Label: SYMPROIC- naldemedine tablet
- NDC Code(s): 59385-041-07, 59385-041-30
- Packager: BioDelivery Sciences International Inc
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated July 25, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use SYMPROIC safely and effectively. See full prescribing information for SYMPROIC.
SYMPROIC® (naldemedine tablets for oral use)
Initial U.S. Approval: 2017RECENT MAJOR CHANGES
Warnings and Precautions, Gastrointestinal Perforation (5.1) 07/2025 INDICATIONS AND USAGE
SYMPROIC is an opioid antagonist indicated for the treatment of opioid-induced constipation (OIC) in adult patients with chronic non-cancer pain, including patients with chronic pain related to prior cancer or its treatment who do not require frequent (e.g., weekly) opioid dosage escalation (1)
DOSAGE AND ADMINISTRATION
Administration (2.1):
- Alteration of analgesic dosing regimen prior to initiating SYMPROIC is not required
- Patients receiving opioids for less than 4 weeks may be less responsive to SYMPROIC
- Discontinue SYMPROIC if treatment with the opioid pain medication is also discontinued
Dosage (2.2):
- In adults, the recommended dosage is 0.2 mg once daily with or without food
DOSAGE FORMS AND STRENGTHS
Tablets: 0.2 mg naldemedine (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Gastrointestinal perforation: Consider the overall risk benefit in patients with known or suspected lesions of the GI tract. Monitor for severe, persistent, or worsening abdominal pain; discontinue if development of symptoms (5.1)
- Opioid withdrawal: Consider the overall risk benefit in patients with disruptions to the blood-brain barrier. Monitor symptoms of opioid withdrawal (5.2)
ADVERSE REACTIONS
Most common adverse reactions (≥2%) are: abdominal pain, diarrhea, nausea and gastroenteritis (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact BioDelivery Sciences International, Inc. at 1-800-469-0261 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Strong CYP3A inducers (e.g., rifampin): Decreased naldemedine concentrations; avoid concomitant use (7)
- Other opioid antagonists: Potential for additive effect and increased risk of opioid withdrawal; avoid concomitant use (7)
- Moderate (e.g., fluconazole) and strong (e.g., itraconazole) CYP3A4 inhibitors: Increased naldemedine concentrations; monitor for adverse reactions (7)
- P-gp inhibitors (e.g., cyclosporine): Monitor for adverse reactions (7)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 7/2025
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Administration
2.2 Adult Dosage
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Gastrointestinal Perforation
5.2 Opioid Withdrawal
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Administration
- Alteration of analgesic dosing regimen prior to initiating SYMPROIC is not required.
- Patients receiving opioids for less than 4 weeks may be less responsive to SYMPROIC [see Clinical Studies (14)].
- Discontinue SYMPROIC if treatment with the opioid pain medication is also discontinued.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
SYMPROIC is contraindicated in:
- Patients with known or suspected gastrointestinal obstruction and patients at increased risk of recurrent obstruction, due to the potential for gastrointestinal perforation [see Warnings and Precautions (5.1)].
- Patients with a history of a hypersensitivity reaction to naldemedine. Reactions have included bronchospasm and rash [see Adverse Reactions (6.1)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Gastrointestinal Perforation
Cases of gastrointestinal (GI) perforation have been reported with use of another peripherally acting opioid antagonist, including SYMPROIC. Postmarketing cases of GI perforation, including fatal cases, were reported when SYMPROIC was used in patients at risk of GI perforation (e.g., GI cancer, past GI surgery, diverticulitis, chemotherapy/radiation). SYMPROIC is contraindicated in patients with known or suspected gastrointestinal obstruction or in patients at risk of recurrent obstruction. Take into account the overall risk-benefit profile when using SYMPROIC in patients with these conditions or other conditions which might result in impaired integrity of the gastrointestinal tract wall (e.g., Crohn's disease). Monitor for the development of severe, persistent, or worsening abdominal pain; discontinue SYMPROIC in patients who develop this symptom.
5.2 Opioid Withdrawal
Clusters of symptoms consistent with opioid withdrawal, including hyperhidrosis, chills, increased lacrimation, hot flush/flushing, pyrexia, sneezing, feeling cold, abdominal pain, diarrhea, nausea, and vomiting have occurred in patients treated with SYMPROIC [see Adverse Reactions (6.1)].
Patients having disruptions to the blood-brain barrier may be at increased risk for opioid withdrawal or reduced analgesia. Take into account the overall risk-benefit profile when using SYMPROIC in such patients. Monitor for symptoms of opioid withdrawal in such patients.
