Label: NITROSTAT- nitroglycerin tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 54868-0691-0, 54868-0691-1, 54868-1538-1 - Packager: Physicians Total Care, Inc.
- This is a repackaged label.
- Source NDC Code(s): 0071-0417, 0071-0418
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated January 12, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Description
Nitrostat is a stabilized sublingual compressed nitroglycerin tablet that contains 0.3 mg (1/200 grain), 0.4 mg (1/150 grain), or 0.6 mg (1/100 grain) nitroglycerin; as well as lactose monohydrate, NF; glyceryl monostearate, NF; pregelatinized starch, NF; calcium stearate, NF powder; and silicon dioxide, colloidal, NF. Nitroglycerin, an organic nitrate, is a vasodilating agent. The chemical name for nitroglycerin is 1, 2, 3 propanetriol trinitrate and the chemical structure is:
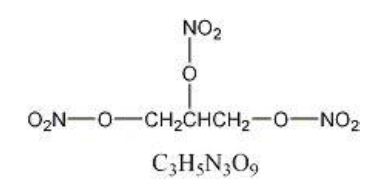
Molecular weight: 227.09
-
Clinical Pharmacology
The principal pharmacological action of nitroglycerin is relaxation of vascular smooth muscle. Although venous effects predominate, nitroglycerin produces, in a dose-related manner, dilation of both arterial and venous beds. Dilation of postcapillary vessels, including large veins, promotes peripheral pooling of blood, decreases venous return to the heart, and reduces left ventricular end-diastolic pressure (preload). Nitroglycerin also produces arteriolar relaxation, thereby reducing peripheral vascular resistance and arterial pressure (afterload), and dilates large epicardial coronary arteries; however, the extent to which this latter effect contributes to the relief of exertional angina is unclear. Therapeutic doses of nitroglycerin may reduce systolic, diastolic, and mean arterial blood pressure. Effective coronary perfusion pressure is usually maintained, but can be compromised if blood pressure falls excessively or increased heart rate decreases diastolic filling time. Elevated central venous and pulmonary capillary wedge pressures, and pulmonary and systemic vascular resistance are also reduced by nitroglycerin therapy. Heart rate is usually slightly increased, presumably due to a compensatory response to the fall in blood pressure. Cardiac index may be increased, decreased, or unchanged. Myocardial oxygen consumption or demand (as measured by the pressure-rate product, tension-time index, and stroke-work index) is decreased and a more favorable supply-demand ratio can be achieved. Patients with elevated left ventricular filling pressures and increased systemic vascular resistance in association with a depressed cardiac index are likely to experience an improvement in cardiac index. In contrast, when filling pressures and cardiac index are normal, cardiac index may be slightly reduced following nitroglycerin administration.
Mechanism of Action
Nitroglycerin forms free radical nitric oxide (NO) which activates guanylate cyclase, resulting in an increase of guanosine 3'5' monophosphate (cyclic GMP) in smooth muscle and other tissues. These events lead to dephosphorylation of myosin light chains, which regulate the contractile state in smooth muscle, and result in vasodilatation.Pharmacodynamics
Consistent with the symptomatic relief of angina, digital plethysmography indicates that onset of the vasodilatory effect occurs approximately 1 to 3 minutes after sublingual nitroglycerin administration and reaches a maximum by 5 minutes postdose. Effects persist for at least 25 minutes following Nitrostat administration.Pharmacokinetics and Drug Metabolism Absorption
Nitroglycerin is rapidly absorbed following sublingual administration of Nitrostat tablets. Mean peak nitroglycerin plasma concentrations occur at a mean time of approximately 6 to 7 minutes postdose (Table 1). Maximum plasma nitroglycerin concentrations (Cmax) and area under the plasma concentration-time curves (AUC) increase dose-proportionally following 0.3 to 0.6 mg Nitrostat. The absolute bioavailability of nitroglycerin from Nitrostat tablets is approximately 40% but tends to be variable due to factors influencing drug absorption such as sublingual hydration and mucosal metabolism.DistributionTable 1
Mean Nitroglycerin (SD) Values
Parameter
2 x 0.3 mg
Nitrostat Tablets
1 x 0.6 mg
Nitrostat Tablets
Cmax, ng/mL
2.3 (1.7)
2.1 (1.5)
tmax, min
6.4 (2.5)
7.2 (3.2)
AUC (0–¥) min
14.9 (8,2)
14.9 (11.4)
t1/2, min
2.8 (1.1)
2.6 (0.6)
The volume of distribution (VArea) of nitroglycerin following intravenous administration is 3.3 L/kg. At plasma concentrations between 50 and 500 ng/mL, the binding of nitroglycerin to plasma proteins is approximately 60%, while that of 1,2- and 1,3-dinitroglycerin is 60% and 30%, respectively.
