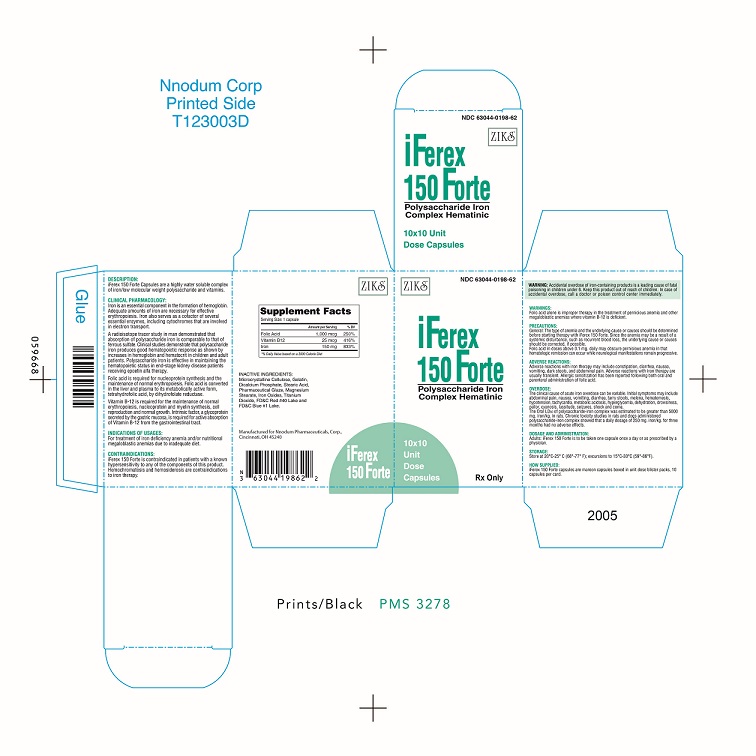
Label: IFEREX 150 FORTE- iron, cyanocobalamin and folic acid capsule
- NHRIC Code(s): 63044-198-01, 63044-198-62
- Packager: Nnodum Pharmaceuticals
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated December 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- STATEMENT OF IDENTITY
- HEALTH CLAIM
- WARNINGS:
- PRECAUTIONS:
- SAFE HANDLING WARNING:
- DOSAGE AND ADMINISTRATION:
- iFerex 150 Forte PACKAGE LABEL/PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
IFEREX 150 FORTE
iron, cyanocobalamin and folic acid capsuleProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:63044-198 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IRON (UNII: E1UOL152H7) (IRON - UNII:E1UOL152H7) IRON 150 mg CYANOCOBALAMIN (UNII: P6YC3EG204) (CYANOCOBALAMIN - UNII:P6YC3EG204) CYANOCOBALAMIN 25 mg FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 1 mg Inactive Ingredients Ingredient Name Strength CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) GELATIN (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) D&C RED NO. 28 (UNII: 767IP0Y5NH) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C RED NO. 3 (UNII: PN2ZH5LOQY) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:63044-198-01 100 in 1 BOTTLE 2 NHRIC:63044-198-62 10 in 1 BOX 2 10 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date dietary supplement 12/01/2021 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color imprint shape scoring 1 size (solid drugs) 7 mm Labeler - Nnodum Pharmaceuticals (960457273) Establishment Name Address ID/FEI Business Operations Contract Pharmacal Corporation 057795122 manufacture