-
6 ADVERSE REACTIONS
Serious and important adverse reactions described elsewhere in labeling include:
- Gastrointestinal perforation [see Warnings and Precautions (5.1)]
- Opioid withdrawal [see Warnings and Precautions (5.2)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to SYMPROIC in 1163 patients in clinical trials, including 487 patients with exposures greater than six months and 203 patients with exposures of 12 months.
The following safety data are derived from three double-blind, placebo-controlled trials in patients with OIC and chronic non-cancer pain: two 12-week studies (Studies 1 and 2) and one 52-week study (Study 3) [see Clinical Studies (14)].
In Studies 1 and 2, patients on laxatives were required to discontinue their use prior to study enrollment. All patients were restricted to bisacodyl rescue treatment during the study. In Study 3, approximately 60% of patients in both treatment groups were on a laxative regimen at baseline; patients were allowed to continue using their laxative regimen throughout the study duration. The safety profile of SYMPROIC relative to placebo was similar regardless of laxative use.
Tables 1 and 2 list common adverse reactions occurring in at least 2% of patients receiving SYMPROIC and at an incidence greater than placebo. Table 1 shows pooled 12-week data from Studies 1 and 2. Table 2 shows 12-week data from Study 3.
Table 1: Common Adverse Reactions* in Patients with OIC and Chronic Non-Cancer Pain (12-week data from Studies 1 and 2) Adverse Reaction SYMPROIC
0.2 mg once daily
N=542Placebo
N=546Abdominal pain† 8% 2% Diarrhea 7% 2% Nausea 4% 2% Gastroenteritis 2% 1% Table 2: Common Adverse Reactions* in Patients with OIC and Chronic Non-Cancer Pain (12-week data from Study 3) Adverse Reaction SYMPROIC
0.2 mg once daily
N=621Placebo
N=619Abdominal pain† 11% 5% Diarrhea 7% 3% Nausea 6% 5% Vomiting 3% 2% Gastroenteritis 3% 1% Adverse reactions up to 12 months in Study 3 are similar to those listed in Tables 1 and 2 (diarrhea: 11% vs. 5%, abdominal pain: 8% vs. 3%, and nausea: 8% vs. 6% for SYMPROIC and placebo, respectively).
Opioid Withdrawal
In Studies 1, 2 and 3, adverse reactions consistent with opioid withdrawal were based on investigator assessment and adjudicated based upon the occurrence of at least 3 adverse reactions potentially related to opioid withdrawal with onset of a constellation of those symptoms occurring on the same day or within one day of each other.
Adverse reactions of possible opioid withdrawal could include non-gastrointestinal (GI) symptoms (e.g., hyperhidrosis, hot flush or flushing, chills, tremor, tachycardia, anxiety, agitation, yawning, rhinorrhea, increased lacrimation, sneezing, feeling cold, and pyrexia), GI symptoms (e.g., vomiting, diarrhea, or abdominal pain), or both GI and non-GI symptoms.
In pooled Studies 1 and 2, the incidence of adverse reactions of opioid withdrawal was 1% (8/542) for SYMPROIC and 1% (3/546) for placebo. In Study 3 (52-week data), the incidence was 3% (20/621) for SYMPROIC and 1% (9/619) for placebo. Most SYMPROIC treated subjects experienced nearly equal incidence of GI only or both GI and non-GI symptoms.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of SYMPROIC. Because reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate the frequency or establish a causal relationship to drug exposure.
Gastrointestinal disorders: Gastrointestinal perforation [see Warnings and Precautions (5.1)].
-
7 DRUG INTERACTIONS
Table 3 includes drugs with clinically important drug interactions with SYMPROIC and instructions for preventing or managing the interaction.