Metabolism
A liver reductase enzyme is of primary importance in the metabolism of nitroglycerin to glycerol di- and mononitrate metabolites and ultimately to glycerol and organic nitrate. Known sites of extrahepatic metabolism include red blood cells and vascular walls. In addition to nitroglycerin, 2 major metabolites 1,2- and 1,3-dinitroglycerin, are found in plasma. Mean peak 1,2- and1,3-dinitroglycerin plasma concentrations occur at approximately 15 minutes postdose. The elimination half-life of 1,2- and 1,3-dinitroglycerin is 36 and 32 minutes, respectively. The 1,2- and 1,3 dinitroglycerin metabolites have been reported to possess approximately 2% and 10% of the pharmacological activity of nitroglycerin. Higher plasma concentrations of the dinitro metabolites, along with their nearly 10-fold longer elimination half-lives, may contribute significantly to the duration of pharmacologic effect. Glycerol mononitrate metabolites of nitroglycerin are biologically inactive.
Elimination
Nitroglycerin plasma concentrations decrease rapidly with a mean elimination half-life of 2 to 3 minutes. Half-life values range from 1.5 to 7.5 minutes. Clearance (13.6 L/min) greatly exceeds hepatic blood flow. Metabolism is the primary route of drug elimination.
- Indications and Usage
-
Contraindications
Allergic reactions to organic nitrates are extremely rare, but they do occur. Nitroglycerin is contraindicated in patients who are allergic to it. Sublingual nitroglycerin therapy is contraindicated in patients with early myocardial infarction, severe anemia, increased intracranial pressure, and those with a known hypersensitivity to nitroglycerin. Administration of Nitrostat (nitroglycerin tablets, USP) is contraindicated in patients who are using Viagra® since Viagra has been shown to potentiate the hypotensive effects of organic nitrates.
-
Warnings
The benefits of sublingual nitroglycerin in patients with acute myocardial infarction or congestive heart failure have not been established. If one elects to use nitroglycerin in these conditions, careful clinical or hemodynamic monitoring must be used because of the possibility of hypotension and tachycardia.
-
Precautions
General
Only the smallest dose required for effective relief of the acute anginal attack should be used. Excessive use may lead to the development of tolerance. Nitrostat tablets are intended for sublingual or buccal administration and should not be swallowed. Severe hypotension, particularly with upright posture, may occur with small doses of nitroglycerin. This drug should therefore be used with caution in patients who may be volume-depleted or who, for whatever reason, are already hypotensive. Hypotension induced by nitroglycerin may be accompanied by paradoxical bradycardia and increased angina pectoris. Nitrate therapy may aggravate the angina caused by hypertrophic cardiomyopathy. As tolerance to other forms of nitroglycerin develops, the effects of sublingual nitroglycerin on exercise tolerance, although still observable, is blunted. In industrial workers who have had long-term exposure to unknown (presumably high) doses of organic nitrates, tolerance rarely occurs. Chest pain, acute myocardial infraction, and even sudden death have occurred during temporary withdrawal of nitrates from these workers, demonstrating the existence of true physical dependence. Several clinical trials of nitroglycerin patches or infusions in patients with angina pectoris have evaluated regimens which incorporated a 10 to 12 hour nitrate free interval. In some of these trials, an increase in the frequency of anginal attacks during the nitrate free interval was observed in a small number of patients. In one trial, patients had decreased exercise tolerance at the end of the nitrate interval. Hemodynamic rebound has been observed only rarely; on the other hand, few studies were so designed that rebound, if it had occurred, would have been detected. Nitrate tolerance as a result of sublingual nitroglycerin administration is probably possible, but only in patients who maintain high continuous nitrate levels for more than 10 or 12 hours daily. Such use of sublingual nitroglycerin would entail administration of scores of tablets daily and is not recommended.
The drug should be discontinued if blurring of vision or drying of the mouth occurs. Excessive dosage of nitroglycerin may produce severe headaches.
Information for Patients
If possible, patients should sit down when taking Nitrostat tablets. This eliminates the possibility of falling due to lightheadedness or dizziness. Nitroglycerin may produce a burning or tingling sensation when administered sublingually; however, the ability to produce a burning or tingling sensation should not be considered a reliable method for determining the potency of the tablets.
