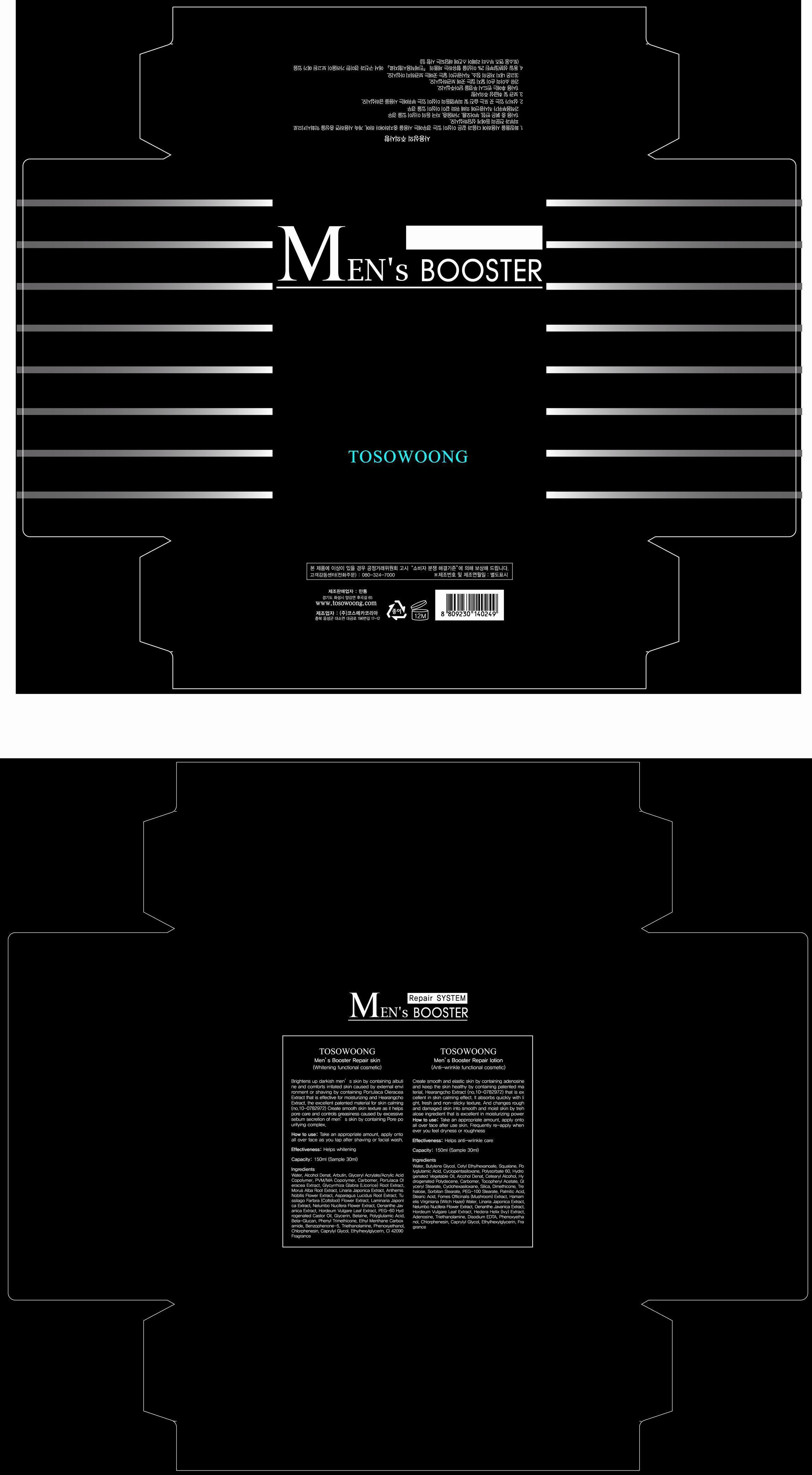
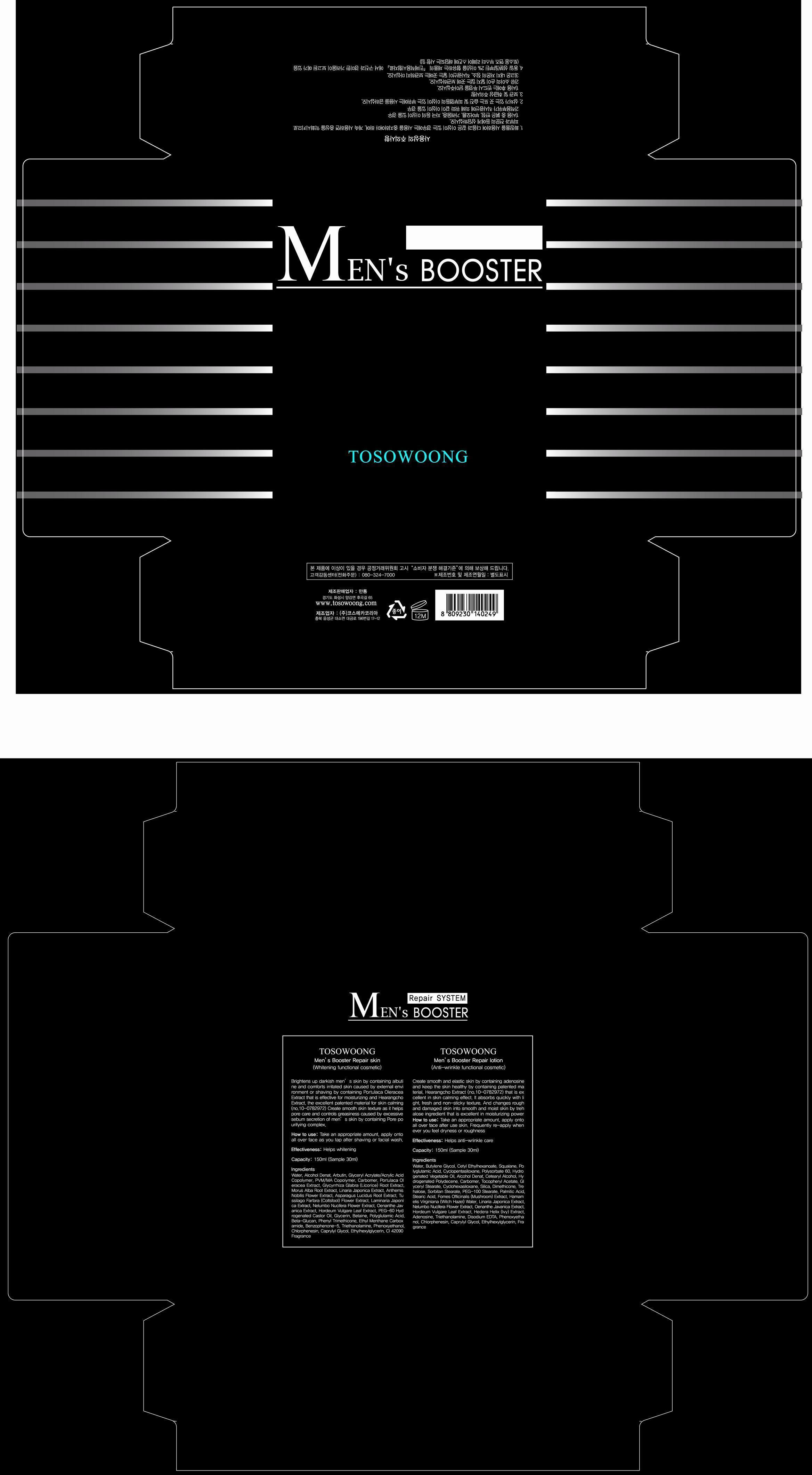
Label: TOSOWOONG MENS BOOSTER REPAIR SKIN- arbutin liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 55249-1001-1 - Packager: Mantong
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated July 5, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
water, alcohol, glyceryl acrylate/acrylic acid copolymer, PVM/MA copolymer, carbomer, portulaca oleracea extract, licorice root extract, morus alba root extract, linaria japonica extract, anthemis nobilis flower extract, asparagus lucidus root extract, coltsfood flower extract, laminaria japonica extract, nelumbo nucifera flower extract, oenanthe javanica extract, hordeum vulgare leaf extract, PEG=60 hydrogenated castor oil, glycerin, betaine, polyglutamic acid, beta-glucan, phenyl trimethicone, ethyl menthane carboxamide, benzophenone-5, triethanolamine, phenoxyethanol, chlorphenesin, caprylyl glycol, ethylhexylglycerin, CI 42090, fragrance
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TOSOWOONG MENS BOOSTER REPAIR SKIN
