Label: PLUS RINSE EMERGENCY EYEWASH- purified water liquid
- NDC Code(s): 72598-022-01, 72598-022-02, 72598-022-20
- Packager: INNOVATIVE SAFETY SUPPLY INC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 26, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
-
Directions
- do not dilute solution or reuse bottle
- use only unopened bottle
- to open, twist cap in the direction of the arrow
- avoid contamination of the integrated eyecup
- place integrated eyecup over affected eye(s)
- tilt head backward
- open eyelid(s) wide
- control rate of flow by pressure on bottle
- thoroughly bathe eye(s) with solution
- allow solution to flow away from eye(s)
- rinse until the bottle is empty and continue rinsing with Plum Emergency Eyewash until you reach a doctor
- continue rinsing with water if necessary
- obtain medical treatment.
- Other information
- Inactive ingredients
- Questions or Comments?
- Imported and distributed by
-
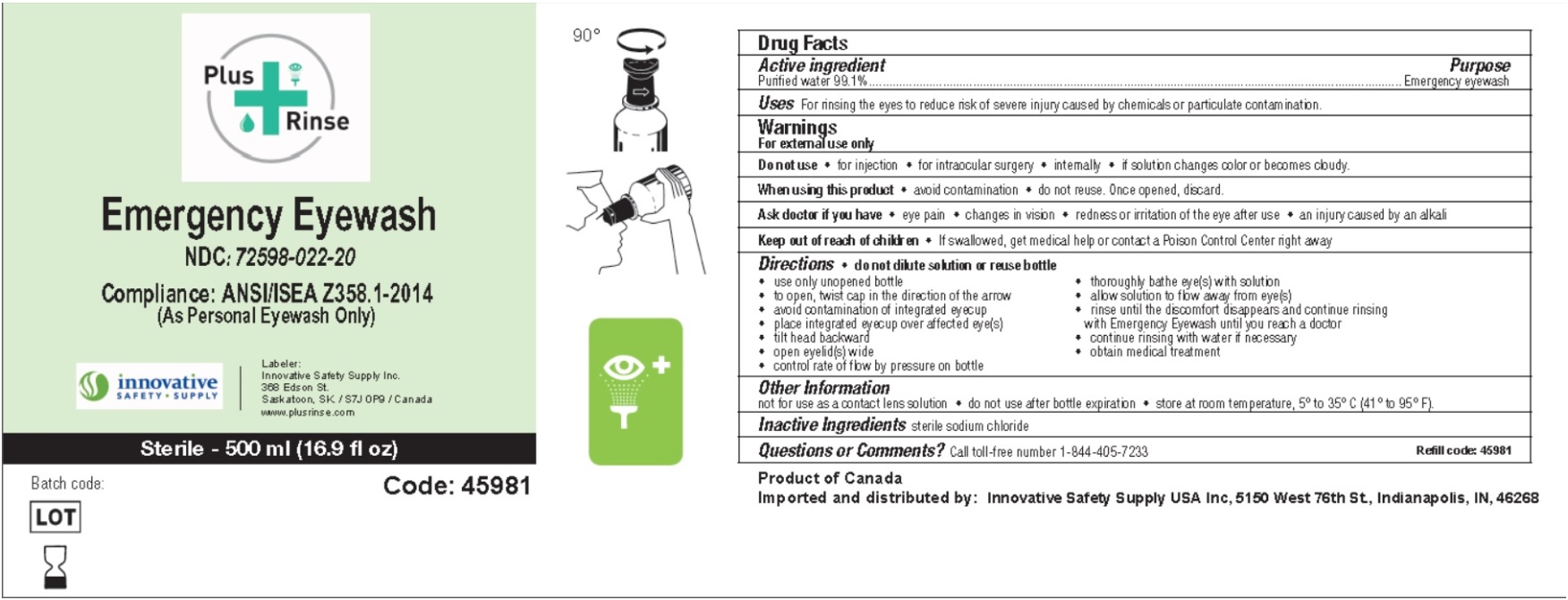
Principal Display Panel
NDC : 72598-022-20
Plus Rinse
Emergency Eyewash
Compliance: ANSI/ISEA Z358.1-2014
(As Personal Eyewash Only)
Innovative SAFETY SUPPLY
Labeler: Innovative Safety Supply Inc, 368 Edson St, Saskatoon, SK/S7J 0P9/Canada.
www.plusrinse.com
Sterile - 500 ml (16.9 fl oz)
NDC: 72598-022-01
Plus Rinse
Emergency Eyewash
Compliance: ANSI/ISEA Z358.1-2014
(As Personal Eyewash Only)
Innovative SAFETY SUPPLY
Labeler: Innovative Safety Supply Inc, 368 Edson St, Saskatoon, SK/S7J 0P9/Canada.
www.plusrinse.com
Sterile - 1000 ml (33.8 fl oz)

NDC: 72598-022-02
Plus Rinse
Emergency Eyewash
Compliance: ANSI/ISEA Z358.1-2014
(As Personal Eyewash Only)
Innovative SAFETY SUPPLY
Labeler: Innovative Safety Supply Inc, 368 Edson St, Saskatoon, SK/S7J 0P9/Canada.
www.plusrinse.com
Sterile - 200 ml (6.8 fl oz)

-
INGREDIENTS AND APPEARANCE
PLUS RINSE EMERGENCY EYEWASH
purified water liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72598-022 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 99.1 mL in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72598-022-20 500 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/06/2019 2 NDC:72598-022-02 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/06/2019 3 NDC:72598-022-01 1000 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/06/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M018 05/06/2019 Labeler - INNOVATIVE SAFETY SUPPLY INC. (203168349)