Headaches can sometimes accompany treatment with nitroglycerin. In patients who get these headaches, the headaches may be a marker of the activity of the drug. Treatment with nitroglycerin may be associated with lightheadedness on standing, especially just after rising from a recumbent or seated position. This effect may be more frequent in patients who have also consumed alcohol. Nitroglycerin should be kept in the original glass container, tightly capped.Drug Interactions
Patients receiving antihypertensive drugs, beta-adrenergic blockers, or phenothiazines and nitrates should be observed for possible additive hypotensive effects. Marked orthostatic hypotension has been reported when calcium channel blockers and organic nitrates were used concomitantly. Concomitant use of nitrates and alcohol may cause hypotension. The vasodilatory and hemodynamic effects of nitroglycerin may be enhanced by concomitant administration of aspirin. Intravenous administration of nitroglycerin decreases the thrombolytic effect of alteplase. Therefore, caution should be observed in patients receiving sublingual nitroglycerin during alteplase therapy. Intravenous nitroglycerin reduces the anticoagulant effect of heparin and activated partial thromboplastin times (APTT) should be monitored in patients receiving heparin and intravenous nitroglycerin. It is not known if this effect occurs following single sublingual nitroglycerin doses. Tricyclic antidepressants (amitriptyline, desipramine, doxepin, others) and anticholinergic drugs may cause dry mouth and diminished salivary secretions. This may make dissolution of sublingual nitroglycerin difficult. Increasing salivation with chewing gum or artificial saliva products may prove useful in aiding dissolution of sublingual nitroglycerin.
Oral administration of nitroglycerin markedly decreases the first-pass metabolism of dihydroergotamine and subsequently increases its oral bioavailability. Ergotamine is known to precipitate angina pectoris. Therefore, patients receiving sublingual nitroglycerin should avoid ergotamine and related drugs or be monitored for symptoms of ergotism if this is not possible. Administration of nitroglycerin is contraindicated in patients who are using Viagra (sildenafil citrate). Viagra has been shown to potentiate the hypotensive effects of organic nitrates.
A decrease in therapeutic effect of sublingual nitroglycerin may result from use of long-acting nitrates.Drug/Laboratory test Interactions
Nitrates may interfere with the Zlatkis-Zak color reaction causing a false report of decreased serum cholesterol.Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal carcinogenesis studies with sublingually administered nitroglycerin have not been performed. Rats receiving up to 434 mg/kg/day of dietary nitroglycerin for 2 years developed dose-related fibrotic and neoplastic changes in liver, including carcinomas, and interstitial cell tumors in testes. At high dose, the incidences of hepatocellular carcinomas in males was 48% and in females was 33% compared to 0% in untreated controls. Incidences of testicular tumors were 52% vs 8% in controls. Lifetime dietary administration of up to 1058 mg/kg/day of nitroglycerin was not tumorigenic in mice. Nitroglycerin was weakly mutagenic in Ames tests performed in 2 different laboratories. Nevertheless, there was no evidence of mutagenicity in an in vivo dominant lethal assay with male rats treated with doses up to about 363 mg/kg/day, PO, or in ex vivo cytogenetic tests in rat and dog tissues. In a 3-generation reproduction study, rats received dietary nitroglycerin at doses up to about 434 mg/kg/day for 6 months prior to mating of the F0 generation with treatment continuing through successive F1 and F2 generations. The high dose was associated with decreased feed intake and body weight gain in both sexes at all matings. No specific effect on the fertility of the F0 generation was seen. Infertility noted in subsequent generations, however, was attributed to increased interstitial cell tissue and aspermatogenesis in the high-dose males. In this 3-generation study there was no clear evidence of teratogenicity.Pregnancy Category C
Animal reproduction and teratogenicity studies have not been conducted with nitroglycerin sublingual tablets. Teratology studies in rats and rabbits, however, were conducted with topically applied nitroglycerin ointment at doses up to 80 mg/kg/day and 240 mg/kg/ day, respectively. No toxic effects on dams or fetuses were seen at any dose tested. There are no adequate and well-controlled studies in pregnant women. Nitroglycerin should be given to a pregnant woman only if clearly needed.Nursing Mothers
It is not known whether nitroglycerin is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when nitroglycerin is administered to a nursing woman.
Pediatric Use
The safety and effectiveness of nitroglycerin in pediatric patients have not been established.Geriatric Use
Clinical studies of Nitrostat did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. -
Adverse Reactions
Headache which may be severe and persistent may occur immediately after use. Vertigo, dizziness, weakness, palpitation, and other manifestations of postural hypotension may develop occasionally, particularly in erect, immobile patients. Marked sensitivity to the hypotensive effects of nitrates (manifested by nausea, vomiting, weakness, diaphoresis, pallor, and collapse) may occur at therapeutic doses. Syncope due to nitrate vasodilatation has been reported. Flushing, drug rash, and exfoliative dermatitis have been reported in patients receiving nitrate therapy.