arbutin liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55249-1001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ARBUTIN (UNII: C5INA23HXF) (ARBUTIN - UNII:C5INA23HXF) ARBUTIN 1 mg in 150 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) PORTULACA OLERACEA WHOLE (UNII: D5J3623SV2) GLYCYRRHIZA GLABRA (UNII: 2788Z9758H) MORUS ALBA ROOT BARK (UNII: SV6B76DK9N) CHAMAEMELUM NOBILE FLOWER (UNII: O2T154T6OG) ASPARAGUS COCHINCHINENSIS TUBER (UNII: IZW9WN28HW) PETASITES FRAGRANS (UNII: 7MKJ1A3A9T) NELUMBO NUCIFERA FLOWER (UNII: 61W322NLDV) BARLEY (UNII: 5PWM7YLI7R) POLYOXYL 60 HYDROGENATED CASTOR OIL (UNII: 02NG325BQG) GLYCERIN (UNII: PDC6A3C0OX) BETAINE (UNII: 3SCV180C9W) PHENYL TRIMETHICONE (UNII: DR0K5NOJ4R) ETHYL MENTHANE CARBOXAMIDE (UNII: 6S7S02945H) BENZOPHENONE-5 (UNII: 853Z42ZYAS) PHENOXYETHANOL (UNII: HIE492ZZ3T) CHLORPHENESIN (UNII: I670DAL4SZ) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55249-1001-1 150 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 07/05/2013 Labeler - Mantong (688226855) Registrant - Mantong (688226855) Establishment Name Address ID/FEI Business Operations Cosmecca Korea Co.,Ltd. 688830827 manufacture(55249-1001)