Table 3: Clinically Relevant Interactions Affecting Naldemedine When Co-Administered with Other Drugs Strong CYP3A Inducers (e.g., rifampin, carbamazepine, phenytoin, St. John's Wort) Clinical Impact Significant decrease in plasma naldemedine concentrations, which may reduce efficacy [see Clinical Pharmacology (12.3)] Intervention Avoid use of SYMPROIC with strong CYP3A inducers. Other Opioid Antagonists Clinical Impact Potential for additive effect of opioid receptor antagonism and increased risk of opioid withdrawal. Intervention Avoid use of SYMPROIC with another opioid antagonist. Moderate (e.g., fluconazole, atazanavir, aprepitant, diltiazem, erythromycin) and Strong (e.g., itraconazole, ketoconazole, clarithromycin, ritonavir, saquinavir) CYP3A Inhibitors Clinical Impact Increase in plasma naldemedine concentrations [see Clinical Pharmacology (12.3)] Intervention Monitor for potential naldemedine-related adverse reactions [see Adverse Reactions (6.1)]. P-glycoprotein (P-gp) Inhibitors (e.g., amiodarone, captopril, cyclosporine, quercetin, quinidine, verapamil) Clinical Impact Increase in plasma naldemedine concentrations [see Clinical Pharmacology (12.3)] Intervention Monitor for potential naldemedine-related adverse reactions [see Adverse Reactions (6.1)]. -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data with naldemedine in pregnant women to inform a drug-associated risk of major birth defects and miscarriage. There is a potential for opioid withdrawal in a fetus when SYMPROIC is used in pregnant women [see Clinical Considerations]. SYMPROIC should be used during pregnancy only if the potential benefit justifies the potential risk.
In a rat embryo-fetal development study following oral administration of naldemedine during the period of organogenesis at doses resulting in systemic exposure approximately 23,000 times the human area under the plasma-concentration time curve (AUC) at the recommended human dose of 0.2 mg/day, no developmental abnormalities were observed. In rabbits, there were no adverse effects on embryo-fetal development following oral administration of naldemedine during the period of organogenesis at doses resulting in systemic exposure approximately 226 times the human AUC at the recommended human dose of 0.2 mg/day [see Data]. No effects on pre- and postnatal development were observed in rats at exposures 12 times human exposures at the recommended human dose.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
In rats, there were no adverse effects on embryo-fetal development following oral administration of naldemedine during the period of organogenesis at doses up to 1000 mg/kg/day (approximately 23,000 times the human exposures (AUC) at the recommended human dose). In rabbits, there were no adverse effects on embryo-fetal development following oral administration of naldemedine during the period of organogenesis at doses up to 100 mg/kg/day (approximately 226 times the human exposures (AUC) at the recommended human dose). At 400 mg/kg/day (approximately 844 times the human exposures (AUC) at the recommended human dose), effects in maternal animals included body weight loss/decreased body weight gain and food consumption, fetal loss, and premature delivery. Decreased fetal body weights at this dose may be related to the maternal toxicity observed.
In the pre- and postnatal development study, pregnant rats were administered naldemedine at oral doses up to 1000 mg/kg/day from gestation day 7 through lactation day 20. No effects on pre- and postnatal development were observed in rats at 1 mg/kg/day (approximately 12 times the human exposures (AUC) at the recommended human dose). A single dam died at parturition at 1000 mg/kg/day, and decreased body weights/body weight gain and food consumption, poor nursing, and total litter loss were noted at 30 and 1000 mg/kg/day (approximately 626 and 17,000 times the human exposures (AUC) at the recommended human dose, respectively). Decreases in the offspring viability index on Day 4 after birth were noted at 30 and 1000 mg/kg/day, and low body weights and delayed pinna unfolding in pups were noted at 1000 mg/kg/day.
8.2 Lactation
Risk Summary
There is no information regarding the presence of naldemedine in human milk, the effects on the breastfed infant, or the effects on milk production. Naldemedine was present in the milk of rats [see Data]. Because of the potential for serious adverse reactions, including opioid withdrawal in breastfed infants, a decision should be made to discontinue breastfeeding or discontinue the drug, taking into account the importance of the drug to the mother. If drug is discontinued in order to minimize drug exposure to a breastfed infant, advise women that breastfeeding may be resumed 3 days after the final dose of SYMPROIC.
8.4 Pediatric Use
The safety and effectiveness of SYMPROIC have not been established in pediatric patients.
8.5 Geriatric Use
Of the 1163 patients exposed to SYMPROIC in clinical studies, 183 (16%) were 65 years of age and over, while 37 (3%) were 75 years and over. No overall differences in safety or effectiveness between these and younger patients were observed, but greater sensitivity of some older individuals cannot be ruled out. In a population pharmacokinetic analysis, no age-related alterations in the pharmacokinetics of naldemedine were observed [see Clinical Pharmacology (12.3)].
