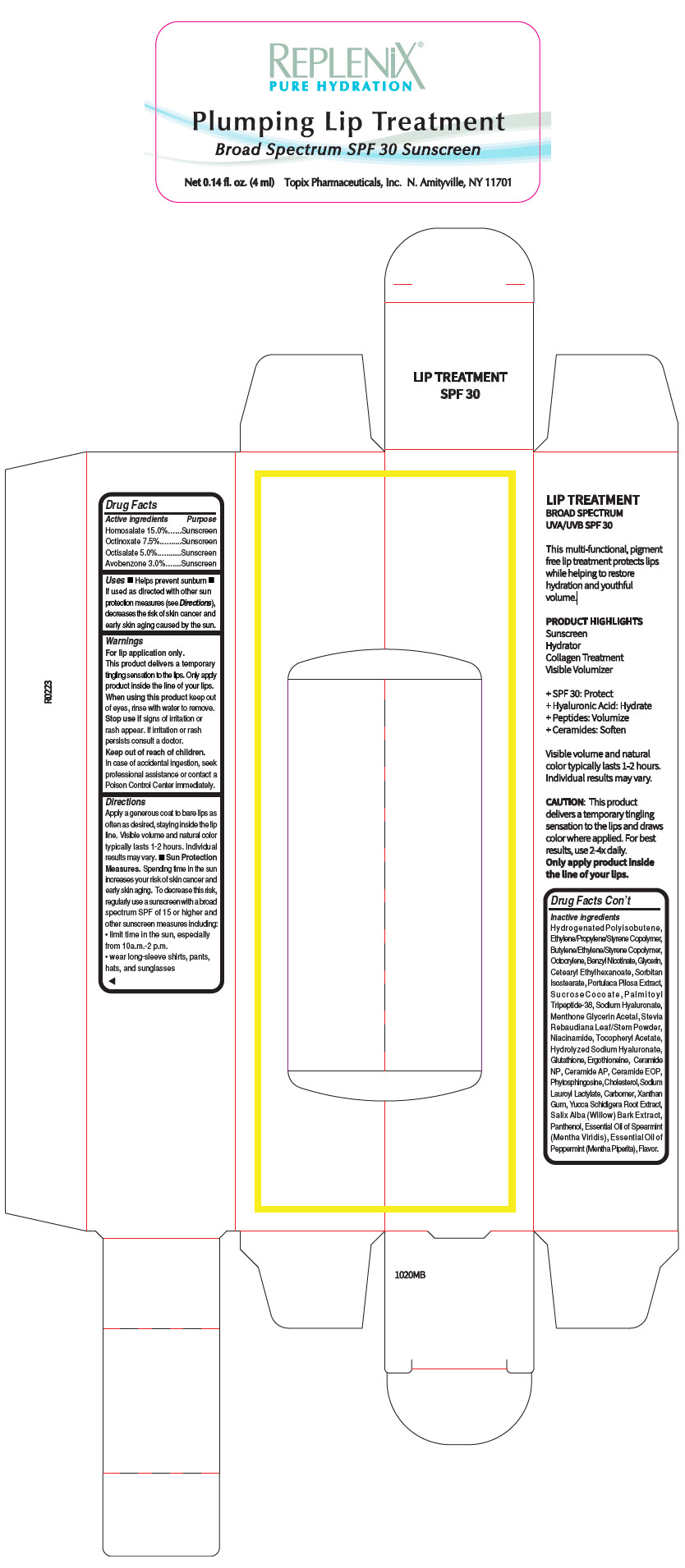
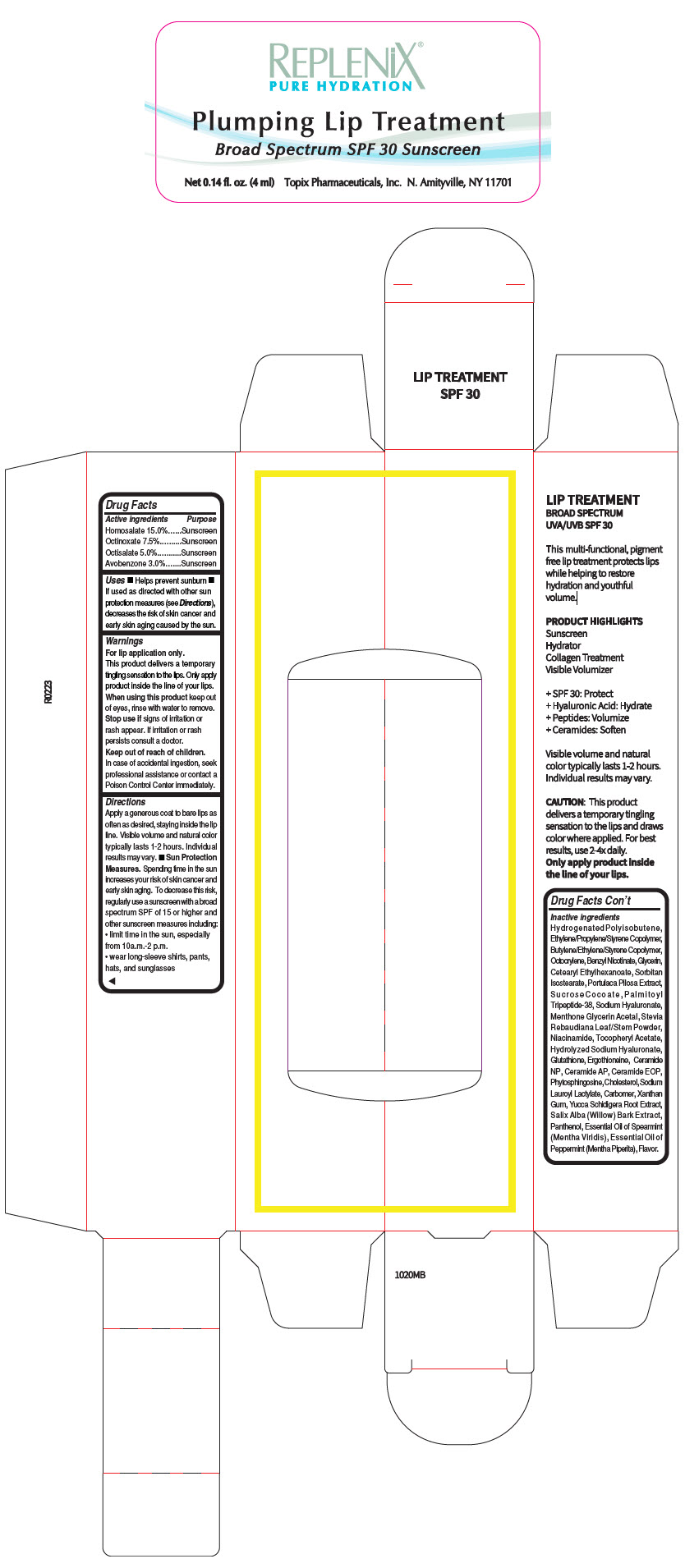
Label: REPLENIX LIP THERAPY HOMOSALATE- homosalate lipstick
- NDC Code(s): 51326-102-04
- Packager: Topiderm, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 2, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses
- Helps prevent sunburn
- If used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun.
- Warnings
-
Directions
Apply a generous coat to bare lips as often as desired, staying inside the lip line. Visible volume and natural color typically lasts 1-2 hours. Individual results may vary.
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sunscreen measures including:
- limit time in the sun, especially from 10a.m.-2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sunscreen measures including:
-
Inactive ingredients
Hydrogenated Polyisobutene, Ethylene/Propylene/Styrene Copolymer, Butylene/Ethylene/Styrene Copolymer, Octocrylene, Benzyl Nicotinate, Glycerin, Cetearyl Ethylhexanoate, Sorbitan Isostearate, Portulaca Pilosa Extract, Sucrose Cocoate, Pa l m i t o y l Tripeptide-38, Sodium Hyaluronate, Menthone Glycerin Acetal, Stevia Rebaudiana Leaf/Stem Powder, Niacinamide, Tocopheryl Acetate, Hydrolyzed Sodium Hyaluronate, Glutathione, Ergothioneine, Ceramide NP, Ceramide AP, Ceramide EOP, Phytosphingosine, Cholesterol, Sodium Lauroyl Lactylate, Carbomer, Xanthan Gum, Yucca Schidigera Root Extract, Salix Alba (Willow) Bark Extract, Panthenol, Essential Oil of Spearmint (Mentha Viridis), Essential Oil of Peppermint (Mentha Piperita), Flavor.
- PRINCIPAL DISPLAY PANEL - 4 ml Bottle Carton
-
INGREDIENTS AND APPEARANCE
REPLENIX LIP THERAPY HOMOSALATE
