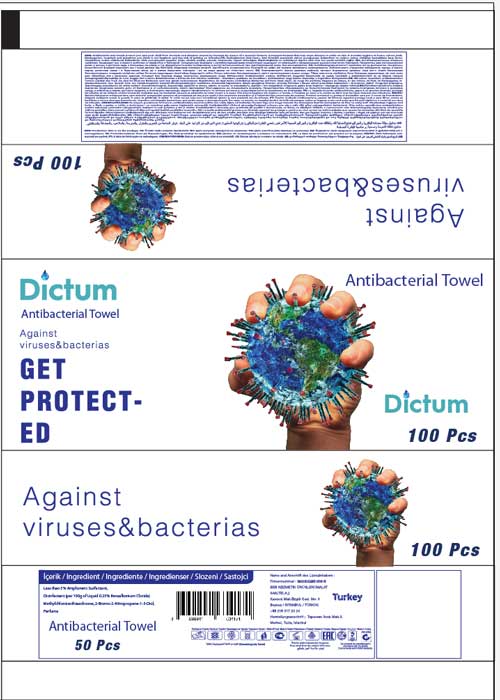
Label: DICTUM ANTIBACTERIAL TOWEL- benzalkonium chloride swab
-
Contains inactivated NDC Code(s)
NDC Code(s): 79780-105-01, 79780-105-02, 79780-105-03, 79780-105-04 - Packager: BEB Kozmetik Urunleri Imalat Sanayi Ticaret AS
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 23, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Purpose
-
INDICATIONS & USAGE
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes
thoroughly with warter.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious contiditon.
Keep our of reac of children. If swallowed, get medical help or contact a Poison Control Center right
away.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right
away.
Place enough product on hands to cover all surfaces. Rub hand together until dry.
Supervise children under 6 years of age when using this product to avoid swallowing.
Store between 15-43C (59-110F)
Avoid freezing and excessive heat above 43C (110F)When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes
thoroughly with warter.Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right
away.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right
away. - Active Ingredients
- Inactive Ingredients
- Warnings
- Dosage
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DICTUM ANTIBACTERIAL TOWEL
benzalkonium chloride swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:79780-105 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.2 g in 100 g DIDECYLDIMONIUM CHLORIDE (UNII: JXN40O9Y9B) (DIDECYLDIMONIUM - UNII:Z7F472XQPA) DIDECYLDIMONIUM CHLORIDE 0.05 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) FRAGRANCE CLEAN ORC0600327 (UNII: 329LCV5BTF) 0.25 g in 100 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:79780-105-01 25 g in 1 PACKET; Type 0: Not a Combination Product 03/30/2020 2 NDC:79780-105-02 50 g in 1 PACKET; Type 0: Not a Combination Product 03/30/2020 3 NDC:79780-105-03 100 g in 1 PACKET; Type 0: Not a Combination Product 03/30/2020 4 NDC:79780-105-04 150 g in 1 PACKET; Type 0: Not a Combination Product 03/30/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 03/30/2020 Labeler - BEB Kozmetik Urunleri Imalat Sanayi Ticaret AS (502991071) Establishment Name Address ID/FEI Business Operations BEB Kozmetik Urunleri Imalat Sanayi Ticaret AS 502991071 manufacture(79780-105)