-
8.6 Hepatic Impairment
The effect of severe hepatic impairment (Child-Pugh Class C) on the pharmacokinetics of naldemedine has not been evaluated. Avoid use of SYMPROIC in patients with severe hepatic impairment. No dose adjustment of SYMPROIC is required in patients with mild or moderate hepatic impairment [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
Single doses of naldemedine up to 100 mg (500 times the recommended dose) and multiple doses of up to 30 mg (150 times the recommended dose) for 10 days have been administered to healthy subjects in clinical studies. Dose-dependent increases in gastrointestinal-related adverse reactions, including abdominal pain, diarrhea, and nausea, were observed.
Single doses of naldemedine up to 3 mg (15 times the recommended dose) and multiple doses of 0.4 mg (twice the recommended dose) for 28 days have been administered to patients with OIC in clinical studies. Dose-dependent increases in gastrointestinal-related adverse reactions, including abdominal pain, diarrhea, nausea, and vomiting, were observed. Also, chills, hyperhidrosis, and dizziness were reported more frequently at 1 and 3 mg doses and hyperhidrosis at the 0.4 mg dose.
No antidote for naldemedine is known. Hemodialysis is not an effective means to remove naldemedine from the blood [see Clinical Pharmacology (12.3)].
-
11 DESCRIPTION
SYMPROIC (naldemedine), an opioid antagonist, contains naldemedine tosylate as the active ingredient.
The chemical name for naldemedine tosylate is: 17-(cyclopropylmethyl)-6,7-didehydro-4,5α-epoxy-3,6,14-trihydroxy-N-[2-(3-phenyl-1,2,4-oxadiazol-5-yl)propan-2-yl]morphinan-7-carboxamide 4-methylbenzenesulfonic acid.
The structural formula is:

The empirical formula for naldemedine tosylate is C32H34N4O6∙C7H8O3S and the molecular weight is 742.84.
Naldemedine tosylate is a white to light tan powder, soluble in dimethylsulfoxide and methanol, slightly soluble in alcohol and water, and independent of pH.
SYMPROIC (naldemedine) tablets for oral use contain 0.2 mg naldemedine (equivalent to 0.26 mg of naldemedine tosylate).
Excipients are: D-mannitol, croscarmellose sodium, magnesium stearate, hypromellose, talc, and yellow ferric oxide.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Naldemedine is an opioid antagonist with binding affinities for mu-, delta-, and kappa-opioid receptors. Naldemedine functions as a peripherally-acting mu-opioid receptor antagonist in tissues such as the gastrointestinal tract, thereby decreasing the constipating effects of opioids.
Naldemedine is a derivative of naltrexone to which a side chain has been added that increases the molecular weight and the polar surface area, thereby reducing its ability to cross the blood-brain barrier (BBB). Naldemedine is also a substrate of the P-glycoprotein (P-gp) efflux transporter. Based on these properties, the CNS penetration of naldemedine is expected to be negligible at the recommended dose levels, limiting the potential for interference with centrally-mediated opioid analgesia.
12.2 Pharmacodynamics
Use of opioids induces slowing of gastrointestinal motility and transit. Antagonism of gastrointestinal mu-opioid receptors by naldemedine inhibits opioid-induced delay of gastrointestinal transit time.
12.3 Pharmacokinetics
Absorption
Following oral administration, naldemedine is absorbed with a time to achieve peak concentrations (Tmax) of approximately 0.75 hours in a fasted state. Across the range of doses evaluated, the maximum plasma concentration (Cmax) and area under the plasma concentration-time curve (AUC) increased in a dose-proportional or almost dose-proportional manner. Accumulation was minimal following multiple daily doses of naldemedine.
Food Effect
A high-fat meal decreased the rate but not the extent of naldemedine absorption. The Cmax was decreased by approximately 35% and time to achieve Cmax was delayed from 0.75 hours in the fasted state to 2.5 hours in the fed state, whereas there was no meaningful change in the AUC in the fed state [see Dosage and Administration (2.2)].
Distribution
Plasma protein binding of naldemedine in humans is 93% to 94%. The mean apparent volume of distribution during the terminal phase (Vz/F) is 155 L.
Elimination
The terminal elimination half-life of naldemedine is 11 hours.
Metabolism
Naldemedine is primarily metabolized by CYP3A to nor-naldemedine, with minor contribution from UGT1A3 to form naldemedine 3-G. Nor-naldemedine and naldemedine 3-G have been shown to have antagonistic activity for opioid receptors, with less potent effect than naldemedine.
Following oral administration of [14C]-labeled naldemedine, the primary metabolite in plasma was nor-naldemedine, with a relative exposure compared to naldemedine of approximately 9% to 13%. Naldemedine 3-G was a minor metabolite in plasma, with a relative exposure to naldemedine of less than 3%.