homosalate lipstickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51326-102 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 150 mg in 1 g OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 g OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 g AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 g Inactive Ingredients Ingredient Name Strength HYDROGENATED POLYBUTENE (1300 MW) (UNII: 7D1YQ9Y5EZ) OCTOCRYLENE (UNII: 5A68WGF6WM) BENZYL NICOTINATE (UNII: S497LCF9C9) GLYCERIN (UNII: PDC6A3C0OX) CETEARYL ETHYLHEXANOATE (UNII: 9M64UO4C25) SORBITAN ISOSTEARATE (UNII: 01S2G2C1E4) PORTULACA PILOSA WHOLE (UNII: II29C6E5R3) SUCROSE COCOATE (UNII: 3H18P0UK73) PALMITOYL LYSYLDIOXYMETHIONYLLYSINE (UNII: T7A529FB8O) HYALURONATE SODIUM (UNII: YSE9PPT4TH) MENTHONE 1,2-GLYCEROL KETAL, (+/-)- (UNII: 7QQ1EE6RCP) STEVIA REBAUDIUNA LEAF (UNII: 6TC6NN0876) NIACINAMIDE (UNII: 25X51I8RD4) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) GLUTATHIONE (UNII: GAN16C9B8O) ERGOTHIONEINE (UNII: BDZ3DQM98W) CERAMIDE NP (UNII: 4370DF050B) CERAMIDE AP (UNII: F1X8L2B00J) CERAMIDE 1 (UNII: 5THT33P7X7) PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) CHOLESTEROL (UNII: 97C5T2UQ7J) SODIUM LAUROYL LACTYLATE (UNII: 7243K85WFO) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) XANTHAN GUM (UNII: TTV12P4NEE) YUCCA SCHIDIGERA ROOT (UNII: E2H9ET15AT) SALIX ALBA BARK (UNII: 205MXS71H7) PANTHENOL (UNII: WV9CM0O67Z) SPEARMINT OIL (UNII: C3M81465G5) PEPPERMINT OIL (UNII: AV092KU4JH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51326-102-04 4 g in 1 BOTTLE; Type 0: Not a Combination Product 01/02/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/02/2022 Labeler - Topiderm, Inc. (049121643) Establishment Name Address ID/FEI Business Operations Topiderm, Inc. 049121643 MANUFACTURE(51326-102)