-
Overdosage
HEMODYNAMIC EFFECTS: The effects of nitroglycerin overdose are generally the results of nitroglycerin's capacity to induce vasodilatation, venous pooling, reduced cardiac output, and hypotension. These hemodynamic changes may have protean manifestations, including increased intracranial pressure, with any or all of persistent throbbing headache, confusion, and moderate fever; vertigo; palpitations; tachycardia; visual disturbances; nausea and vomiting (possibly with colic and even bloody diarrhea); syncope (especially in the upright posture); dyspnea, later followed by reduced ventilatory effort, diaphoresis, with the skin either flushed or cold and clammy; heart block and bradycardia; paralysis; coma; seizures; and death. No specific antagonist to the vasodilator effects of nitroglycerin is known, and no intervention has been subject to controlled study as a therapy of nitroglycerin overdose. Because the hypotension associated with nitroglycerin overdose is the result of venodilatation and arterial hypovolemia, prudent therapy in this situation should be directed toward increase in central fluid volume. Passive elevation of
the patient's legs may be sufficient, but intravenous infusion of normal saline or similar fluid may also be necessary. The use of epinephrine or other arterial vasoconstrictors in this setting is likely to do more harm than good. In patients with renal disease or congestive heart failure, therapy resulting in central volume expansion is not without hazard. Treatment of nitroglycerin overdose in these patients may be subtle and difficult, and invasive monitoring may be required.
METHEMOGLOBINEMIA: Methemoglobinemia has been rarely reported in association with organic nitrates. The diagnosis should be suspected in patients who exhibit signs of impaired oxygen delivery despite adequate cardiac output and adequate arterial PO2. Classically, methemoglobinemic blood is described as chocolate brown, without color change on exposure to air. If methemoglobinemia is present, intravenous administration of methylene blue, 1 to 2 mg/kg of body weight, may be required. -
Dosage and Administration
One tablet should be dissolved under the tongue or in the buccal pouch at the first sign of an acute anginal attack. The dose may be repeated approximately every 5 minutes, until relief is obtained. If the pain persists after a total of 3 tablets in a 15-minute period, prompt medical attention is recommended. Nitrostat may be used prophylactically 5 to 10 minutes prior to engaging in activities which might precipitate an acute attack. During administration the patient should rest, preferably in the sitting position. No dosage adjustment is required in patients with renal failure.
-
How Supplied
Nitrostat is supplied as white, round, flat-faced tablets in 3 strengths (0.3 mg, 0.4 mg, and 0.6 mg) in
Store at Controlled Room Temperature 20°–25°C (68°–77°F) [see USP].0.3 mg (1/200
grain):
Coded "N" on one side and "3" on the other.
Bottles of 100 NDC 54868-1538-1
0.4 mg (1/150
grain):
Coded "N" on one side and "4" on the other.
Bottles of 25 NDC 54868-0691-0
Bottles of 100 NDC 54868-0691-1
Rx only

LAB-0180-2.0
December 2005
Relabeling of "Additional" label by:
Physicians Total Care, Inc.
Tulsa, OK 74146
- PACKAGE LABEL PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NITROSTAT
nitroglycerin tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-1538(NDC:0071-0417) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NITROGLYCERIN (UNII: G59M7S0WS3) (NITROGLYCERIN - UNII:G59M7S0WS3) NITROGLYCERIN 0.3 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) CALCIUM STEARATE (UNII: 776XM7047L) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color white Score no score Shape ROUND Size 4mm Flavor Imprint Code N;4 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-1538-1 100 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021134 09/22/1994 NITROSTAT
nitroglycerin tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-0691(NDC:0071-0418) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NITROGLYCERIN (UNII: G59M7S0WS3) (NITROGLYCERIN - UNII:G59M7S0WS3) NITROGLYCERIN 0.4 mg Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) CALCIUM STEARATE (UNII: 776XM7047L) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color white Score no score Shape ROUND Size 4mm Flavor Imprint Code N;4 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-0691-0 25 in 1 BOTTLE 2 NDC:54868-0691-1 100 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021134 08/11/1995 Labeler - Physicians Total Care, Inc. (194123980) Establishment Name Address ID/FEI Business Operations Physicians Total Care, Inc. 194123980 relabel