Naldemedine also undergoes cleavage in the GI tract to form benzamidine and naldemedine carboxylic acid.
Excretion
Following oral administration of [14C]-labeled naldemedine, the total amount of radioactivity excreted in the urine and feces was 57% and 35% of the administered dose of naldemedine, respectively. The amount of naldemedine excreted unchanged in the urine was approximately 16% to 18% of the administered dose. Benzamidine was the most predominant metabolite excreted in the urine and feces, representing approximately 32% and 20% of the administered dose of naldemedine, respectively. The percentage of unchanged drug in feces has not been estimated.
Use in Specific Populations
Age: Geriatric Population, Sex, Race/Ethnicity
A population pharmacokinetic analysis from clinical studies with naldemedine did not identify a clinically meaningful effect of age, sex, or race on the pharmacokinetics of naldemedine.
Renal Impairment
The pharmacokinetics of naldemedine after administration of a 0.2 mg single oral dose of SYMPROIC was studied in subjects with mild (n=8, estimated glomerular filtration rate [eGFR] of 60 to 89 mL/min/1.73 m2), moderate (n=8, eGFR 30 to 59 mL/min/1.73 m2), and severe (n=6, eGFR less than 30 mL/min/1.73 m2) renal impairment, and subjects with end-stage renal disease (ESRD) requiring hemodialysis (n=8), and compared to healthy subjects with normal renal function (n=8, estimated creatinine clearance of at least 90 mL/min). The pharmacokinetics of naldemedine between subjects in all groups were similar.
Plasma concentrations of naldemedine in subjects with ESRD requiring hemodialysis were similar when SYMPROIC was administered either pre- or post-hemodialysis, indicating that naldemedine was not removed from the blood by hemodialysis.
Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of a 0.2 mg single oral dose of SYMPROIC was studied in subjects with hepatic impairment classified as mild (n=8, Child-Pugh Class A) or moderate (n=8, Child-Pugh Class B) and compared with healthy subjects with normal hepatic function (n=8). The pharmacokinetics of naldemedine between subjects in all groups were similar.
The effect of severe hepatic impairment (Child-Pugh Class C) on the pharmacokinetics of naldemedine was not evaluated [see Use in Specific Populations (8.6)].
Drug Interaction Studies
Effect of Naldemedine on Other Drugs
In in vitro studies at clinically relevant concentrations, naldemedine did not inhibit the major CYP enzymes (including CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1, CYP3A4/5, or CYP4A11 isozymes) and is not an inhibitor of transporters (including OATP1B1, OATP1B3, OCT1, OCT2, OAT1, OAT3, BCRP, or P-gp). Naldemedine did not cause significant induction of CYP1A2, CYP2B6, CYP3A4, UGT1A2, UGT1A6, or UGT2B7 isozymes.
Effect of Other Drugs on Naldemedine
Naldemedine is primarily metabolized by the CYP3A4 enzyme with a minor contribution from UGT1A3. Naldemedine is a substrate of P-gp. The effects of co-administered drugs on the pharmacokinetics of naldemedine are summarized in Figure 1.
Figure 1: Effect of Co-Administered Drugs on the Pharmacokinetics of Naldemedine
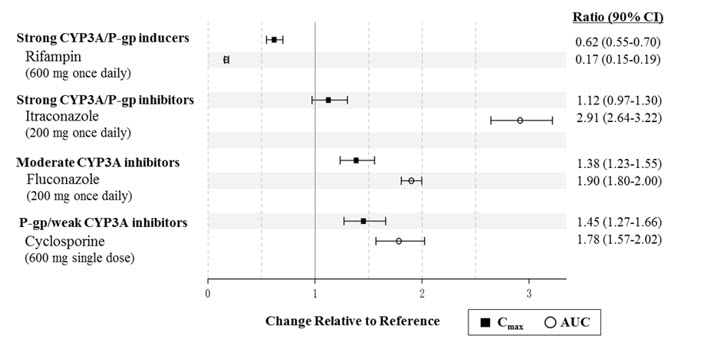
Efavirenz (moderate CYP3A inducer): Simulation using physiologically-based pharmacokinetic modeling suggested that concomitant use of efavirenz decreases exposure to naldemedine by 43%. The clinical consequence of this decreased exposure is unknown.
No drug interaction studies have been conducted for SYMPROIC with drugs that alter gastric pH (e.g., antacids, proton-pump inhibitors).
