Label: FOLIVANE-OB- ferrous fumarate, iron, ascorbic acid, folic acid, thiamine mononitrate, riboflavin, niacin, calcium pantothenate, pyridoxine hydrochloride, biotin, cyanocobalamin, cupric sulfate, magnesium sulfate, manganese sulfate, and zinc sulfate capsule
- NHRIC Code(s): 13811-535-30
- Packager: Trigen Laboratories, LLC
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated January 7, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SUPPLEMENT FACTS
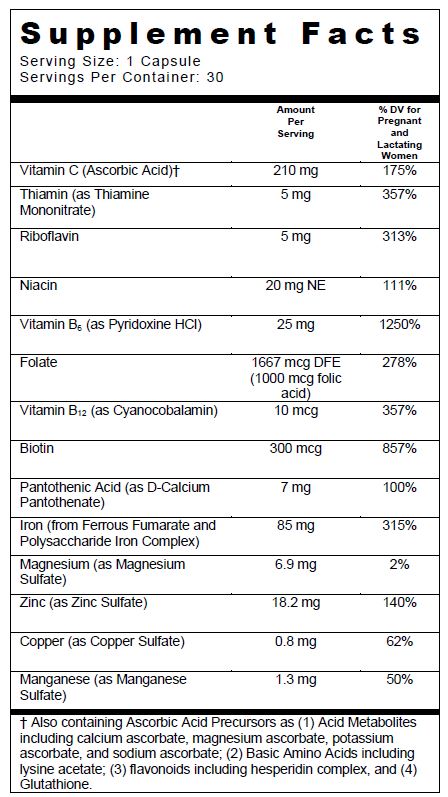
Other Ingredients: Capsule (Hypromellose, Titanium Dioxide, FD&C Blue #1, FD&C Red #40) Microcrystalline Cellulose, Magnesium Stearate, and Fumed Silica.
Folivane™-OB is a prescription prenatal supplement designed to improve the nutritional status of women throughout pregnancy and during the postnatal period to lactating and non-lactating mothers. Folivane™-OB may also be used to improve the nutritional status of women. DO NOT ADMINISTER TO CHILDREN UNDER THE AGE OF 12.
-
CONTRAINDICATIONS
Folivane™-OB is contraindicated in patients with known hypersensitivity to any of its ingredients; also, all iron compounds are contraindicated in patients with hemosiderosis, hemochromatosis, or hemolytic anemia. Pernicious anemia is a contraindication, as folic acid may obscure its signs and symptoms.
- WARNING
-
PRECAUTION
General
Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where B12 is deficient. Folic acid in doses above 0.1 mg daily may obscure pernicious anemia assessment, such that hematologic remission can occur while neurological manifestations remain progressive.
Biotin levels higher than the recommended daily allowance may cause interference with some laboratory tests, including cardiovascular diagnostic tests (e.g. troponin) and hormone test, and may lead to incorrect test results. Tell your healthcare provider about all prescription and over-the-counter medicines, vitamins, and dietary supplements that you take, including biotin.
- DRUG INTERACTIONS
-
ADVERSE REACTIONS
Folic Acid
Allergic sensitization have been reported following both oral and parenteral administration of folic acid.
Ferrous Fumarate
Gastrointestinal disturbances (anorexia, nausea, diarrhea, constipation) occur occasionally, but are usually mild and may subside with continuation of therapy. Although the absorption of iron is best when taken between meals, giving Folivane™-OB after meals may control occasional G.I. disturbances. Folivane™-OB is best absorbed when taken at bedtime.
-
OVERDOSAGE
Acute overdosage of iron may cause abdominal pain, nausea and vomiting and, in severe cases, cardiovascular collapse and death. Other more chronic symptoms include pallor and cyanosis, melena, shock, drowsiness and coma. The estimated overdose of orally ingested iron is 300 mg/kg body weight. Toxic effects are seen at 10-20 mg/kg elemental iron. When overdoses are ingested by children, severe reactions, including fatalities, have resulted. Folivane™-OB should be stored beyond the reach of children to prevent against accidental iron poisoning.
