASCORBATE (UNII: 9LBV3F07AZ) HYDROLYZED WHEAT PROTEIN (ENZYMATIC, 3000 MW) (UNII: J2S07SB0YL) ACAI (UNII: 46AM2VJ0AW) PUNICA GRANATUM ROOT BARK (UNII: CLV24I3T1D) SOYBEAN OIL (UNII: 241ATL177A) TOCOPHEROL (UNII: R0ZB2556P8) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) VITIS VINIFERA SEED (UNII: C34U15ICXA) PREZATIDE (UNII: 39TG2H631E) BETADEX (UNII: JV039JZZ3A) GARLIC (UNII: V1V998DC17) GARDEN CRESS SPROUT (UNII: PWQ18YNR62) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) SODIUM ACRYLATE/SODIUM ACRYLOYLDIMETHYLTAURATE COPOLYMER (4000000 MW) (UNII: 1DXE3F3OZX) PEG-100 STEARATE (UNII: YD01N1999R) POTASSIUM CETYL PHOSPHATE (UNII: 03KCY6P7UT) ISOHEXADECANE (UNII: 918X1OUF1E) CAPRYLYL GLYCOL (UNII: 00YIU5438U) XANTHAN GUM (UNII: TTV12P4NEE) DECARBOXY CARNOSINE HYDROCHLORIDE (UNII: 6X7K9I5QR7) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) POLYSORBATE 80 (UNII: 6OZP39ZG8H) PERFLEXANE (UNII: FX3WJ41CMX) PERFLUOROPERHYDROPHENANTHRENE (UNII: ZZ3T53GWV9) ETHYLBISIMINOMETHYLGUAIACOL MANGANESE CHLORIDE (UNII: SM5YJ88LTU) TETRAHYDRODIFERULOYLMETHANE (UNII: 00U0645U03) PERFLUORODIMETHYLCYCLOHEXANE (UNII: Q1Y54IOL0P) TETRAHYDRODEMETHOXYDIFERULOYLMETHANE (UNII: 44D8X9U00T) PERFLUNAFENE (UNII: 54A06VV62N) PHENOXYETHANOL (UNII: HIE492ZZ3T) EDETATE SODIUM (UNII: MP1J8420LU) CITRAL (UNII: T7EU0O9VPP) LIMONENE, (+)- (UNII: GFD7C86Q1W) LINALOOL, (+)- (UNII: F4VNO44C09) BASIC YELLOW 5 (UNII: 07BP340B4T) SOLVENT RED 4 (UNII: EVE9WNU99R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54111-131-50 50 mL in 1 TUBE; Type 0: Not a Combination Product 12/01/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 12/01/2016 Labeler - Bentley Laboratories, LLC (068351753)