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
In 2-year carcinogenicity studies, there were no drug-related neoplastic findings following oral administration of naldemedine to mice and rats at doses up to 100 mg/kg/day (approximately 17,500 and 6,300 times the human exposures (AUC) at the recommended human dose, respectively).
Mutagenesis
Naldemedine was not genotoxic in the in vitro bacterial reverse mutation (Ames) assay, a chromosomal aberration assay with cultured Chinese hamster lung cells, or an in vivo micronucleus assay with rat bone marrow cells.
Impairment of Fertility
Naldemedine was found to have no effect on fertility or reproductive performance in male and female rats at oral doses up to 1000 mg/kg/day (approximately 17,000 times the human exposures (AUC) at the recommended human dose). In female rats, prolongation of diestrous phase was noted at 10 mg/kg/day (approximately 179 times the human exposures (AUC) at the recommended human dose).
-
14 CLINICAL STUDIES
SYMPROIC was evaluated in two replicate, 12-week, randomized, double-blind, placebo-controlled trials (Study 1 and Study 2) in which SYMPROIC was used without laxatives in patients with OIC and chronic non-cancer pain.
Patients receiving a stable opioid morphine equivalent daily dose of at least 30 mg for at least 4 weeks before enrollment and self-reported OIC were eligible for clinical trial participation.
Patients with evidence of significant structural abnormalities of the GI tract were not enrolled in these trials.
In Studies 1 and 2, patients had to either be not using laxatives or willing to discontinue laxative use at the time of screening and willing to use only the provided rescue laxatives during the screening and treatment periods.
In Studies 1 and 2, OIC was confirmed through a two-week run in period and was defined as no more than 4 spontaneous bowel movements (SBMs) total over 14 consecutive days and less than 3 SBMs in a given week with at least 25% of the SBMs associated with one or more of the following conditions: (1) straining; (2) hard or lumpy stools; (3) having a sensation of incomplete evacuation; and (4) having a sensation of anorectal obstruction/blockage.
An SBM was defined as a bowel movement (BM) without rescue laxative taken within the past 24 hours. Patients with no BMs over the 7 consecutive days prior to and during the 2-week screening period or patients who had never taken laxatives were excluded.
In the screening and treatment periods, bisacodyl was used as rescue laxative if patients had not had a BM for 72 hours and were allowed one-time use of an enema, if after 24 hours of taking bisacodyl they still had not had a BM.
A total of 547 patients in Study 1 and 553 patients in Study 2 were randomized in a 1:1 ratio to receive SYMPROIC 0.2 mg once daily or placebo for 12 weeks. Study medication was administered without regard to meals.
The mean age of subjects in Studies 1 and 2 was 54 years; 59% were women; and 80% were white. The most common types of pain in Studies 1 and 2 were back or neck pain (61%). The mean baseline number of SBMs was 1.3 and 1.2 per week for Studies 1 and 2, respectively.
Prior to enrollment, patients were using their current opioid for a mean duration of approximately 5 years. A wide range of types of opioids were used. The mean baseline opioid morphine equivalent daily dosage was 132 mg and 121 mg per day for Studies 1 and 2, respectively.
The efficacy of SYMPROIC was assessed in Studies 1 and 2 using a responder analysis. A responder was defined as a patient who had at least 3 SBMs per week and a change from baseline of at least 1 SBM per week for at least 9 out of the 12 weeks and 3 out of the last 4 weeks in Studies 1 and 2.
The responder rates in Studies 1 and 2 are shown in Table 4.
Table 4: Efficacy Responder Rates in Studies 1 and 2 in Patients with OIC and Chronic Non-Cancer Pain Study 1 Study 2 SYMPROIC
0.2 mg once daily
(N=273)Placebo
(N=272)Treatment Difference
[95% CI]SYMPROIC
0.2 mg once daily
(N=276)Placebo
(N=274)Treatment Difference
[95% CI]CI=Confidence Interval - *
- The primary endpoint was defined as a patient who had at least 3 SBMs per week and a change from baseline of at least 1 SBM per week for at least 9 out of the 12 study weeks and 3 out of the last 4 weeks.
- †
- Cochran-Mantel-Haenszel test adjusted for opioid dose strata (30 to 100 mg; greater than 100 mg)
Responder* 130 (48%) 94 (35%) 13%
[5%, 21%]145 (53%) 92 (34%) 19%
[11%, 27%]p value† 0.0020 <0.0001 In Studies 1 and 2, the mean increase in frequency of SBMs per week from baseline to the last 2 weeks of the 12-week treatment period was 3.1 for SYMPROIC vs. 2.0 for placebo (difference 1.0, 95% CI 0.6, 1.5), and 3.3 for SYMPROIC vs. 2.1 for placebo (difference 1.2, 95% CI 0.8, 1.7), respectively.