Treatment: For specific therapy, exchange transfusion and chelating agents should be used. For general management, perform gastric lavage with sodium bicarbonate solution or milk. Administer intravenous fluids and electrolytes and use oxygen.
- DESCRIPTION
- DIRECTIONS FOR USE
- HOW SUPPLIED
- STORAGE
- HEALTH CLAIM
- PRINCIPAL DISPLAY PANEL - 30 Capsule Bottle Label
-
INGREDIENTS AND APPEARANCE
FOLIVANE-OB
ferrous fumarate, iron, ascorbic acid, folic acid, thiamine mononitrate, riboflavin, niacin, calcium pantothenate, pyridoxine hydrochloride, biotin, cyanocobalamin, cupric sulfate, magnesium sulfate, manganese sulfate, and zinc sulfate capsuleProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:13811-535 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Ferrous Fumarate (UNII: R5L488RY0Q) (Ferrous Cation - UNII:GW89581OWR) Ferrous Cation 42.5 mg Iron (UNII: E1UOL152H7) (Iron - UNII:E1UOL152H7) Iron 42.5 mg Ascorbic Acid (UNII: PQ6CK8PD0R) (Ascorbic Acid - UNII:PQ6CK8PD0R) Ascorbic Acid 210 mg Folic Acid (UNII: 935E97BOY8) (Folic Acid - UNII:935E97BOY8) Folic Acid 1 mg Thiamine Mononitrate (UNII: 8K0I04919X) (Thiamine Ion - UNII:4ABT0J945J) Thiamine 5 mg Riboflavin (UNII: TLM2976OFR) (Riboflavin - UNII:TLM2976OFR) Riboflavin 5 mg Niacin (UNII: 2679MF687A) (Niacin - UNII:2679MF687A) Niacin 20 mg Calcium Pantothenate (UNII: 568ET80C3D) (Calcium Cation - UNII:2M83C4R6ZB) Calcium Cation 7 mg Pyridoxine Hydrochloride (UNII: 68Y4CF58BV) (Pyridoxine - UNII:KV2JZ1BI6Z) Pyridoxine Hydrochloride 25 mg Biotin (UNII: 6SO6U10H04) (Biotin - UNII:6SO6U10H04) Biotin 300 ug Cyanocobalamin (UNII: P6YC3EG204) (Cyanocobalamin - UNII:P6YC3EG204) Cyanocobalamin 10 ug Cupric Sulfate (UNII: LRX7AJ16DT) (Cupric Cation - UNII:8CBV67279L) Cupric Cation 800 ug MAGNESIUM SULFATE HEPTAHYDRATE (UNII: SK47B8698T) (Magnesium Cation - UNII:T6V3LHY838) MAGNESIUM SULFATE HEPTAHYDRATE 6.9 mg Manganese Sulfate (UNII: W00LYS4T26) (MANGANOUS CATION - UNII:H6EP7W5457) MANGANOUS CATION 1.3 mg Zinc Sulfate (UNII: 89DS0H96TB) (Zinc Cation - UNII:13S1S8SF37) Zinc Cation 18.2 mg Inactive Ingredients Ingredient Name Strength Hypromelloses (UNII: 3NXW29V3WO) Cellulose, Microcrystalline (UNII: OP1R32D61U) Magnesium Stearate (UNII: 70097M6I30) Titanium Dioxide (UNII: 15FIX9V2JP) FD&C Blue No. 1 (UNII: H3R47K3TBD) FD&C Red No. 40 (UNII: WZB9127XOA) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:13811-535-30 30 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 01/01/2010 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color imprint scoring 1 shape size (solid drugs) 22 mm Labeler - Trigen Laboratories, LLC (830479668)