During week 1 of the treatment period, the mean increase in frequency of SBMs per week from baseline was 3.3 for SYMPROIC vs. 1.3 for placebo (difference 2.0, 95% CI 1.5, 2.5) in Study 1 and 3.7 for SYMPROIC vs. 1.6 for placebo (difference 2.1, 95% CI 1.5, 2.6) in Study 2.
The mean increase in the frequency of complete SBM (CSBM) per week from baseline to the last 2 weeks of 12-week treatment period was 2.3 for SYMPROIC vs. 1.5 for placebo (difference 0.8, 95% CI 0.4, 1.2) in Study 1 and 2.6 for SYMPROIC vs. 1.6 for placebo (difference 1.1, 95% CI 0.6, 1.5) in Study 2. A CSBM was defined as a SBM that was associated with a sense of complete evacuation.
The change in the frequency of SBMs without straining per week from baseline to the last 2 weeks of the treatment period was 1.3 for SYMPROIC vs. 0.7 for placebo (difference 0.6, 95% CI 0.2, 0.9) in Study 1 and 1.8 for SYMPROIC vs. 1.1 for placebo (difference 0.7, 95% CI 0.3, 1.2) in Study 2.
- 16 HOW SUPPLIED/STORAGE AND HANDLING
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Administration
Advise patients to discontinue SYMPROIC if treatment with the opioid pain medication is also discontinued.
Gastrointestinal Perforation
Advise patients to discontinue SYMPROIC and to promptly seek medical attention if they develop unusually severe, persistent or worsening abdominal pain [see Warnings and Precautions (5.1)].
Opioid Withdrawal
Advise patients that clusters of symptoms consistent with opioid withdrawal may occur while taking SYMPROIC and to contact their healthcare provider if these symptoms occur [see Warnings and Precautions (5.2)].
Pregnancy
Advise females of reproductive potential, who become pregnant or are planning to become pregnant, that the use of SYMPROIC during pregnancy may precipitate opioid withdrawal in a fetus due to the undeveloped blood-brain barrier [see Use in Specific Populations (8.1)].
Lactation
Advise women that breastfeeding is not recommended during treatment with SYMPROIC and for 3 days after the final dose [see Use in Specific Populations (8.2)].
- SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDE
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: 07/2025 MEDICATION GUIDE
SYMPROIC® (sim proe' ik)
(naldemedine) tablets, for oral useWhat is the most important information I should know about SYMPROIC?
SYMPROIC may cause serious side effects, including: - Tear in your stomach or intestinal wall (perforation). Stomach pain that is severe can be a sign of a serious medical condition. If you get stomach pain that does not go away, stop taking SYMPROIC and get emergency medical help right away.
- Opioid withdrawal. You may have symptoms of opioid withdrawal during treatment with SYMPROIC including sweating, chills, tearing, warm or hot feeling to your face (flush), sneezing, fever, feeling cold, abdominal pain, diarrhea, nausea, and vomiting. Tell your healthcare provider if you have any of these symptoms.
What is SYMPROIC? SYMPROIC is a prescription medicine used to treat constipation that is caused by prescription pain medicines called opioids in adults with long-lasting (chronic) pain that is not caused by active cancer.
It is not known if SYMPROIC is safe and effective in children.Do not take SYMPROIC if you: - have a bowel blockage (intestinal obstruction) or have a history of bowel blockage.
- are allergic to SYMPROIC or any of the ingredients in SYMPROIC. See the end of this Medication Guide for a complete list of ingredients in SYMPROIC. Tell your healthcare provider or pharmacist before you start or stop any medicines during treatment with SYMPROIC.
Before you take SYMPROIC, tell your healthcare provider about all of your medical conditions, including if you: - have any stomach or bowel (intestines) problems, including, cancer or past surgery of the stomach or bowel, diverticulitis, or had chemotherapy or radiation treatment..
- have liver problems.
- are pregnant or plan to become pregnant. Taking SYMPROIC during pregnancy may cause opioid withdrawal symptoms in your unborn baby. Tell your healthcare provider right away if you become pregnant during treatment with SYMPROIC.
- are breastfeeding or plan to breastfeed. It is not known if SYMPROIC passes into your breast milk. You should not breastfeed during treatment with SYMPROIC and for 3 days after your last dose. Taking SYMPROIC while you are breastfeeding may cause opioid withdrawal symptoms in your baby. You and your healthcare provider should decide if you will take SYMPROIC or breastfeed. You should not do both.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Other medicines may affect the way SYMPROIC works. How should I take SYMPROIC? - Take SYMPROIC exactly as your healthcare provider tells you to take it.
- Take your prescribed dose of SYMPROIC 1 time each day.
- SYMPROIC can be taken with or without food.
- SYMPROIC has been shown to be effective in people who have taken opioid pain medicines for at least 4 weeks.
- Tell your healthcare provider if you stop taking your opioid pain medicine. If you stop taking your opioid pain medicine, you should also stop taking SYMPROIC.
What are the possible side effects of SYMPROIC? See "What is the most important information I should know about SYMPROIC?" The most common side effects of SYMPROIC include stomach (abdomen) pain, diarrhea, nausea and vomiting (gastroenteritis).
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of SYMPROIC. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store SYMPROIC? - Store SYMPROIC at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep SYMPROIC in the bottle that it comes in.
Keep SYMPROIC and all medicines out of the reach of children. General information about the safe and effective use of SYMPROIC. Medicines are sometimes prescribed for purposes other than those in a Medication Guide. Do not take SYMPROIC for a condition for which it was not prescribed. Do not give SYMPROIC to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about SYMPROIC that is written for health professionals. What are the ingredients in SYMPROIC? Active Ingredient: naldemedine tosylate Inactive ingredients: D-mannitol, croscarmellose sodium, magnesium stearate, hypromellose, talc, and yellow ferric oxide.
Manufactured for: Collegium Pharmaceutical, Inc. Stoughton, MA 02072SYM-001-MG-JULY2025
SYMPROIC is a registered trademark of Shionogi & Co., Ltd. For more information, go to www.symproic.com or call 1-800-469-0261. - PRINCIPAL DISPLAY PANEL - 0.2 mg Tablet Bottle Label - 30 Tablets
-
PRINCIPAL DISPLAY PANEL - 0.2 mg Tablet Blister Pack
7 Tablets
Each tablet contains 0.2 mg of naldemedine
(equivalent to 0.26 mg of naldemedine tosylate).
Usual adult dosage: See package insert.Store at 20°C to 25°C (68°F to 77°F).
Mfd. for BioDelivery Sciences International, Inc.
Raleigh, NC 27612Symproic®
(naldemedine) tablets
0.2 mgNDC 59385-041-07 Rx ONLY
Keep out of reach of children.
PHYSICIAN SAMPLES NOT FOR SALE
319623 04/19 SYM-0001.1bDAY 1
DAY 2
DAY 3
DAY 4
DAY 5
DAY 6
DAY 7LOT:
EXP:
-
PRINCIPAL DISPLAY PANEL - 0.2 mg Tablet Blister Pack Carton
NDC 59385-041-07
7 Tablets
Dosage: Take 1 tablet
once dailyEach tablet contains 0.2 mg of
naldemedine (equivalent to
0.26 mg of naldemedine tosylate).Please see the included
Prescribing Information.Keep out of reach of children.
Dispense the enclosed Medication Guide
to each patient.Symproic®
(naldemedine) tablets
0.2 mgPHYSICIAN SAMPLES NOT FOR SALE
FOR ORAL USE ONLY
Rx ONLY
-
INGREDIENTS AND APPEARANCE
SYMPROIC
naldemedine tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:59385-041 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength naldemedine tosylate (UNII: V1N8F1RVVO) (naldemedine - UNII:03KSI6WLXH) naldemedine 0.2 mg Inactive Ingredients Ingredient Name Strength Mannitol (UNII: 3OWL53L36A) Croscarmellose Sodium (UNII: M28OL1HH48) Magnesium Stearate (UNII: 70097M6I30) Hypromellose, Unspecified (UNII: 3NXW29V3WO) Talc (UNII: 7SEV7J4R1U) Ferric Oxide Yellow (UNII: EX438O2MRT) Product Characteristics Color YELLOW Score no score Shape ROUND Size 7mm Flavor Imprint Code 222;0;2 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59385-041-30 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/14/2019 2 NDC:59385-041-07 1 in 1 CARTON 07/01/2020 07/31/2024 2 7 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208854 06/14/2019 Labeler - BioDelivery Sciences International Inc (016058955)